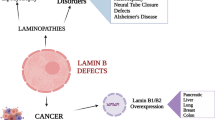
Abstract
The A-type lamins, lamin A and lamin C, generated from a single gene, LMNA, are major structural components of the nuclear lamina. The two alternative splice products have mostly been studied together because they have been considered to be interchangeable. However, several lines of evidence indicate that in spite of being generated from the same gene and having high similarities in their primary sequences, the two isoforms are not equivalent in different biological aspects in both health and disease. The key question is whether they have both overlapping and unique functions and whether they are distinctly regulated. Based on the so far available experimental evidence, lamin A appears to be the most regulated A-type isoform during development, aging, and disease which indicates that lamin A is implicated in many different biological aspects and may have a greater repertoire of specialized functions than lamin C. The aim of this review is to point out differences between the two major LMNA splice variants and the consequences of these differences on their functions. This may guide further research and be of prime importance for the understanding of the pathogenesis of LMNA mutations.




Similar content being viewed by others
References
Afilalo J, Sebag IA, Chalifour LE, Rivas D, Akter R, Sharma K, Duque G (2007) Age-related changes in lamin A/C expression in cardiomyocytes. Am J Physiol Heart Circ Physiol 293(3):H1451–H1456. doi:10.1152/ajpheart.01194.2006
Agrawal S, Pilarski R, Eng C (2005) Different splicing defects lead to differential effects downstream of the lipid and protein phosphatase activities of PTEN. Hum Mol Genet 14(16):2459–2468. doi:10.1093/hmg/ddi246
Al-Saaidi R, Rasmussen TB, Palmfeldt J, Nissen PH, Beqqali A, Hansen J, Bross P (2013) The LMNA mutation p.Arg321Ter associated with dilated cardiomyopathy leads to reduced expression and a skewed ratio of lamin A and lamin C proteins. Exp Cell Res. doi:10.1016/j.yexcr.2013.08.024
Arimura T, Helbling-Leclerc A, Massart C, Varnous S, Niel F, Lacene E, Bonne G (2005) Mouse model carrying H222P-Lmna mutation develops muscular dystrophy and dilated cardiomyopathy similar to human striated muscle laminopathies. Hum Mol Genet 14(1):155–169. doi:10.1093/hmg/ddi017
Beck LA, Hosick TJ, Sinensky M (1990) Isoprenylation is required for the processing of the lamin A precursor. J Cell Biol 110(5):1489–1499
Ben-Harush K, Wiesel N, Frenkiel-Krispin D, Moeller D, Soreq E, Aebi U, Medalia O (2009) The supramolecular organization of the C. elegans nuclear lamin filament. J Mol Biol 386(5):1392–1402. doi:10.1016/j.jmb.2008.12.024
Broers JL, Kuijpers HJ, Ostlund C, Worman HJ, Endert J, Ramaekers FC (2005) Both lamin A and lamin C mutations cause lamina instability as well as loss of internal nuclear lamin organization. Exp Cell Res 304(2):582–592. doi:10.1016/j.yexcr.2004.11.020
Burke B, Stewart CL (2006) The laminopathies: the functional architecture of the nucleus and its contribution to disease. Annu Rev Genomics Hum Genet 7:369–405. doi:10.1146/annurev.genom.7.080505.115732
