Abstract
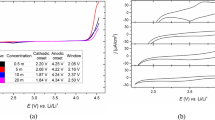
Conducting poly(o-anisidine) coatings were obtained on low carbon steel in aqueous oxalic acid solution by using the galvanostatic technique. The coatings were characterised by potential-time relations, UV-VIS absorption spectroscopy, scanning electron microscopy, and X-ray diffraction measurements. The electrochemical performance of coated steel electrodes was evaluated on the basis of galvanostatic charge-discharge performance and electrochemical impedance spectroscopy in 0.5 M H2SO4. Maximum charging current was found in the case of the coating obtained at a current density of 8 mA cm−2 for 600 s duration at the supply voltage of 0.5 V. The estimated capacitance of the coated steel electrode for charging is 42.67 mF and 7.2 mF for discharging. It was also found that there was an increase in capacitance as a function of supply voltage and the maximum value was obtained at 0.5 V. The study reveals the possibility of using conducting poly(o-anisidine)-coated low carbon steel from oxalic acid medium as supercapacitor electrode materials.
Similar content being viewed by others
References
Arsov, L. D., Plieth, W., & Koßmehl, G. (1998). Electrochemical and Raman spectroscopic study of polyaniline; influence of the potential on the degradation of polyaniline. Journal of Solid State Electrochemistry, 2, 355–361. DOI: 10.1007/s100080050112.
Cheng, Q., Tang, J., Ma, J., Zhang, H., Shinya, N., & Qin, L. C. (2011). Polyaniline-coated electro-etched carbon fibre cloth electrodes for supercapacitors. Journal of Physical Chemistry, 115, 23584–23590. DOI: 10.1021/jp203852p.
Li, X. W., Li, X. H., Dai, N., Wang, G. C., & Wang, Z. (2010). Preparation and electrochemical capacitance performances of super-hydrophilic conducting polyaniline. Journal of Power Sources, 195, 5417–5421. DOI: 10.1016/j.jpowsour.2010.03.034.
Li, L. I., Liu, E. H., Shen, H. J., Yang, Y. J., Huang, Z. H., Xiang, X. X., & Tian, Y. Y. (2011). Charge storage performance of doped carbons prepared from polyaniline for supercapacitors. Journal of Solid State Electrochemistry, 15, 175–182. DOI: 10.1007/s10008-010-1087-8.
Pawar, P., Wankhede, M. G., Patil, P. P., & Sainkar, S. R. (2003). Investigations on growth mechanism of poly(oanisidine) coatings on low carbon steel by electrochemical synthesis method. Materials Science and Engineering, A347, 365–373. DOI: 10.1016/s0921-5093(02)00617-2.
Peng, C., Zhang, S. W., Jewell, D., & Chen, G. Z. (2008). Carbon nanotubes and conducting polymer composites for supercapacitors. Progress in Natural Science, 18, 777–788. DOI: 10.1016/j.pnsc.2008.03.002.
Rajendra Prasad, K., & Munichandraiah, N. (2002). Potentiodynamically deposited polyaniline on stainless steel inexpensive, high-performance electrodes for electrochemical supercapacitors. Journal of the Electrochemical Society, 149, A1393–A1399. DOI: 10.1149/1.1509458.
Trivedi, D. C. (1997). Polyanilines. In H. S. Nalwa (Ed.), Handbook of organic conductive molecules and polymers (pp. 506–566). Chichester, UK: Wiley.
Zhang, Y., Feng, H., Wu, X. B., Wang, L. Z., Zhang, A. Q., Xia, T. C., Dong, H. C., Li, X. F., & Zhang, L. S. (2009). Progress of electrochemical capacitor electrode materials: A review. International Journal of Hydrogen Energy, 34, 4889–4899. DOI: 10.1016/j.ijhydene.2009.04.005.
Zhou, H. H., Chen, H., Luo, S. L., Lu, G. W., Wei, W. Z., & Kuang, Y. F. (2005). The effect of the polyaniline morphology on the performance of polyaniline supercapacitors. Journal of Solid State Electrochemistry, 9, 574–580. DOI: 10.1007/s10008-004-0594-x.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Deshpande, P.P., Murali, M., Deshpande, P.P. et al. Conducting poly(o-anisidine)-coated steel electrodes for supercapacitors. Chem. Pap. 67, 1066–1071 (2013). https://doi.org/10.2478/s11696-013-0317-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11696-013-0317-9