Abstract
This study evaluated the dietary lysine requirement by measuring the plasma free lysine concentrations in rainbow trout, Oncorhynchus mykiss after dorsal aorta cannulation. A basal diet containing 36.6 % crude protein (29.6 % crystalline amino acids mixture, 5 % casein and 2 % gelatin) was formulated to one of the seven L-amino acid based diets containing graded levels of lysine (0.72, 1.12, 1.52, 1.92, 2.32, 2.72 or 3.52 % dry diet). A total of 35 fish averaging 512 ± 6.8 g (mean ± SD) were randomly distributed into seven groups with five fish in each group. After 48 h of feed deprivation, each group of fish was fed one of the experimental diets by intubation at 1 % body weight. Blood samples were taken at 0, 5 and 24 h after intubation. Post-prandial plasma free lysine concentrations (PPlys, 5 h after intubation) of fish fed diets containing ≥ 2.32 % lysine were higher than those of fish fed diets containing ≤ 1.92 % lysine. Post-absorptive free lysine concentrations (PAlys, 24 h after intubation) of fish fed diets containing 2.32 and 3.52 % lysine were higher than those of fish fed diets containing ≤ 1.52 % lysine. The broken-line regression analysis on the basis of PPlys and PAlys indicated that the lysine requirement of rainbow trout could be 2.34 and 2.20 % in diet.
Therefore, these results strongly suggested that the dietary lysine requirement based on the broken-line model analyses of PPlys and PAlys could be greater than 2.2 but less than 2.34 % (corresponding to be 6.01 % ≤, but ≤ 6.39 % in dietary protein basis, respectively) in rainbow trout. Also, these results shown that the quantitative estimation of lysine requirement by using PPlys and PAlys could be an acceptable method in fish.
Similar content being viewed by others
Background
Lysine is one of the essential amino acids, which is not synthesized in animals; so it must be ingested as lysine or lysine-containing proteins (Wilson 2002). It is the most limiting amino acid in the plant protein sources used in formulating fish feeds (Harris 1980; Forster and Ogata 1998; Small and Soares 2000; Ahmed and Khan 2004). Lysine deficiency causes loss of appetite, growth retardation, fin erosion and increased mortality as shown in milk fish Chanos chanos (Borlongan and Benitez 1990), Japanese sea bass Lateolabrax japanicus (Mai et al. 2006) and rainbow trout Oncorhynchus mykiss (Ketola 1983). So, it is important to determine the lysine requirement of cultured fish species for normal growth and feed utilization.
Dietary lysine requirement has been estimated for commonly cultured species of fish, ranging from 3.2 to 6.2 % of dietary protein (Wilson 2002). Several researchers have reported the lysine requirement of rainbow trout to range from 1.30 (3.17) to 2.9 (6.1) % of diet (dietary protein) (Ogino 1980; Walton et al. 1984; Kim et al. 1992; Rodehutscord et al. 1997). The above requirements were determined by feeding trials. Feeding trials are expensive and last for several weeks or months before appreciable responses are observed. While, determination of amino acid requirements by measuring plasma amino acid and ammonia concentrations could be employed to obtain the desired results with less labor and cost. This method has been successfully employed in several species of fish and results are similar to those obtained by feeding trials (Cowey 1995; Ok et al. 2001). Hence, the method can be a reliable alternative to determine the amino acids or other important nutrient requirement in commercially important fish species (Bae et al. 2011).
The relationship between the concentration of PFAA and intake of dietary amino acids have been investigated by several researchers (Plakas et al. 1980; Murai et al. 1987; Bai et al. 2003). Plasma free amino acid (PFAA) concentrations were used to investigate the amino acids metabolism and to evaluate the quality of dietary protein in rainbow trout Oncorhynchus mykiss (Nose 1972; Walton and Wilson 1986; Schuhmacher et al. 1997) carp Cyprinuscarpio (Plakas et al. 1980; Dabrowski 1982) and Atlantic salmon Salmo salar (Espe et al. 1993; Sunde et al. 2003). Evaluation of PFAA concentrations has led to the discovery of genetic defects of amino acid metabolism which lead to the diagnosis of primary renal or liver disease including the effects of amino acid deficiencies, imbalances and toxicities related to amino acid metabolism (Zicker and Rogers 1990). Although the effects of dietary protein or amino acid mixtures on PFAAs concentrations in rainbow trout, (Schuhmacher et al. 1997; Murai et al. 1987; Vermeirssen et al. 1997) and sea bass Dicentrarchus labrax (Thebault et al. 1985) have been reported, the complete dose response relationships for amino acids have not been investigated.