Candelario J, Sudhakar S, Navarro S, Reddy S, Comai L (2008) Perturbation of wild-type lamin A metabolism results in a progeroid phenotype. Aging Cell 7(3):355–367. doi:10.1111/j.1474-9726.2008.00393.x
Capanni C, Del Coco R, Squarzoni S, Columbaro M, Mattioli E, Camozzi D, Lattanzi G (2008) Prelamin A is involved in early steps of muscle differentiation. Exp Cell Res 314(20):3628–3637. doi:10.1016/j.yexcr.2008.09.026
Cenni V, Sabatelli P, Mattioli E, Marmiroli S, Capanni C, Ognibene A, Lattanzi G (2005) Lamin A N-terminal phosphorylation is associated with myoblast activation: impairment in Emery-Dreifuss muscular dystrophy. J Med Genet 42(3):214–220. doi:10.1136/jmg.2004.026112
Coffinier C, Jung HJ, Li Z, Nobumori C, Yun UJ, Farber EA, Young SG (2010) Direct synthesis of lamin A, bypassing prelamin A processing, causes misshapen nuclei in fibroblasts but no detectable pathology in mice. J Biol Chem 285(27):20818–20826. doi:10.1074/jbc.M110.128835
Dagenais A, LeMyre A, Bibor-Hardy V (1990) Differential transport and integration into the nuclear lamina for lamins A, B, and C. Biochem Cell Biol 68(5):827–831
De Sandre-Giovannoli A, Bernard R, Cau P, Navarro C, Amiel J, Boccaccio I, Levy N (2003) Lamin A truncation in Hutchinson-Gilford progeria. Science 300(5628):2055. doi:10.1126/science.1084125
Dechat T, Gotzmann J, Stockinger A, Harris CA, Talle MA, Siekierka JJ, Foisner R (1998) Detergent-salt resistance of LAP2alpha in interphase nuclei and phosphorylation-dependent association with chromosomes early in nuclear assembly implies functions in nuclear structure dynamics. EMBO J 17(16):4887–4902. doi:10.1093/emboj/17.16.4887
Dhe-Paganon S, Werner ED, Chi YI, Shoelson SE (2002) Structure of the globular tail of nuclear lamin. J Biol Chem 277(20):17381–17384. doi:10.1074/jbc.C200038200
Dittmer TA, Misteli T (2011) The lamin protein family. Genome Biol 12(5):222. doi:10.1186/gb-2011-12-5-222
Duque G, Rivas D (2006) Age-related changes in lamin A/C expression in the osteoarticular system: laminopathies as a potential new aging mechanism. Mech Ageing Dev 127(4):378–383. doi:10.1016/j.mad.2005.12.007
Eckersley-Maslin MA, Bergmann JH, Lazar Z, Spector DL (2013) Lamin A/C is expressed in pluripotent mouse embryonic stem cells. Nucleus 4(1):53–60. doi:10.4161/nucl.23384
Erber A, Riemer D, Hofemeister H, Bovenschulte M, Stick R, Panopoulou G, Weber K (1999) Characterization of the hydra lamin and its gene: a molecular phylogeny of metazoan lamins. J Mol Evol 49(2):260–271
Eriksson M, Brown WT, Gordon LB, Glynn MW, Singer J, Scott L, Collins FS (2003) Recurrent de novo point mutations in lamin A cause Hutchinson-Gilford progeria syndrome. Nature 423(6937):293–298. doi:10.1038/nature01629
Fatkin D, MacRae C, Sasaki T, Wolff MR, Porcu M, Frenneaux M, McDonough B (1999) Missense mutations in the rod domain of the lamin A/C gene as causes of dilated cardiomyopathy and conduction-system disease. N Engl J Med 341(23):1715–1724. doi:10.1056/NEJM199912023412302
Fisher DZ, Chaudhary N, Blobel G (1986) cDNA sequencing of nuclear lamins A and C reveals primary and secondary structural homology to intermediate filament proteins. Proc Natl Acad Sci U S A 83(17):6450–6454
