Abstract
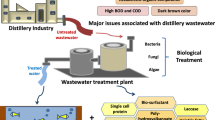
Distilleries are one of the most polluting industries generating enormous amount of wastewater from which an average of 10–15 L of effluent is released with the production of 1 L of alcohol. The distillery wastewater known as spent wash is characterized by its dark brown color, high temperature, low pH, and high percentage of dissolved organic and inorganic matter. It also contains nearly 2 % of the dark brown recalcitrant pigment called melanoidin which imparts dark brown color to the effluent. Various physical, chemical, and alternate treatment methods have been adopted for the removal of color from this wastewater. But these methods only change the form of contaminants rather than degrading them completely.
Biological methods produce relatively little amount of product after treatment by resolving a large amount of organism elements into carbon dioxide to be stabilized, or by removing organic matters contained in wastewater with the generation of methane gas. In the biological treatment methods, pollutants in wastewater can be resolved, detoxified, and separated by using mainly microorganisms. Due to the relatively low cost and the variations of work progress, the biological methods have been most widely used all over the world. A number of fungi, bacteria, yeast, and algae have been reported to have effluent treatment capabilities by the process of absorption, adsorption, and enzymatic degradation techniques. Toxicity studies of the biologically treated wastewaters also suggested that the process is efficient enough to reduce the toxicity of the spent wash by around 80 %. Hence, compared to the common and expensive physical or chemical ways for decolorization, an efficient bioremediation system has been found successful through biosorption and enzymatic ways of decolorization.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Abadulla E, Tzanov T, Costa S, Robra KH, Cavaco-Paulo A, Gübitz GM (2000) Decolorization and detoxification of texile dyes with laccase from Trametes hirsute. Appl Environ Microbiol 66:3357–3362
Aksu Z, Sag Y, Kutsal T (1992) The biosorption copper (II) by C. vulgaris and Z. ramigera. Environ Technol 13:579–586
Arana A, Roda A, Téllez A, Loera O, Carbajo JM, Terrón MC, et al (2004) Comparative analysis of laccase-isozymes patterns of several related Polyporaceae species under different culture conditions. J Basic Microbiol 44:79–87
Baldrian P (2006) Fungal laccases-occurrence and properties. FEMS Microbiol Rev 30:215–242
Bally R, Thomas-Bauzon D, Heulin T, Balandreau J, Richard C, De-Ley J (1983) Determination of the most frequent N2 fixing bacteria in the rice rhizosphere. Can J Microbiol 29:881–887
Bayramoglu G, Bektas S, Arica MY (2003) Biosorption of heavy metal ions on immobilized white-rot fungus Trametes versicolor. J Hazard Mater 101:285–300
Bourbonnais R, Paice MG (1992) Demethylation and delignification of kraft pulp by Trametes versicolor laccase in the presence of 2,2ʹ-azinobis-(3-ethylbenzthiazoline-6-sulphonate). Appl Environ Microbiol 36:823–827
Bourbonnais R, Paice MG, Reid I, Lanthier P, Yaguchi M (1995) Lignin oxidation by laccase isozymes from Trametes versicolor and role of the mediator 2,2-azinobis (3-ethylbenzthiazoline-6-sulfonate) in kraft lignin depolymerization. Appl Environ Microbiol 61:1876–1880
Bourbonnais R, Paice MG, Freiermuth B, Bodie E, Borneman S (1997) Reactivities of various mediators and laccases with kraft pulp and lignin model compounds. Appl Environ Microbiol 63:4627–1632
Breen A, Singleton FL (1999) Fungi in lignocellulose breakdown and biopulping. Curr Opin Biotechnol 10:252–258
Bressler DC, Fedorak PM, Pickard MA (2000) Oxidation of carbazole, N-ethylcarbazole, fluorene, and dibenzothiophene by the laccase of Coriolopsis gallica. Biotechnol Lett 22:1119–1125
Carliell CM, Barclay SJ, Naidoo N, Buckley CA, Mulholland DA, Senio E (1995) Microbial decolourization of a reactive azo dye under anaerobic conditions. Water SA 21:61–69
Claus H (2003) Laccases and their occurrence in prokaryotes. Arch Microbiol 179:145–150
