Abstract
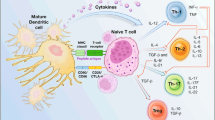
Glioblastomas are characterized by immunosuppression, rapid proliferation, angiogenesis, and invasion into the surrounding brain parenchyma. Limitations in current therapeutic approaches have spurred the development of personalized, patient-specific treatments. Among these, active immunotherapy has emerged as a viable option for glioma treatment. The ability to generate an immune response utilizing patient-derived dendritic cells (DCs) (professional antigen-presenting cells) is especially attractive. This approach to glioma treatment allows for the immunologic targeting and destruction of malignant cells. Data acquired in multiple pre-clinical models and clinical trials have shown significant responses and prolonged survival. Here we provide an overview of the current status of DC vaccination for the treatment of gliomas.



Similar content being viewed by others
References
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ et al (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–996
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144:646–674
Yamanaka R, Kajiwara K (2012) Dendritic cell vaccines. Adv Exp Med Biol 746:187–200
McLendon R, Friedman A, Bigner D, Van Meir EG, Brat DJ, Mastrogianakis GM et al (2008) Comprehensive genomic characterization defines human glioblastoma genes and core pathways. Nature 455:1061–1068
Trumpfheller C, Longhi MP, Caskey M, Idoyaga J, Bozzacco L, Keler T et al (2012) Dendritic cell-targeted protein vaccines: a novel approach to induce T-cell immunity. J Intern Med 271:183–192
Steinman RM, Idoyaga J (2010) Features of the dendritic cell lineage. Immunol Rev 234:5–17
Steinman RM, Banchereau J (2007) Taking dendritic cells into medicine. Nature 449:419–426
Shurin MR, Salter RD (2009) Dendritic cells in cancer. Springer, New York
Liu K, Nussenzweig MC (2010) Origin and development of dendritic cells. Immunol Rev 234:45–54
Liau LM, Prins RM, Kiertscher SM, Odesa SK, Kremen TJ, Giovannone AJ et al (2005) Dendritic cell vaccination in glioblastoma patients induces systemic and intracranial T-cell responses modulated by the local central nervous system tumor microenvironment. Clin Cancer Res 11:5515–5525
Prins RM, Soto H, Konkankit V, Odesa SK, Eskin A, Yong WH et al (2011) Gene expression profile correlates with T-cell infiltration and relative survival in glioblastoma patients vaccinated with dendritic cell immunotherapy. Clin Cancer Res 17:1603–1615
Caruso DA, Orme LM, Neale AM, Radcliff FJ, Amor GM, Maixner W et al (2004) Results of a phase 1 study utilizing monocyte-derived dendritic cells pulsed with tumor RNA in children and young adults with brain cancer. Neuro-oncology 6:236–246
Tacken PJ, De Vries IJM, Torensma R, Figdor CG (2007) Dendritic-cell immunotherapy: from ex vivo loading to in vivo targeting. Nat Rev Immunol 7:790–802
Kalinski P, Okada H (2010) Polarized dendritic cells as cancer vaccines: directing effector-type T cells to tumors. Semin Immunol 22(3):173–182
Okada H, Kalinski P, Ueda R, Hoji A, Kohanbash G, Donegan TE et al (2011) Induction of CD8+ T-cell responses against novel glioma-associated antigen peptides and clinical activity by vaccinations with alpha-type 1 polarized dendritic cells and polyinosinic–polycytidylic acid stabilized by lysine and carboxymethylcellulose in patients with recurrent malignant glioma. J Clin Oncol 29:330–336
Liau LM, Black KL, Martin NA, Sykes SN, Bronstein JM, Jouben-Steele L et al (2000) Treatment of a patient by vaccination with autologous dendritic cells pulsed with allogeneic major histocompatibility complex class I-matched tumor peptides. Case Report. Neurosurg Focus 9:e8
Prins RM, Wang X, Soto H, Young E, Lisiero DN, Fong B et al (2013) Comparison of glioma-associated antigen peptide-loaded versus autologous tumor lysate-loaded dendritic cell vaccination in malignant glioma patients. J Immunother 36:152–157
Yu JS, Wheeler CJ, Zeltzer PM, Ying H, Finger DN, Lee PK et al (2001) Vaccination of malignant glioma patients with peptide-pulsed dendritic cells elicits systemic cytotoxicity and intracranial T-cell infiltration. Cancer Res 61:842–847
Polyzoidis S, Ashkan K (2014) DCVax(R)-L-developed by Northwest Biotherapeutics. Hum Vaccine Immunother 10:3139–3145
Jouanneau E, Black KL, Veiga L, Cordner R, Goverdhana S, Zhai Y et al (2014) Intrinsically de-sialylated CD103(+) CD8 T cells mediate beneficial anti-glioma immune responses. Cancer Immunol Immunother 63:911–924
Wheeler CJ, Black KL, Liu G, Mazer M, Zhang XX, Pepkowitz S et al (2008) Vaccination elicits correlated immune and clinical responses in glioblastoma multiforme patients. Cancer Res 68:5955–5964
De Vleeschouwer S, Fieuws S, Rutkowski S, Van Calenbergh F, Van Loon J, Goffin J et al (2008) Postoperative adjuvant dendritic cell-based immunotherapy in patients with relapsed glioblastoma multiforme. Clin Cancer Res 14:3098–3104