In our previous studies, requirements for dietary essential amino acids such as arginine, methionine, valine and threonine were obtained in rainbow trout by using surgically modified methods in our laboratory (Ok et al. 2001; Bae et al. 2011; Ok 2002; Park et al. 2005). As a part of our consecutive studies on essential amino acid requirements of rainbow trout, the purpose of the present study was to estimate the dietary lysine requirement based on the plasma free lysine concentrations in dorsal aorta cannulated rainbow trout.
Methods
Experimental fish
Rainbow trout, averaging 512 ± 6.8 g (body weight, mean ± SD) were obtained and the experiment was carried out in Ewhajung trout farm of Sangju, Korea. In the present experiment, seven net cages (1.3 × 1.3 × 1.3 m) were placed in a flow-through concrete raceway with a water flow rate of 60 L/min. All the fish in each group were arranged as four cages in a single row placed above the three cages in another row with the same environmental condition in all. Fish were fed with a commercial rainbow trout diet (Woo-sung Feed Co. Ltd., Daejon, Korea) for 72 h until the fish recovered from the dorsal aorta cannulation operation. Supplemental aeration was also provided to maintain the dissolved oxygen at 6.5 ± 0.5 mg/L, and water temperature was maintained at 16 ± 0.4 °C.
Preparation of experimental diets
A basal diet containing 36.6 % crude protein (29.6 % crystalline amino acids mixture, 5 % casein and 2 % gelatin) was formulated by the modification of Kim (1997). Seven experimental diets were formulated to contain graded levels of lysine (0.72, 1.12, 1.52, 1.92, 2.32, 2.72 or 3.52 % of diet, on a dry matter basis) based on the basal diet. Equal amounts of aspartic acid and glutamic acid by weight were substituted for the graded amounts of lysine in the basal diet to maintain the seven experimental diets isonitrogenous. Formulation and amino acid composition of the basal diet are shown in Tables 1 and 2, respectively. The ingredient mixtures without oil were stored at −80 °C until used and the diets were prepared by adding fish oil (10 % of diet) and water (four parts of distilled water : ten parts of diet, w/w) before intubation.
Dorsal aorta cannulation and intubation (force-feeding)
The experiment was conducted under the guidelines of Animal Ethics Committee Regulations, No. 554 issued from Pukyong National University, Busan, Korea. Rainbow trout were anaesthetized with 200 mg/L 3-aminobenzoic acid ethyl ester methanesulfonate (MS-222, Sigma Chemical Company, St. Louis, MO, USA) for 3–5 min, placed on a V-shaped table, and their gills were irrigated continuously with 16 °C water containing100mg/L of MS-222 during the operation.A50 cm long cannula (Clay Adams PE 50 tubing, Parsippany, NJ, USA) with a bubble of about 5–6 cm on one end was used and the cannula was washed with the heparinized Cortland saline solution (Houston 1990) before use. A13-gauge needle was used to pierce a hole on the right nostrum of fish (ventral side up) as an exit for the cannula. A 19-gauge needle was used to bore a small hole in the roof of the mouth at the mid-line behind the third gill arch at a 30° angle, and a piano wire was inserted into the PE 50 tubing as a guide. The proper insertion was verified by the observation of a slow blood flow after the wire was withdrawn from the cannula. A 3 mL syringe with a 23-gauge needle was used to remove air and blood clot and the cannula was flushed with the heparinized saline solution. The cannula was sutured behind the bubble on the roof of the mouth, led outfrom the right nostrum, plugged with a color headpin and sutured at the dorsal fin (Ok et al. 2001; Bae et al. 2011; Bai et al. 2003; Park et al. 2005). Thirty five dorsal aorta cannulated rainbow trout were randomly distributed into seven groups of five fish per group and fed with one of the seven experimental diets per group. After 24 h of feed deprivation, the fish were anesthetized with 200 mg/L of MS-222 and fed the experimental diet by the stomach intubation method (diet plus 0.4 parts of distilled water per diet) using a 3 mL syringe.