Fong LG, Ng JK, Lammerding J, Vickers TA, Meta M, Cote N, Young SG (2006) Prelamin A and lamin A appear to be dispensable in the nuclear lamina. J Clin Invest 116(3):743–752. doi:10.1172/JCI27125
Foster HA, Stokes P, Forsey K, Leese HJ, Bridger JM (2007) Lamins A and C are present in the nuclei of early porcine embryos, with lamin A being distributed in large intranuclear foci. Chromosom Res 15(2):163–174. doi:10.1007/s10577-006-1088-8
Furukawa K, Inagaki H, Hotta Y (1994) Identification and cloning of an mRNA coding for a germ cell-specific A-type lamin in mice. Exp Cell Res 212(2):426–430. doi:10.1006/excr.1994.1164
Gerace L, Huber MD (2012) Nuclear lamina at the crossroads of the cytoplasm and nucleus. J Struct Biol 177(1):24–31. doi:10.1016/j.jsb.2011.11.007
Gerace L, Comeau C, Benson M (1984) Organization and modulation of nuclear lamina structure. J Cell Sci Suppl 1:137–160
Han X, Feng X, Rattner JB, Smith H, Bose P, Suzuki K, Riabowol K (2008) Tethering by lamin A stabilizes and targets the ING1 tumour suppressor. Nat Cell Biol 10(11):1333–1340. doi:10.1038/ncb1792
Haque F, Lloyd DJ, Smallwood DT, Dent CL, Shanahan CM, Fry AM, Shackleton S (2006) SUN1 interacts with nuclear lamin A and cytoplasmic nesprins to provide a physical connection between the nuclear lamina and the cytoskeleton. Mol Cell Biol 26(10):3738–3751. doi:10.1128/MCB.26.10.3738-3751.2006
Izumi M, Vaughan OA, Hutchison CJ, Gilbert DM (2000) Head and/or CaaX domain deletions of lamin proteins disrupt preformed lamin A and C but not lamin B structure in mammalian cells. Mol Biol Cell 11(12):4323–4337
Johnson BR, Nitta RT, Frock RL, Mounkes L, Barbie DA, Stewart CL, Kennedy BK (2004) A-type lamins regulate retinoblastoma protein function by promoting subnuclear localization and preventing proteasomal degradation. Proc Natl Acad Sci U S A 101(26):9677–9682. doi:10.1073/pnas.0403250101
Jung HJ, Coffinier C, Choe Y, Beigneux AP, Davies BS, Yang SH, Fong LG (2012) Regulation of prelamin A but not lamin C by miR-9, a brain-specific microRNA. Proc Natl Acad Sci U S A 109(7):E423–E431. doi:10.1073/pnas.1111780109
Koch AJ, Holaska JM (2014) Emerin in health and disease. Semin Cell Dev Biol 29C:95–106. doi:10.1016/j.semcdb.2013.12.008
Kolb T, Maass K, Hergt M, Aebi U, Herrmann H (2011) Lamin A and lamin C form homodimers and coexist in higher complex forms both in the nucleoplasmic fraction and in the lamina of cultured human cells. Nucleus 2(5):425–433. doi:10.4161/nucl.2.5.17765
Krimm I, Ostlund C, Gilquin B, Couprie J, Hossenlopp P, Mornon JP, Zinn-Justin S (2002) The Ig-like structure of the C-terminal domain of lamin A/C, mutated in muscular dystrophies, cardiomyopathy, and partial lipodystrophy. Structure 10(6):811–823
Lammerding J, Fong LG, Ji JY, Reue K, Stewart CL, Young SG, Lee RT (2006) Lamins A and C but not lamin B1 regulate nuclear mechanics. J Biol Chem 281(35):25768–25780. doi:10.1074/jbc.M513511200
Liang Y, Chiu PH, Yip KY, Chan SY (2011) Subcellular localization of SUN2 is regulated by lamin A and Rab5. PLoS One 6(5):e20507. doi:10.1371/journal.pone.0020507