Claus H (2004) Laccases: structure, reactions, distribution. Micron 35:93–96
Clifford DA (1999) Ion exchange and inorganic adsorption, In: Letterman RD (ed), Water quality and treatment, 5th edn. McGraw-Hill, New York
Dec J, Bollag JM (1994) Use of plant-material for the decontamination of water polluted with phenols. Biotechnol Bioeng 44:1132–1139
Dedeyan B, Klonowska A, Tagger S, Tron T, Iacazio G, Gil G, Petit LJ (2000) Biochemical and molecular characterization of a laccase from Marasmius quercophilus. Appl Environ Microbiol 6:925–929
Delval F, Crini G, Bertini S, Filiatre C, Torri G (2005) Preparation characterization and sorption properties of crosslinked starch-based exchangers. Carbohydr Polym 60:67–75
Ethanol India (2007) A sugar industry perspective & ethanol production (Ethanol Information India). http://www.ethanolindia.net/sugarind.html. Accessed July 2012
Ferrer I, Dezotti M, Durán N (1991) Decolorization of kraft effluent by free and immobilized lignin peroxidase and horseradish peroxidase. Biotechnol Lett 13:577–582
Fu YZ, Viraraghavan T (2001) Fungal decolorization of dye wastewaters: a review. Bioresour Technol 79:251–262
Gadd GM, White C, De-Rome L (1988) Heavy metal and radionuclide by fungi and yeasts. In: Norris PR, Kelly DP (eds) Biohydrometallurgy A. Rowe, Chippenham
Galun M, Galun E, Siegel B, Séller E, Leer H, Siegel S (1987) Removal of metal ions from aqueous solutions by Pencillium biomass: kinetic and uptake parameters. Water Air Soil Pollut 33:359–371
Ghosh M, Ganguli A, Tripathi AK (2002) Treatment of anaerobically digested distillery spentwash in a two-stage bioreactor using Pseudomonas putida and Aeromonas sp. Process Biochem 37:857–862
Godbole J (2002) Ethanol from cane molasses, Fuel Ethanol Workshop, Honululu, Hawaii. http://www.hawaii.gov/dbedt/ert/new-fuel/files/ethanol-workshop/10-Godbole-DOE-HI. Accessed 14 Nov 2002
González T, Terrón MC, Yagüe S, Zapico E, Galletti GC, González AE (2000) Pyrolysis/gas chromatography/mass spectrometry monitoring of fungal-biotreated distillery wastewater using Trametes sp. I-62 (CECT 20197). Rapid Commun Mass Spectrom 14:1417–1424
Hoff T, Liu SY, Bollag JM (1985) Transformation of halogen, alkyl, and alkoxy-substituted anilines by a laccase of Trametes versicolor. Appl Environ Microbiol 49:1040–1045
Jain N, Minocha AK, Verma CL (2002) Degradation of predigested distillery effluent by isolated bacterial strains. Indian J Exp Biol 40:101–105
Jhung JK, Choi E (1995) A comparative study of UASB and anaerobic fixed film reactors with development of sludge granulation. Water Res 29:271–277
Kalavathi DF, Uma L, Subramanian G (2001) Degradation and metabolization of the pigment-melanoidin in distillery effluent by the marine cyanobacterium Oscillatoria boryana BDU 92181. Enzyme Microb Technol 29:246–251
Kambe TN, Shimomura M, Nomura N, Chanpornpong T, Nakahara T (1999) Decolourization of molasses wastewater by Bacillus sp. under thermophilic and anaerobic conditions. J Biosci Bioeng 87:119–121
Karam J, Nicell JA (1997) Potential applications of enzymes in waste treatment. J Chem Technol Biotechnol 69:141–153
Kaushik G, Thakur IS (2009a) Isolation of fungi and optimization of process parameters for decolorization of distillery mill effluent. World J Microbiol Biotechnol 25:955–964
Kaushik G, Thakur IS (2009b) Isolation and characterization of distillery spent wash color reducing bacteria and process optimization by Taguchi approach. Int Biodeterior Biodegrad 63:420–426
Kaushik G, Thakur IS (2013) Biodegradation of synthetic dyes and purification, characterization and Mass spectroscopic analysis of thermotolerant laccase by Bacillus sp. Appl Biochem Microbiol 49:352–359
Kim SJ, Shoda M (1999) Batch decolourization of molasses by suspended and immobilizes fungus of Geotrichum candidum. J Biosci Bioeng 88:586–589
Kinae N, Hashu T, Makita T, Tomita I, Kimura I, Kanamori H (1981) Studies on the toxicity of pulp and paper mill effluents: mutagenicity of the sediment samples derived from kraft paper mills. Water Res 15:17–24
