Abstract
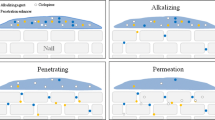
The main objective of the current work is to demonstrate the process of passive lateral diffusion in the human nail plate and its effect on the passive transungual permeation of antifungal drug ciclopirox olamine (CPO). A water soluble dye, methyl red sodium salt (MR) was used to visualize the process of lateral diffusion using a novel suspended nail experiment. The decline in concentration of CPO correlates with that of concentration of MR from the proximal to the distal end of the nail in suspended nail study. Three toenails each were trimmed to 5 mm × 5 mm (25 mm2), 7 mm × 7 mm (49 mm2), and 9 mm × 9 mm (81 mm2) to study the extent and effect of lateral diffusion of the CPO on its in vitro transungual permeation. The permeation flux of CPO decreased as the surface area of the toenail increased. There was a positive correlation between the concentrations of CPO and MR in the area of application and in the peripheral area of the toenails of the three surface areas, confirming the findings in the suspended nail experiment. Profound lateral diffusion of CPO was demonstrated and shown to reduce the in vitro passive transungual drug permeation and prolong the lag-time in human toenails. The study data implies that during passive in vitro transungual permeation experiments, the peripheral nail around the area of drug application has to be kept to a minimum, in order to get reliable data which mimics the in vivo situation.







Similar content being viewed by others
REFERENCES
Hao J, Smith K, Li K. Iontophoretically enhanced ciclopirox delivery into and across human nail plate. J Pharm Sci. 2009;98(10):3608–16.
Hui X, Wester RC, Barbadillo S, Lee C, Patel B, Wortzmman M, et al. Ciclopirox delivery into the human nail plate. J Pharm Sci. 2004;93:2545–8.
Monti D, Saccomani L, Chetoni P, Burgalassi S, Tampucci S, Mailland F. Validation of bovine hoof slices as a model for infected human toenails: in vitro ciclopirox transungual permeation. Br J Dermatol. 2011;165(1):99–105.
Palliyil B, Lebo DB, Patel PR. A preformulation strategy for the selection of penetration enhancers for a transungual formulation. AAPS PharmSciTech. 2013. doi:10.1208/s12249-013-9954-0.
Chouhan P, Saini TR. Hydration of nail plate: a novel screening model for transungual drug permeation enhancers. Int Pharm. 2012;436(1–2):179–82.
Hao J, Smith KA, Li KS. Chemical method to enhance transungual transport and iontophoresis efficiency. Int J Pharm. 2008;357(1–2):61–9.
Khengar R, Jones S, Turner R, Forbes B, Brown M. Nail swelling as a pre-formulation screen for the selection and optimisation of ungual penetration enhancers. Pharm Res. 2007;24(12):2207–12.
Kobayashi Y, Miyamoto M, Sugibayashi K, Morimoto Y. Enhancing effect of N-acetyl-L-cysteine or 2-Mercaptoethanol on the in vitro permeation of 5-fluorouracil or tolnaftate through the human nail plate. Chem Pharm Bull. 1998;46(11):1797–802.
Malhotra GG, Zatz JL. Investigation of nail permeation enhancement by chemical modification using water as a probe. J Pharm Sci. 2002;91(2):312–23.
Mohorcic M, Torkar A, Friedrich J, Kristl J, Murdan S. An investigation into keratinolytic enzymes to enhance ungual drug delivery. Int J Pharm. 2007;332(196–201):196.
Murdan S. Enhancing the nail permeability of topically applied drugs. Expert Opin Drug Deliv. 2008;5(11):1267–82.
Murthy SN, Vaka SR, Sammeta SM, Nair AB. TranScreen-N: method for rapid screening of trans-ungual drug delivery enhancers. J Pharm Sci. 2009;98(11):4264–71.
Nair AB, Sammeta SM, Vaka SR, Narasimha MS. A study on the effect of inorganic salts in transungual drug delivery of terbinafine. J Pharm Pharmacol. 2009;61(4):431–7.
Nair AB, Sammeta SM, Kim HD, Chakraborty B, Friden PM, Murthy N. Alteration of the diffusional barrier property of the nail leads to greater terbinafine drug loading and permeation. Int J Pharm. 2009;375:22–7.
Nair AB, Chakraborty B, Murthy SN. Effect of polyethylene glycols on the trans-ungual delivery of terbinafine. Curr Drug Deliv. 2010;7(5):407–14.
Quintanar-Guerrero D, Ganem-Quintanar A, Tapia-Olquin P, Kalia YN, Buri P. The effect of keratolytic agents on the permeability of three imidazole antimycotic drugs through the human nail. Drug Dev Ind Pharm. 1998;24(7):685–90.