Libotte T, Zaim H, Abraham S, Padmakumar VC, Schneider M, Lu W, Karakesisoglou I (2005) Lamin A/C-dependent localization of Nesprin-2, a giant scaffolder at the nuclear envelope. Mol Biol Cell 16(7):3411–3424. doi:10.1091/mbc.E04-11-1009
Lin F, Worman HJ (1993) Structural organization of the human gene encoding nuclear lamin A and nuclear lamin C. J Biol Chem 268(22):16321–16326
Liu B, Ghosh S, Yang X, Zheng H, Liu X, Wang Z, Zhou Z (2012a) Resveratrol rescues SIRT1-dependent adult stem cell decline and alleviates progeroid features in laminopathy-based progeria. Cell Metab 16(6):738–750. doi:10.1016/j.cmet.2012.11.007
Liu Y, Berendsen AD, Jia S, Lotinun S, Baron R, Ferrara N, Olsen BR (2012b) Intracellular VEGF regulates the balance between osteoblast and adipocyte differentiation. J Clin Invest 122(9):3101–3113. doi:10.1172/JCI61209
Lloyd DJ, Trembath RC, Shackleton S (2002) A novel interaction between lamin A and SREBP1: implications for partial lipodystrophy and other laminopathies. Hum Mol Genet 11(7):769–777
Lopez-Mejia IC, de Toledo M, Chavey C, Lapasset L, Cavelier P, Lopez-Herrera C, Tazi J (2014) Antagonistic functions of LMNA isoforms in energy expenditure and lifespan. EMBO Rep 15(5):529–539. doi:10.1002/embr.201338126
Luo YB, Mitrpant C, Johnsen R, Fabian V, Needham M, Fletcher S, Mastaglia FL (2013a) Investigation of splicing changes and post-translational processing of LMNA in sporadic inclusion body myositis. Int J Clin Exp Pathol 6(9):1723–1733
Luo YB, Mitrpant C, Johnsen RD, Fabian VA, Fletcher S, Mastaglia FL, Wilton SD (2013b) Investigation of age-related changes in LMNA splicing and expression of progerin in human skeletal muscles. Int J Clin Exp Pathol 6(12):2778–2786
Machiels BM, Zorenc AH, Endert JM, Kuijpers HJ, van Eys GJ, Ramaekers FC, Broers JL (1996) An alternative splicing product of the lamin A/C gene lacks exon 10. J Biol Chem 271(16):9249–9253
Markiewicz E, Venables R, Mauricio Alvarez R, Quinlan R, Dorobek M, Hausmanowa-Petrusewicz I, Hutchison C (2002) Increased solubility of lamins and redistribution of lamin C in X-linked Emery-Dreifuss muscular dystrophy fibroblasts. J Struct Biol 140(1–3):241–253
McKeon FD, Kirschner MW, Caput D (1986) Homologies in both primary and secondary structure between nuclear envelope and intermediate filament proteins. Nature 319(6053):463–468. doi:10.1038/319463a0
Melcer S, Gruenbaum Y, Krohne G (2007) Invertebrate lamins. Exp Cell Res 313(10):2157–2166. doi:10.1016/j.yexcr.2007.03.004
Miranda M, Chacon MR, Vidal F, Megia A, Richart C, Veloso S, Vendrell J (2007) LMNA messenger RNA expression in highly active antiretroviral therapy-treated HIV-positive patients. J Acquir Immune Defic Syndr 46(4):384–389
Miranda M, Chacon MR, Gutierrez C, Vilarrasa N, Gomez JM, Caubet E, Vendrell J (2008) LMNA mRNA expression is altered in human obesity and type 2 diabetes. Obesity (Silver Spring) 16(8):1742–1748. doi:10.1038/oby.2008.276
Mislow JM, Holaska JM, Kim MS, Lee KK, Segura-Totten M, Wilson KL, McNally EM (2002) Nesprin-1alpha self-associates and binds directly to emerin and lamin A in vitro. FEBS Lett 525(1–3):135–140
Moir RD, Yoon M, Khuon S, Goldman RD (2000) Nuclear lamins A and B1: different pathways of assembly during nuclear envelope formation in living cells. J Cell Biol 151(6):1155–1168