Knapp JS, Vantoch-Wood EJ, Zhang F (2001) Use of wood-rotting fungi for the decolourisation of dyes and industrial effluents. In: Gadd GM (ed) Fungi in bioremediation. British mycological society. Cambridge University Press, Cambridge, p 242
Kumar P, Chandra R (2004) Detoxification of distillery effluent through Bacillus thuringiensis (MTCC 4714) enhanced phytoremediation potential of Spirodela polyrrhiza (L.) Schliden. Bull Environ Contam Toxicol 73:903–910
Kumar P, Chandra R (2006) Decolurisation and detoxification of synthetic molasses melanoidins by individual and mixed cultures of Bacillus spp. Bioresour Technol 97:2096–2102
Lackner R, Srebotnik E, Messner K (1991) Oxidative degradation of high molecular weight chlorolignin by manganese peroxidase of Phanerochaete chrysosporium. Biochem Biophys Res Commun 178:1092–1098
Leonowicz A, Cho NS, Luterek J, Wilkolazka A, Wojtas- Wasilewska M, Matuszewska A, Hofrichter M, Wesenberg D (2001) Fungal laccase: properties and activity on lignin. J Basic Microbiol 41:185–227
Leontievsky AA, Myasoedova NM, Baskunov BP, Evans CS, Golovleva LA (2000) Transformation of 2,4,6-trichlorophenol by the white rot fungi Panus tigrinus and Coriolus versicolor. Biodegradation 11:331–340
Lettinga G, Hulshoff-Pol LW (1991) UASB process design for various types of wastewaters. Water Sci Technol 24:87–107
Li K, Xu F, Eriksson KEL (1999) Comparison of fungal laccases and redox mediators in oxidation of a nonphenolic lignin model compound. Appl Environ Microbiol 65:2654–2660
Liu CH, Wu JS, Chiu HC, Suen SY, Chu KH (2007) Removal of anionic reactive dyes from water using anion exchange membranes as adsorbers. Water Res 41:1491–1500
Mailleret L, Bernard O, Steyer JP (2003) Robust regulation of anaerobic digestion process. Water Sci Technol 48:87–94
Mall ID, Kumar V (1997) Removal of organic matter from distillery effluent using low cost adsorbent. Chem Eng World 32(7):89–96
Martins SIFS, van-Boekel MAJS (2004) A kinetic model for the glucose/glycine Maillard reaction pathways. Food Chem 90(1–2):257–269
Martins LO, Soares CM, Pereira MM, Teixeira M, Costa T, Jones GH, Henriques AO (2002) Molecular and biochemical characterization of a highly stable bacterial laccase that occurs as a structural component of the Bacillus subtilis endospore coat. J Biol Chem 277:18849–18859
Mohana S, Desai C, Madamwar D (2007) Biodegradation and decolourization of anaerobically treated distillery spent wash by a novel bacterial consortium. Bioresour Technol 98:333–339
Moriya K, Iefuji H, Shimoi H, Sato S, Tadenuma M (1990) Treatment of distillery wastewater discharged from beet molasses spirits production using yeast. J Ferment Bioeng 69:138–140
Nandy T, Shastry S, Kaul SN (2002) Wastewater management in cane molasses distillery involving bioresource recovery. J Environ Manag 65(1):25–38
Niku-Paavola ML, Viikari L (2000) Enzymatic oxidation of alkenes. J Mol Catal B Enzym 10:435–444
Ohmomo S, Kaneko Y, Sirianuntapiboon S, Somachi P, Atthasampunna P, Nakamura I (1987) Decolorization of molasses wastewater by a thermophilic strain Aspergillus fumigatus G-2-6. Agric Biol Chem 52:3339–3346
Ohmomo S, Daengsabha W, Yoshikawa H, Yui M, Nozaki K, Nakajima T, Nakamura I (1988) Screening of anaerobic bacteria with the ability to decolourize molasses melanoidin. Agric Biol Chem 57:2429–2435
Paice MG, Reid ID, Boubonnais R, Archibald FS, Jurasek L (1993) Manganese peroxidase, produced by Trametes cersicolor during pulp bleaching, demethylates and delignifies kraft pulp. Appl Environ Microbiol 59:260–265
Patil PU, Kapadnis BP, Dhamankar VS (2003) Decolorization of synthetic melanoidin and biogas effluent by immobilized fungal isolate of Aspergillus niger UM2. All India Distiller’s Association (AIDA) Newsletter 53–56
Perez J, Jeffries TW (1992) Roles of manganese and organic acid chelators in regulating lignin degradation and biosynthesis of peroxidases by Phanerochaete chrysosporium. Appl Environ Microbiol 58:2402–2409
Pointing SB, Jones EBG, Vrijmoed LLP (2000) Optimization of laccase production by Pycnoporus sanguineus in submerged liquid culture. Mycologia 92:139–44
