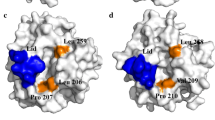
Abstract
Combinatorial libraries of the lid domain of Rhizopus oryzae lipase (ROL; Phe88Xaa, Ala91Xaa, Ile92Xaa) were displayed on the yeast cell surface using yeast cell-surface engineering. Among the 40,000 transformants in which ROL mutants were displayed on the yeast cell surface, ten clones showed clear halos on soybean oil-containing plates. Among these, some clones exhibited high activities toward fatty acid esters of fluorescein and contained non-polar amino acid residues in the mutated positions. Computer modeling of the mutants revealed that hydrophobic interactions between the substrates and amino acid residues in the open form of the lid might be critical for ROL activity. Based on these results, Thr93 and Asp94 were further combinatorially mutated. Among 6,000 transformants, the Thr93Thr, Asp94Ser and Thr93Ser, Asp94Ser transformants exhibited a significant shift in substrate specificity toward a short-chain substrate. Computer modeling of these mutants suggested that a unique oxyanion hole, which is composed of Thr85 Oγ and Ser94 Oγ, was formed and thus the substrate specificity was changed. Therefore, coupling combinatorial mutagenesis with the cell surface display of ROL could lead to the production of a unique ROL mutant.




Similar content being viewed by others
References
Beer HD, Wohlfahrt G, Mccarthy JE, Schomburg D, Schmid, RD (1996) Analysis of the catalytic mechanism of a fungal lipase using computer-aided design and structural mutants. Protein Eng 9:507–517
Beer HD, McCarthy JEG, Bornscheuer UT, Schmid RD (1998) Cloning, expression, characterization and role of the leader sequence of a lipase from Rhizopus oryzae. Biochim Biophys Acta 1399:173–180
Benhar I (2001) Biotechnological applications of phage and cell display. Biotechnol Adv 19:1–33
Berg OG, Cajal Y, Butterfoss GL, Grey RL, Alsina MA, Yu BZ, Jain MK (1998) Interfacial activation of triglyceride lipase from Thermomyces (Humicola) lanuginosa: kinetic parameters and a basis for control of the lid. Biochemistry 37:6615–6627
Brocca S, Secundo F, Ossola M, Alberghina L, Carrea G, Lotti M (2003) Sequence of the lid affects activity and specificity of Candida rugosa lipase isoenzymes. Protein Sci 12:2312–2319
Cajal Y, Svendsen A, Girona V, Patkar SA, Alsina MA (2000) Interfacial control of lid opening in Thermomyces lanuginosa lipase. Biochemistry 39:413–423
Carlos JL, Klenotic PA, Paetzel M, Strynadka NCJ, Dalbey RE (2000) Mutational evidence of transition state stabilization by serine 88 in Escherichia coli type I signal peptidase. Biochemistry 39:7276–7283
Cygler M, Schrag JD (1997) Lipases and alpha/beta hydrolase fold. Methods Enzymol 284:3–28
Derewenda S, Brzozowski AM, Lawson DM, Derewenda ZM (1992) Catalysis at the interface: the anatomy of a conformational change in a triglyceride lipase. Biochemistry 31:1532–1541
Fischer M, Pleiss J (2003) The lipase engineering database: a navigation and analysis tool for protein families. Nucleic Acids Res 31:319–321
Herrgard S, Gibas CJ, Subramaniam S (2000) Role of an electrostatic network of residues in the enzymatic action of the Rhizomucor miehei lipase family. Biochemistry 39:2921–2930
Kohno M, Funatsu J, Mikami B, Kugiyama W, Matsuo T, Morita Y (1996) The crystal structure of lipase II from Rhizopus niveus at 2.2 A resolution. J Biochem 120:505–510
Kramer DN, Guilbault GG (1963) A substrate for the fluorometric determination of lipase activity. Anal Chem 35:4–5
Murai T, Ueda M, Atomi H, Shibasaki Y, Kamasawa N, Osumi M, Kawaguchi T, Arai M, Tanaka A (1997) Construction of a starch-utilizing yeast by cell surface engineering. Appl Microbiol Biotechnol 64:4857–4861
Sambrook J, Russel DW (2001) Molecular cloning, 3rd Edn. Cold Harbor Laboratory, Cold Harbor, N.Y.
Secundo F, Carrea G, Tarabiono C, Brocca S, Lotti M (2004) Activity and enantioselectivity of wildtype and lid mutated Candida rugosa lipase isoform 1 in organic solvents. Biotechnol Bioeng 86:236–240
Shibamoto H, Matsumoto T, Fukuda H, Kondo A (2004) Molecular engineering of Rhizopus oryzae lipase using a combinatorial protein library constructed on the yeast cell surface. J Mol Catal B Enzym 28:235–239
Shiraga S, Ueda M, Takahashi S, Tanaka A (2002) Construction of the combinatorial library of Rhizopus oryzae lipase mutated in the lid domain by displaying on years cell surface. J Mol Catal B Enzym 17:167–173
Shiraga S, Kawakami M, Ueda M (2004) Construction of combinatorial library of starch-binding domain of Rhizopus oryzae glucoamylase and screening of clones with enhanced activity by yeast display method. J Mol Catal B Enzym 28:229–234
Svendsen A (2000) Lipase protein engineering. Biochim Biophys Acta 1543:223–238
Tajima M, Nogi Y, Fukasawa T (1985) Transcriptional regulation of yeast genes for galactose metabolism. Yeast 1:67–77
Takahashi S, Ueda M, Atomi H, Beer HD, Bornscheuer UT, Schmid RD, Tanaka A (1998) Extracelluar production of active Rhizopus oryzae lipase by Saccharomyces cerevisiae. J Ferment Bioeng 86:164
Ueda M, Takahashi S, Washida M, Shiraga S, Tanaka A (2002) Expression of Rhizopus oryzae lipase gene in Saccharomyces cerevisiae. J Mol Catal B Enzym 17:113–124
Washida M, Takahashi S, Ueda M, Tanaka A (2001) Spacer-mediated display of active lipase on the yeast cell surface. Appl Microbiol Biotechnol 56:681–686
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shiraga, S., Ishiguro, M., Fukami, H. et al. Creation of Rhizopus oryzae lipase having a unique oxyanion hole by combinatorial mutagenesis in the lid domain. Appl Microbiol Biotechnol 68, 779–785 (2005). https://doi.org/10.1007/s00253-005-1935-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-005-1935-0