Mory PB, Crispim F, Freire MB, Salles JE, Valerio CM, Godoy-Matos AF, Moises RS (2012) Phenotypic diversity in patients with lipodystrophy associated with LMNA mutations. Eur J Endocrinol 167(3):423–431. doi:10.1530/EJE-12-0268
Motsch I, Kaluarachchi M, Emerson LJ, Brown CA, Brown SC, Dabauvalle MC, Ellis JA (2005) Lamins A and C are differentially dysfunctional in autosomal dominant Emery-Dreifuss muscular dystrophy. Eur J Cell Biol 84(9):765–781. doi:10.1016/j.ejcb.2005.04.004
Mounkes LC, Kozlov SV, Rottman JN, Stewart CL (2005) Expression of an LMNA-N195K variant of A-type lamins results in cardiac conduction defects and death in mice. Hum Mol Genet 14(15):2167–2180. doi:10.1093/hmg/ddi221
Niebroj-Dobosz I, Fidzianska A, Hausmanowa-Petrusewicz I (2003) Expression of emerin and lamins in muscle of patients with different forms of Emery-Dreifuss muscular dystrophy. Acta Myol 22(2):52–57
Nissan X, Blondel S, Navarro C, Maury Y, Denis C, Girard M, Peschanski M (2012) Unique preservation of neural cells in Hutchinson-Gilford progeria syndrome is due to the expression of the neural-specific miR-9 microRNA. Cell Rep 2(1):1–9. doi:10.1016/j.celrep.2012.05.015
Oh YS, Kim DG, Kim G, Choi EC, Kennedy BK, Suh Y, Kim S (2010) Downregulation of lamin A by tumor suppressor AIMP3/p18 leads to a progeroid phenotype in mice. Aging Cell 9(5):810–822. doi:10.1111/j.1474-9726.2010.00614.x
Olsen JV, Vermeulen M, Santamaria A, Kumar C, Miller ML, Jensen LJ, Mann M (2010) Quantitative phosphoproteomics reveals widespread full phosphorylation site occupancy during mitosis. Sci Signal 3(104):ra3. doi:10.1126/scisignal.2000475
Pekovic V, Gibbs-Seymour I, Markiewicz E, Alzoghaibi F, Benham AM, Edwards R, Hutchison CJ (2011) Conserved cysteine residues in the mammalian lamin A tail are essential for cellular responses to ROS generation. Aging Cell 10(6):1067–1079. doi:10.1111/j.1474-9726.2011.00750.x
Peter A, Reimer S (2012) Evolution of the lamin protein family: what introns can tell. Nucleus 3(1):44–59
Prufert K, Vogel A, Krohne G (2004) The lamin CxxM motif promotes nuclear membrane growth. J Cell Sci 117(Pt 25):6105–6116. doi:10.1242/jcs.01532
Pugh GE, Coates PJ, Lane EB, Raymond Y, Quinlan RA (1997) Distinct nuclear assembly pathways for lamins A and C lead to their increase during quiescence in Swiss 3 T3 cells. J Cell Sci 110(Pt 19):2483–2493
Raharjo WH, Enarson P, Sullivan T, Stewart CL, Burke B (2001) Nuclear envelope defects associated with LMNA mutations cause dilated cardiomyopathy and Emery-Dreifuss muscular dystrophy. J Cell Sci 114(Pt 24):4447–4457
Reunert J, Wentzell R, Walter M, Jakubiczka S, Zenker M, Brune T, Marquardt T (2012) Neonatal progeria: increased ratio of progerin to lamin A leads to progeria of the newborn. Eur J Hum Genet 20(9):933–937. doi:10.1038/ejhg.2012.36
Rober RA, Weber K, Osborn M (1989) Differential timing of nuclear lamin A/C expression in the various organs of the mouse embryo and the young animal: a developmental study. Development 105(2):365–378
Sakaki M, Koike H, Takahashi N, Sasagawa N, Tomioka S, Arahata K, Ishiura S (2001) Interaction between emerin and nuclear lamins. J Biochem 129(2):321–327