Sample collection and analysis
Five fish per group were anesthetized with 200 mg/L MS-222 and blood was sampled using a 3 mL syringe from each fish at 0,5 and 24 h after intubation of the experimental diets. Plasma samples were prepared by centrifugation at 3000 g for 10 min at room temperature. For deproteinization, plasma samples were mixed with a 10 % 5-sulphosalicylic acid solution in the ratio of 4:1 (v/v), cooled on ice for 30 min and re-centrifuged. The protein-free supernatant was dissolved in pH 2.2 lithium citrate sample dilution buffer in the ratio of 1:1 (v/v), and the samples were stored at −80 °C until analysis. Plasma free amino acids were quantified using aS433 amino acid analyzer (Sykam, Gilching, Germany) using the ninhydrin method. Plasma ammonia concentrations were analyzed using the Berthelot reaction (Sigma).
Statistical analysis
Data were subjected to one way analysis of variance test using Statistix 3.1(Analytical Software, St Paul, MN, USA). When a significant treatment effect was observed, a Least Significant Difference test was used to compare means. Treatment effects were considered significant at P < 0.05. The breakpoints for PPlys, PAlys and PPA were estimated by using the broken line model of Robbins et al. (1979).
Results
Plasma free lysine concentrations
Results of post-prandial plasma free lysine concentrations (PPlys, 5 h after intubation) and post-absorptive plasma free lysine concentrations (PAlys, 24 h after intubation) are summarized in Table 3. PPlys of fish fed diets containing ≥ 2.32 % lysine were significantly higher than those of fish fed diets containing ≤ 1.92 % lysine. There were no significant differences in PPlys among fish fed diets containing ≥ 2.32 % lysine. Broken-line model analyses on the basis of PPlys shown in Fig. 1 indicated that the dietary lysine requirement of trout should be 2.34 (6.39) % of diet (% dietary protein on a dry matter basis). PAlys of fish fed diets containing 2.32 % and 3.52 % lysine were significantly higher than those of fish fed diets containing ≤ 1.52 % lysine. However, there were no significant differences in PAlys among fish fed diets containing ≥ 1.92 % lysine. Broken-line model analyses on the basis of PAlysin Fig. 2 indicated that the dietary lysine requirement of trout could be 2.20 (6.01) % of diet (% dietary protein on a dry matter basis).
Plasma ammonia concentrations
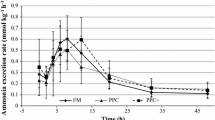
Post-prandial plasma ammonia concentrations (PPA, 5 h after intubation) and post-absorptive plasma ammonia concentrations (PAA, 24 h after intubation) are shown in Table 3. PPA of fish fed diets containing 3.52 % lysine were significantly higher than those of fish fed diets containing 2.32 % or less lysine. There were no significant differences in PPA among fish fed diets containing 2.72 % and 3.52 % lysine. Post-prandial plasma ammonia significantly increased with dietary lysine levels from 0.72 % (115 ± 19 nmol/mL) to 2.72 % (282 ± 15 nmol/mL), and the concentrations remained constant for fish fed diets containing ≥ 2.72 % lysine. Post absorptive plasma ammonia concentrations in the present study were not significantly different among the groups.