Shivakumar HN, Juluri A, Desai BG, Murthy N. Ungual and transungual drug delivery. Drug Dev Ind Pharm. 2012;38(8):901–11.
Traynor MJ, Turner RB, Evans CR, Khengar RH, Jones SA, Brown MB. Effect of a novel penetration enhancer on the ungual permeation of two antifungal agents. J Pharm Pharmacol. 2010;62(6):730–7.
van Hoogdalem EJ, van den Hoven WE, Terpstra IJ, van Zijtveld J, Verschoor JSC, Visser JN. Nail penetration of the antifungal agent oxiconazole after repeated topical application in healthy volunteer, and the effect of acetylcysteine. Eur J Pharm Sci. 1997;5(3):119–27.
Nair AB, Kim HD, Chakraborty B, Singh J, Zaman M, Gupta A, et al. Ungual and trans-ungual iontophoretic delivery of terbinafine for the treatment of onychomycosis. J Pharm Sci. 2009;98(11):4130–40.
Nair AB, Kim HD, Davis SP, Etheredge R, Barsness M, Friden PM, et al. An ex vivo toe model used to assess applicators for the iontophoretic ungual delivery of terbinafine. Pharm Res. 2009;26(9):2194–201.
Mansbridge J, Knapp M. Penetration of Lucifer Yellow into human skin: a lateral diffusion channel in the stratum corneum. J Histochem Cytochem. 1993;41(6):909–14.
Schicksnus G, Muller-Goymann CC. Lateral diffusion of Ibuprofen in human skin during permeation studies. Skin Pharmacol Physiol. 2004;17:84–90.
Escarrone AL, Bittencourt C, Laporta L, dos Santos M, Primel E, Caldas S. LC–UV method with pre-column derivatization for the determination of ciclopirox olamine in raw material and topical solution. Chromatographia. 2008;67:967–71.
Lehr K-H, Damm P. Quantification of ciclopirox by high-performance liquid chromatography after pre-column derivatization. J Chromatogr. 1985;339:451–6.
Myoung Y, Choi H-K. Permeation of ciclopirox across porcine hoof membrane: effect of pressure sensitive adhesives and vehicles. Eur J Pharm Sci. 2003;20:319–25.
Kobayashi Y, Miyamoto M, Sugibayashi K, Morimote Y. Drug permeation through the three layers of the human nail plate. J Pharm Pharmacol. 1999;51:271–8.
Murdan S. Drug delivery to the nail following topical application. Int J Pharm. 2002;236:1–26.
Garson J, Baltenneck F, Leroy F, Riekel C, Müller M. Histological structure of human nail as studied by synchrotron X-ray microdiffraction. Cell Mol Biol. 2000;46(6):1025–34.
Gupta AK, Kohli Y. In vitro susceptibility testing of ciclopirox, terbinafine, ketoconazole and itraconazole against dermatophytes and nondermatophytes, and in vitro evaluation of combination antifungal activity. Br J Dermatol. 2003;149(2):296–305.
Kokjohn K, Bradley M, Griffiths B, Ghannoum M. Evaluation of in vitro activity of ciclopirox olamine, butenafine HCl and econazole nitrate against dermatophytes, yeasts and bacteria. Int J Dermatol. 2003;42(S1):11–7.
Shehata AS, Mukherjee PK, Ghannoum MA. Comparison between the standardized clinical and laboratory standards institute M38-A2 method and a 2,3-Bis(2-Methoxy-4-Nitro-5-[(Sulphenylamino)Carbonyl]-2H-Tetrazolium hydroxide based method for testing antifungal susceptibility of dermatophytes. J Clin Microbiol. 2008;46(11):3668–71.
Singh J, Zaman M, Gupta AK. Evaluation of microdilution and disk diffusion methods for antifungal susceptibility testing of dermatophytes. Med Mycol. 2007;45(7):595–602.
Subissi A, Monti D, Togni G, Maillan F. Ciclopirox : recent nonclinical and clinical data relevant to its use as a topical antimycotic agent. Drugs. 2010;70(16):2133–52.
Gotter B, Faubel W, Neubert RHH. Photothermal imaging in 3D surface analysis of membrane drug delivery. Eur J Pharm Biopharm. 2010;74:26–32.
ACKNOWLEDGMENTS
The study was funded by cGMP services, Temple University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Palliyil, B.B., Li, C., Owaisat, S. et al. Lateral Drug Diffusion in Human Nails. AAPS PharmSciTech 15, 1429–1438 (2014). https://doi.org/10.1208/s12249-014-0169-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-014-0169-9