Sato I, Miyado M, Miwa Y, Sunohara M (2006) Expression of nuclear and mitochondrial thyroid hormone receptors in postnatal rat tongue muscle. Cells Tissues Organs 183(4):195–205. doi:10.1159/000096510
Schirmer EC, Foisner R (2007) Proteins that associate with lamins: many faces, many functions. Exp Cell Res 313(10):2167–2179. doi:10.1016/j.yexcr.2007.03.012
Schirmer EC, Gerace L (2004) The stability of the nuclear lamina polymer changes with the composition of lamin subtypes according to their individual binding strengths. J Biol Chem 279(41):42811–42817. doi:10.1074/jbc.M407705200
Sehgal P, Chaturvedi P, Kumaran RI, Kumar S, Parnaik VK (2013) Lamin A/C haploinsufficiency modulates the differentiation potential of mouse embryonic stem cells. PLoS One 8(2):e57891. doi:10.1371/journal.pone.0057891
Shimi T, Pfleghaar K, Kojima S, Pack CG, Solovei I, Goldman AE, Goldman RD (2008) The A- and B-type nuclear lamin networks: microdomains involved in chromatin organization and transcription. Genes Dev 22(24):3409–3421. doi:10.1101/gad.1735208
Simon DN, Wilson KL (2013) Partners and post-translational modifications of nuclear lamins. Chromosoma 122(1–2):13–31. doi:10.1007/s00412-013-0399-8
Simon DN, Zastrow MS, Wilson KL (2010) Direct actin binding to A- and B-type lamin tails and actin filament bundling by the lamin A tail. Nucleus 1(3):264–272. doi:10.4161/nucl.1.3.11799
Skvortsov S, Schafer G, Stasyk T, Fuchsberger C, Bonn GK, Bartsch G, Huber LA (2011) Proteomics profiling of microdissected low- and high-grade prostate tumors identifies Lamin A as a discriminatory biomarker. J Proteome Res 10(1):259–268. doi:10.1021/pr100921j
Speckman RA, Garg A, Du F, Bennett L, Veile R, Arioglu E, Bowcock AM (2000) Mutational and haplotype analyses of families with familial partial lipodystrophy (Dunnigan variety) reveal recurrent missense mutations in the globular C-terminal domain of lamin A/C. Am J Hum Genet 66(4):1192–1198. doi:10.1086/302836
Stamm S, Ben-Ari S, Rafalska I, Tang Y, Zhang Z, Toiber D, Soreq H (2005) Function of alternative splicing. Gene 344:1–20. doi:10.1016/j.gene.2004.10.022
Stick R (1992) The gene structure of Xenopus nuclear lamin A: a model for the evolution of A-type from B-type lamins by exon shuffling. Chromosoma 101(9):566–574
Strelkov SV, Schumacher J, Burkhard P, Aebi U, Herrmann H (2004) Crystal structure of the human lamin A coil 2B dimer: implications for the head-to-tail association of nuclear lamins. J Mol Biol 343(4):1067–1080. doi:10.1016/j.jmb.2004.08.093
Stuurman N, Heins S, Aebi U (1998) Nuclear lamins: their structure, assembly, and interactions. J Struct Biol 122(1–2):42–66. doi:10.1006/jsbi.1998.3987
Swift J, Harada T, Buxboim A, Shin JW, Tang HY, Speicher DW, Discher DE (2013a) Label-free mass spectrometry exploits dozens of detected peptides to quantify lamins in wildtype and knockdown cells. Nucleus 4(6):450–459. doi:10.4161/nucl.27413
Swift J, Ivanovska IL, Buxboim A, Harada T, Dingal PC, Pinter J, Discher DE (2013b) Nuclear lamin-A scales with tissue stiffness and enhances matrix-directed differentiation. Science 341(6149):1240104. doi:10.1126/science.1240104