Discussion
In the present study, the dietary lysine requirements for the rainbow trout based on plasma free lysine concentrations (PPlys and PAlys) should be between 2.20 (6.01) % and 2.34 (6.39) % of diet (% dietary protein basis). In line with the present study, dietary lysine requirements of rainbow trout have been reported to be 2.23 (5.3) % of diet (dietary protein) by Ogino (1980) and 2.32 (6.63) % by Rodehutscord et al. (1997) based on the growth performance. Also, results of the present study were similar to those reported for several other species, such as grass carp Ctenopharyngodon idella (2.24 % of diet) (Wang et al. 2005) and cobia Rachycentron canadum (2.33 %) (Zhou et al. 2007), but higher than those reported for Japanese flounder Paralichthys olivaceus 1.97–2.16 % and red sea bream Pagrus major 2.09–2.14 % (Forster and Ogata 1998), red drum Sciaenops ocellatus 2.0 % (Brown et al. 1988), milkfish Chanos chanos 2.0 % (Borlongan and Benitez 1990) and gilthead seabream Sparus aurata 2.15 % (Marcouli et al. 2006) and lower than those reported for Japanese seabass 2.49 % (Mai et al. 2006) and grouper Epinephelus coioides 2.83 % (Luo et al. 2006). The wide variation observed in the requirements for lysine among fish species may be due to the differences in dietary protein sources, the amino acid pattern (Forster and Ogata 1998), diet formulation, size and age of fish, genetic differences, feeding practices and rearing conditions (Ruchimat et al. 1997). Furthermore, assimilation rate, amino acid form and energy in feedstuff may also influence amino acid requirements (Simmons et al. 1999; De Silva et al. 2000). In general, lysine requirements of fish species that have high protein requirements, such as marine fish and freshwater carnivorous fish, are higher than those of the species that have low protein requirements including omnivores and herbivorous fish (Zhou et al. 2007).
The pattern of post prandial or absorptive plasma free amino acid concentrations of fish fed with graded levels of the amino acid tested has been used in several studies in an attempt to confirm the requirement values assessed by growth (Thebault et al. 1985; Wilson et al. 1980; Tibaldi and Tulli 1999). The effects of dietary lysine intake on PPlys and PAlys were dependent upon the relative adequacy of the dietary lysine supply, increasing up to the dietary lysine requirement levels with no further increase above the requirement values. The plasma limiting amino acid concentration is expected to be low in fish given sub-optimal levels of the tested amino acid, however when adequate levels are fed, the plasma concentration tends to increase linearly as the dietary concentration rises (Cowey 1995). As indicated above, a similar response was observed in the present study where a breakpoint was observed in the plasma concentration value at 2.20 % (PPlys) or 2.34 % (PAlys) dietary lysine.
Post-prandial plasma ammonia concentrations (PPA, 5 h after intubation) were significantly affected by the dietary lysine level in this study. Ok (2002) reported that PPA remained low and virtually constant but increased significantly when dietary amino acid levels were higher than the requirement values for essential amino acids such as arginine, valine, methionine and threonine in rainbow trout. However, PPA increased linearly in response to dietary lysine level, and the breakpoint was observed in PPA at 3.10 % (8.47) dietary lysine (% dietary protein) in the present study. The results showed that estimated lysine requirement value based on PPA is considerably higher than those based on PPlys or PAlys. Results of the present study indicated that measuring plasma ammonia concentrations is not acceptable in evaluating the lysine requirement of rainbow trout.
The patterns of most plasma free amino acid concentrations from force-fed rainbow trout with and without dorsal aorta cannulation were similar, and the dorsal aorta cannulation did not affect these patterns (Ok et al. 2001). These results indicated that force-fed rainbow trout might recover from the stress of dorsal aorta cannulation within 48 h of the operation, and the dorsal aorta cannulation would allow repeated sampling on the same individual fish to study nutrient metabolism in the blood circulation. Plasma amino acid concentrations may be useful in determining the optimum blood sampling time and evaluating protein quality and essential amino acid requirements. Furthermore, this technique can be used in pharmaceutical and toxicological as well as in nutritional research in rainbow trout. Since results in this and other studies involving measurement of plasma free amino acids are similar to values obtained by growth trial, it could be suggested that this technique could be employed in estimating other amino acid requirements in rainbow trout.
