Summary
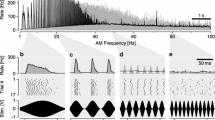
The response dynamics of cercal afferents in the cockroach, Periplaneta americana, were determined by means of a cross-correlation technique using a Gaussian white noise modulation of wind as a stimulus. The white noise stimulus could evoke sustained firing activity in most of the afferents examined (Fig. 1). The spike discharges were unitized and then cross-correlated with the stimulus to compute 1st- and 2nd-order Weiner kernels. The Ist-order kernels from a total of 28 afferents were biphasic and closely matched the time differential of a pulse (Figs. 1, 3 and 4). The amplitude and waveform of the kernels depended on the stimulus angle in such a way that the kernels were the mirror image of those on the polar opposite side (Figs. 2 and 3). The 2nd-order kernels were also differential. They had 2 diagonal peaks and 2 off-diagonal valleys in a 2-dimensional plot with 2 time axes (Figs. 1, 5 and 6). This 4-eye configuration was basically invariant irrespective of the stimulus angle, although the kernels varied in amplitude when the stimulus angle was changed. The time between the peak and a following trough of the 1st-order kernel was constant and had a mean of 4.6±0.1 ms, whereas the time between 2 diagonal peaks of the 2nd-order kernels was 4.7±0.1 ms (Figs. 4 and 6), suggesting that wind receptors (filiform sensilla) on cerci act as a band-pass filter with a peak frequency of about 106 Hz. The peak time, however, varies from 2.3 to 6.9 ms in both kernels, which may reflect the spatial distribution of the corresponding hairs on the cercus. The summation of the 1st- (linear) and 2nd-order (nonlinear) models precisely predicted the timing of the spike firing (Fig. 8). Thus, these 2 lower-order kernels can totally characterize the response dynamics of the wind receptors. The nonlinear response explains the directional sensitivity of the sensory neurons, while the differentiating 1st-order kernel explains the velocity sensitivity of the neurons. The nonlinearity is a signal compression in which one of the diagonal peaks of the 2nd-order kernel always offsets the downward phase of the 1st-order kernel (Fig. 7) and obviously represents a half-wave rectification property of the wind receptors that are excited by hair movement in only one direction and inhibited by hair movement in the polar opposite direction.
Similar content being viewed by others
References
Ball EE, Boyan GS, Stone RC (1982) The cercal receptor system of the praying mantid, Archimantis brunneriana Sauss. II. Cereal nerve structure and projection and electrophysiological responses of individual receptors. Cell Tissue Res 224:71–80
Boyan GS, Ball EE (1986) Wind-sensitive interneurones in the terminal ganglion of praying mantis. J Comp Physiol A 159:773–789
Buño W, Monti-Bloch L, Mateos A, Handler P (1981) Dynamic properties of cockroach cercal “threadlike” hair sensilla. J Neurobiol 12:123–141
Camhi JM (1980) The escape system of the cockroach. Sei Am 243:144–157
Camhi JM, Tom W (1978) The escape behavior of the cockroach Periplaneta americana. I. Turning response to wind puffs. J Comp Physiol 128:193–201
Camhi JM, Tom W, Volman S (1978) The escape behavior of the cockroach Periplaneta americana. II. Detection of natural predators by air displacement. J Comp Physiol 128:203–212
Dagan D, Camhi JM (1979) Response to wind recorded from the cercal nerve of the cockroach Periplaneta americana. II. Directional selectivity of the sensory neurons innervating single columns of filiform hairs. J Comp Physiol 133:103–110
Daley DL, Camhi JM (1988) Connectivity pattern of the cercal-to-giant interneuron system of the American cockroach. J Neurophysiol 60:1350–1368
French AS, Wong RKS (1977) Nonlinear analysis of sensory transduction in an insect mechanoreceptor. Biol Cybern 26:231–240
Gnatzy W (1976) The ultrastructure of the thread-hairs on the cerci of the cockroach Periplaneta americana L.: the intermolt phase. J Ultrastruct Res 54:124–134
Gnatzy W, Tautz J (1980) Ultrastructure and mechanical properties of an insect mechanoreceptor: stimulus-transmitting structures and sensory apparatus of the cercal filiform hairs of Gryllus. Cell Tissue Res 213:441–463
Kämper G, Kleindienst H (1990) Oscillation of cricket sensory hairs in a low-frequency sound field. J Comp Physiol A 167:193–200
Kondoh Y, Morishita H, Arima T, Okuma J, Hasegawa Y (1991) White noise analysis in a wind-sensitive, nonspiking interneuron of the cockroach. J Comp Physiol A 168:429–443
Marmarelis PZ, Marmarelis VZ (1978) Analysis of physiological systems. Plenum Press, New York
Mizunami M, Tateda H (1988) Dynamic relationship between the slow potentials and spikes in cockroach ocellar neurons. J Gen Physiol 91:703–723
Nicklaus R (1965) Die Erregung einzelner Fadenhaare von Periplaneta americana in Abhängigkeit von der Grösse und Richtung der Auslenkung. Z Vergl Physiol 50:331–362
Pearson KG, Stein RB, Malhotra SK (1970) Properties of action potentials from insect motor nerve fibers. J Exp Biol 53:299–316
Sakai HN, Naka K-I (1987a) Signal transmission in the catfish. V. Sensitivity and circuit. J Neurophysiol 58:1329–1350
Sakai HM, Naka K-I (1987b) Neuron network in catfish retina: 1968–1987. In: Osborne N, Chader J (eds) Progress in retina research, vol. 7. Pergamon Press, pp 149–208
Sakai HM, Naka K-I, Korenberg MJ (1988) White-noise analysis in visual neuroscience. Visual Neurosci 1:287–296
Sakuranaga M, Naka K-I (1985a) Signal transmission in the catfish retina. I. Transmission in the outer retina. J Neurophysiol 53:373–389
Sakuranaga M, Naka K-I (1985b) Signal transmission in the catfish retina. II. Transmission to type N cell. J Neurophysiol 53:390–410
Sakuranaga M, Ando Y-I, Naka K-I (1987) Dynamics of ganglioncell response in the catfish and frog retinas. J Gen Physiol 90:229–259
Shimozawa T, Kanou M (1984a) Varieties of filiform hairs: range fractionation by sensory afferents and cercal interneurons of a cricket. J Comp Physiol A 155:485–493
Shimozawa T, Kanou M (1984b) The aerodynamics and sensory physiology of range fractionation in the cercal filiform sensilla of the cricket Gryllus bimaculatus. J Comp Physiol A 155:495–505
Tobias M, Murphey RK (1979) The response of cercal receptors and identified interneurons in the cricket (Gryllus domesticus) to airstream. J Comp Physiol 129:51–59
Westin J (1979) Responses to wind recorded from the cercal nerve of the cockroach Periplaneta americana. I. Response properties of single sensory neurons. J Comp Physiol 133:97–102
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kondoh, Y., Arima, T., Okuma, J. et al. Filter characteristics of cercal afferents in the cockroach. J Comp Physiol A 169, 653–662 (1991). https://doi.org/10.1007/BF00194894
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00194894