Sylvius N, Hathaway A, Boudreau E, Gupta P, Labib S, Bolongo PM, Tesson F (2008) Specific contribution of lamin A and lamin C in the development of laminopathies. Exp Cell Res 314(13):2362–2375. doi:10.1016/j.yexcr.2008.04.017
Tazi J, Bakkour N, Stamm S (2009) Alternative splicing and disease. Biochim Biophys Acta 1792(1):14–26. doi:10.1016/j.bbadis.2008.09.017
Van Berlo JH, Voncken JW, Kubben N, Broers JL, Duisters R, van Leeuwen RE, Pinto YM (2005) A-type lamins are essential for TGF-beta1 induced PP2A to dephosphorylate transcription factors. Hum Mol Genet 14(19):2839–2849. doi:10.1093/hmg/ddi316
Vaughan A, Alvarez-Reyes M, Bridger JM, Broers JL, Ramaekers FC, Wehnert M, Hutchison CJ (2001) Both emerin and lamin C depend on lamin A for localization at the nuclear envelope. J Cell Sci 114(Pt 14):2577–2590
Venables JP (2006) Unbalanced alternative splicing and its significance in cancer. Bioessays 28(4):378–386. doi:10.1002/bies.20390
Wakabayashi T, Mori T, Hirahara Y, Koike T, Kubota Y, Takamori Y, Yamada H (2011) Nuclear lamins are differentially expressed in retinal neurons of the adult rat retina. Histochem Cell Biol 136(4):427–436. doi:10.1007/s00418-011-0853-8
Wang Z, Udeshi ND, Slawson C, Compton PD, Sakabe K, Cheung WD, Hart GW (2010) Extensive crosstalk between O-GlcNAcylation and phosphorylation regulates cytokinesis. Sci Signal 3(104):ra2. doi:10.1126/scisignal.2000526
Weber K, Plessmann U, Traub P (1989) Maturation of nuclear lamin A involves a specific carboxy-terminal trimming, which removes the polyisoprenylation site from the precursor; implications for the structure of the nuclear lamina. FEBS Lett 257(2):411–414
Wegner L, Anthonsen S, Bork-Jensen J, Dalgaard L, Hansen T, Pedersen O, Vaag A (2010) LMNA rs4641 and the muscle lamin A and C isoforms in twins—metabolic implications and transcriptional regulation. J Clin Endocrinol Metab 95(8):3884–3892. doi:10.1210/jc.2009-2675
Willis ND, Cox TR, Rahman-Casans SF, Smits K, Przyborski SA, van den Brandt P, Hutchison CJ (2008) Lamin A/C is a risk biomarker in colorectal cancer. PLoS One 3(8):e2988. doi:10.1371/journal.pone.0002988
Worman HJ, Bonne G (2007) “Laminopathies”: a wide spectrum of human diseases. Exp Cell Res 313(10):2121–2133. doi:10.1016/j.yexcr.2007.03.028
Zhang Y (2011) Biology of the Mi-2/NuRD complex in SLAC (stemness, longevity/ageing, and cancer). Gene Regul Syst Bio 5:1–26. doi:10.4137/GRSB.S6510
Zhang YQ, Sarge KD (2008) Sumoylation regulates lamin A function and is lost in lamin A mutants associated with familial cardiomyopathies. J Cell Biol 182(1):35–39. doi:10.1083/jcb.200712124
Zuo B, Yang J, Wang F, Wang L, Yin Y, Dan J, Liu L (2012) Influences of lamin A levels on induction of pluripotent stem cells. Biol Open 1(11):1118–1127. doi:10.1242/bio.20121586
Acknowledgments
The study was supported by grants from the Institute of Clinical Medicine, Aarhus University Hospital, and the Lundbeck Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Al-Saaidi, R., Bross, P. Do lamin A and lamin C have unique roles?. Chromosoma 124, 1–12 (2015). https://doi.org/10.1007/s00412-014-0484-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00412-014-0484-7