Pokhrel D, Viraraghavan T (2004) Treatment of pulp and paper mill wastewater: a review. Sci Total Environ 333:37–58
Raghukumar C (2000) Fungi from marine habitats: an application in bioremediation. Mycol Res 104:1222–1226
Raghukumar C, Mohandass C, Kamat S, Shailaja MS (2004) Simultaneous detoxification and decolorization of molasses spent wash by the immobilized white-rot fungus Flavodon flavus isolated from a marine habitat. Enzyme Microb Technol 35:197–202
Ringot D, Lerzy B, Bonhoure JP, Auclair E, Oriol E, Larondelle Y (2005) Effect of temperature on in vitro ochratoxin a biosorption onto yeast cell derivatives. Process Biochem 40:3008–3016
Rodriguez E, Pickard MA, Vazquez DR (1999) Industrial dye decolorization by laccases from ligninolytic fungi. Curr Microbiol 38:27–32
Sánchez-Amat A, Solano F (1997) A pluripotent polyphenol oxidase from the melanogenic marine Alteromonas sp. shares catalytic capabilities of tyrosinases and laccases. Biochem Biophys Res Commun 240:787–792
Seth R, Goyal SK, Handa BK (1995) Fixed film biomethanation of distillery spentwash using low cost porous media. Resour Conserv Recycl 14:79–89
Sirianuntapiboon S., Zohsalam P., Ohmomo S. 2003. Decolourization of molasses wastewater by Citeromyces sp. WR-43-6. Process Biochem 39:917–924
Skerratt G (2004) European distilleries: an overview. In: Tewari PK (ed) Liquid asset. Proceedings of the Indo-EU workshop on promoting efficient water use in agro-based industries. TERI, New Delhi, pp 1–11
Srivastava S, Thakur IS (2006) Biosorption potency of Aspergillus niger for removal of chromium (VI). Curr Microbiol 53:232–237
Tewari PK, Batra VS, Balakrishnan M (2007) Water management initiatives in sugarcane molasses based distilleries in India. Resour Conserv Recycl 52:351–367
Thakkar AP, Dhamankar VS, Kapadnis BP (2006) Biocatalytic decolourisation of molasses by Phanerochaete chrysosporium. Bioresour Technol 97:1377–1381
Tsezos M, Bell JP (1989) Comparison of the biosorption and desorption of hazardous organic pollutants by live and dead biomass. Water Res 23:561–568
Valderrama LT, Del-Campo CM, Rodriguez CM, Bashan LE, Bashan Y (2002) Treatment of recalcitrant wastewater from ethanol and citric acid using the microalga Chlorella vulgaris and the macrophyte Lemna minuscula. Water Res 36:4185–4192
Veglio F, Beolchini F (1997) Removal of metals by biosorption: a review. Hydrometallurgy 44:301–316
Wang CJ, Thiele S, Bollag JM (2002) Interaction of 2,4,6-trinitrotoluene (TNT) and 4-amino-2,6-dinitrotoluene with humic monomers in the presence of oxidative enzymes. Arch Environ Contam Toxicol 42:1–8
Wariishi H, Valli K, Gold MH (1991) In vitro depolymerization of lignin by manganese peroxidase of Phanerochaete chrysosporium. Biochem Biophys Res Commun 176:269–275
Watanabe Y, Sugi R, Tanaka Y, Hayashida S (1982) Enzymatic decolorization of melanoidin by Coriolus sp. No. 20. Agric Biol Chem 46:1623–1630
Won SW, Choi SB, Chung BW, Park D, Park JM, Yun YS (2004) Biosorptive decolorization of reactive orange 16 using the waste biomass of Corynebacterium glutamicum. Ind Eng Chem Res 43:7865–7869
Wu JS, Liu CH, Chu KH, Suen SY (2008) Removal of cationic dye methyl violet 2B from water by cation exchange membranes. J Membr Sci 309:239–245
Yeoh BG (1997) Two-phase anaerobic treatment of cane-molasses alcohol stillage. Water Sci Technol 36:(6-7):441–448
Zacharewski T, Berhane K, Gillesby B (1995) Detection of estrogen. and dioxin-like activity in pulp and paper MI black liquor and effluent using in Vitro recombinant receptor (reporter): I gene assays. Environ Sci Technol 29:2140–2146
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer India
About this chapter
Cite this chapter
Kaushik, G. (2015). Bioremediation of Industrial Effluents: Distillery Effluent. In: Kaushik, G. (eds) Applied Environmental Biotechnology: Present Scenario and Future Trends. Springer, New Delhi. https://doi.org/10.1007/978-81-322-2123-4_2
Download citation
DOI: https://doi.org/10.1007/978-81-322-2123-4_2
Published:
Publisher Name: Springer, New Delhi
Print ISBN: 978-81-322-2122-7
Online ISBN: 978-81-322-2123-4
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)