Ardon H, De Vleeschouwer S, Van Calenbergh F, Claes L, Kramm CM, Rutkowski S et al (2010) Adjuvant dendritic cell-based tumour vaccination for children with malignant brain tumours. Pediatr Blood Cancer 54:519–525
Sampson JH, Archer GE, Mitchell DA, Heimberger AB, Herndon JE II, Lally-Goss D et al (2009) An epidermal growth factor receptor variant III-targeted vaccine is safe and immunogenic in patients with glioblastoma multiforme. Mol Cancer Ther 8:2773–2779
Morgan RA, Johnson LA, Davis JL, Zheng Z, Woolard KD, Reap EA et al (2012) Recognition of glioma stem cells by genetically modified T cells targeting EGFRvIII and development of adoptive cell therapy for glioma. Hum Gene Ther 23:1043–1053
Cao JX, Zhang XY, Liu JL, Li D, Li JL, Liu YS et al (2014) Clinical efficacy of tumor antigen-pulsed DC treatment for high-grade glioma patients: evidence from a meta-analysis. PLoS ONE 9:e107173
Butowski N (2010) Immunostimulants for malignant gliomas. Neurosurg Clin N Am 21:53–65
Kamimura K, Suda T, Zhang G, Liu D (2011) Advances in gene delivery systems. Pharm Med 25:293–306
Rainov NG, Heidecke V (2011) Clinical development of experimental virus-mediated gene therapy for malignant glioma. Anti-cancer Agents Med Chem (Former Curr Med Chem Anti-cancer Agents) 11:739–747
Chiocca EA, Smith KM, McKinney B, Palmer CA, Rosenfeld S, Lillehei K et al (2008) A phase I trial of Ad.hIFN-beta gene therapy for glioma. Mol Ther 16:618–626
Hervas-Stubbs S, Perez-Gracia JL, Rouzaut A, Sanmamed MF, Le Bon A, Melero I (2011) Direct effects of type I interferons on cells of the immune system. Clin Cancer Res 17:2619–2627
Pluhar GE, Grogan PT, Seiler C, Goulart M, SantaCruz KS, Carlson C et al (2010) Anti-tumor immune response correlates with neurological symptoms in a dog with spontaneous astrocytoma treated by gene and vaccine therapy. Vaccine 28:3371–3378
Colombo F, Barzon L, Franchin E, Pacenti M, Pinna V, Danieli D et al (2005) Combined HSV-TK/IL-2 gene therapy in patients with recurrent glioblastoma multiforme: biological and clinical results. Cancer Gene Ther 12:835–848
Serre K, Mohr E, Gaspal F, Lane PJ, Bird R, Cunningham AF et al (2010) IL-4 directs both CD4 and CD8 T cells to produce Th2 cytokines in vitro, but only CD4 T cells produce these cytokines in response to alum-precipitated protein in vivo. Mol Immunol 47:1914–1922
Okada H, Lieberman FS, Walter KA, Lunsford LD, Kondziolka DS, Bejjani GK et al (2007) Autologous glioma cell vaccine admixed with interleukin-4 gene transfected fibroblasts in the treatment of patients with malignant gliomas. J Transl Med 5:67
Sonabend AM, Velicu S, Ulasov IV, Han Y, Tyler B, Brem H et al (2008) A safety and efficacy study of local delivery of interleukin-12 transgene by PPC polymer in a model of experimental glioma. Anticancer Drugs 19:133–142
Mitchell DA, Batich KA, Gunn MD, Huang M-N, Sanchez-Perez L, Nair SK et al (2015) Tetanus toxoid and CCL3 improve dendritic cell vaccines in mice and glioblastoma patients. Nature 519:366–369
Zou W (2006) Regulatory T cells, tumour immunity and immunotherapy. Nat Rev Immunol 6:295–307
Ott PA, Hodi FS, Robert C (2013) CTLA-4 and PD-1/PD-L1 blockade: new immunotherapeutic modalities with durable clinical benefit in melanoma patients. Clin Cancer Res 19:5300–5309
Fong B, Jin R, Wang X, Safaee M, Lisiero DN, Yang I et al (2012) Monitoring of regulatory T cell frequencies and expression of CTLA-4 on T cells, before and after DC vaccination, can predict survival in GBM patients. PLoS ONE 7:e32614
Hamid O, Robert C, Daud A, Hodi FS, Hwu W-J, Kefford R et al (2013) Safety and tumor responses with lambrolizumab (anti-PD-1) in melanoma. N Engl J Med 369:134–144
Curran MA, Montalvo W, Yagita H, Allison JP (2010) PD-1 and CTLA-4 combination blockade expands infiltrating T cells and reduces regulatory T and myeloid cells within B16 melanoma tumors. Proc Natl Acad Sci USA 107:4275–4280
Schartner JM, Hagar AR, Van Handel M, Zhang L, Nadkarni N, Badie B (2005) Impaired capacity for upregulation of MHC class II in tumor-associated microglia. Glia 51:279–285
Grauer O, Pschl P, Lohmeier A, Adema GJ, Bogdahn U (2007) Toll-like receptor triggered dendritic cell maturation and IL-12 secretion are necessary to overcome T-cell inhibition by glioma-associated TGF-beta2. J Neurooncol 82:151–161
Ribas A (2012) Tumor immunotherapy directed at PD-1. N Engl J Med 366:2517–2519
Zeng J, See AP, Phallen J, Jackson CM, Belcaid Z, Ruzevick J et al (2013) Anti-PD-1 blockade and stereotactic radiation produce long-term survival in mice with intracranial gliomas. Int J Radiat Oncol Biol Phys 86:343–349
Wainwright DA, Chang AL, Dey M, Balyasnikova IV, Kim C, Tobias AL et al (2014) Durable therapeutic efficacy utilizing combinatorial blockade against IDO, CTLA-4 and PD-L1 in mice with brain tumors. Clin Cancer Res 20(20):5290–5301
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Antonios, J.P., Everson, R.G. & Liau, L.M. Dendritic cell immunotherapy for brain tumors. J Neurooncol 123, 425–432 (2015). https://doi.org/10.1007/s11060-015-1830-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-015-1830-1