Conclusions
We concluded that the dietary lysine requirement based on the Broken-line model analyses of post-prandial and post absorptive plasma free lysine concentrations (PPlys and PAlys) could be greater than 2.2 but less than 2.34 % (corresponding to be 6.01 % ≤, but ≤ 6.39 % in dietary protein basis, respectively) in rainbow trout. Also, we concluded that quantitative estimation of lysine requirement by using PPlys and PAlys could be an acceptable method in fish.
References
Ahmed I, Khan MA. Dietary lysine requirement of fingerling Indian major carp Cirrhinus mrigala (Hamilton). Aquaculture. 2004;235:499–511.
Bae JY, Ok IH, Lee, et al. Reevaluation of dietary methionine requirement by plasma methionine and ammonia concentrations in surgically modified rainbow trout Oncorhynchus mykiss. J Appl Ichthyol. 2011;27:887–91.
Bai SC, Ok IH, Park, et al. Development of modeling system for assessing essential amino acid requirements using surgically modified rainbow trout. J Aquacult. 2003;16:1–7.
Borlongan IG, Benitez LV. Quantitative lysine requirement of milkfish Chanos chanos juveniles. Aquaculture. 1990;87:341–7.
Brown PB, Davis DA, Robinson EH. An estimate of the dietary lysine requirement of juvenile red drum Sciaenops ocellatus. J World Aquac Soc. 1988;19:109–12.
Cowey CB. Protein and amino acid requirements: a critique of methods. J Appl Ichthyol. 1995;11:199–204.
Dabrowski K. Postprandial distribution of free amino acids between plasma and erythrocytes of common carp Cyprinus carpio. Comp Biochem Physiol. 1982;72A:753–63.
De Silva SS, Gunasekera RM, Gooley G. Digestibility and amino acid availability of three protein-rich ingredient-incorporateddiets by Murray cod Maccullochella peelii and the Australian shortfin eel Anguilla australis. Aquac Res. 2000;31:195–205.
Espe M, Lied E, Torrissen KR. Changes in plasma and muscle free amino acids in Atlantic salmon Salmo salar during absorption of diets containing different amounts of hydrolysed cod muscle protein. Comp Biochem Physiol. 1993;105A:555–62.
Forster I, Ogata HY. Lysine requirement of juvenile Japanese flounder Paralichthys olivaceus and juvenile red sea bream Pagrus major. Aquaculture. 1998;161:131–42.
Harris LE. Feedstuffs. In: Pillay TVR, editor. Fish feed technology. Rome, Italy: UNDP/FAO; 1980. p. 111–68.
Houston AH. Blood and circulation. In: Schreck CB, Moyle PB, editors. Methods for fish biology. New York, USA: American Fisheries Society; 1990. p. 273–343.
Ketola HG. Requirement for dietary lysine and arginine by fry of rainbow trout. J Anim Sci. 1983;56:101–7.
Kim KI, Kayes TB, Amundson CH. Requirements for lysine and arginine by rainbow trout Oncorhynchus mykiss. Aquaculture. 1992;106:333–44.
Kim KI. Reevaluation of protein and amino acid requirements of rainbow trout Oncorhynchus mykiss. Aquaculture. 1997;151:3–7.
Luo Z, Liu YJ, Mai, et al. Quantitative L-lysine requirement of juvenile grouper Epinephelus coioides. Aquacult Nutr. 2006;12:165–72.
Mai KS, Zhang L, Aiet, et al. Dietary lysine requirement of juvenile Japanese seabass Lateolabrax japanicus. Aquaculture. 2006;258:535–42.
Marcouli PA, Alexis MN, Andriopoulouet, et al. Dietary lysine requirement of juvenile gilthead seabream Sparus aurata L. Aquacult Nutr. 2006;12:25–33.
Murai T, Ogata H, Hirasawa Y, Akiyama, et al. Portal absorption and hepatic uptake of amino acids in rainbow trout force fed complete diets containing casein or crystalline amino acids. Nippon Suisan Gakkaishi. 1987;53:1847–59.
Nose T. Changes in patterns of free plasma amino acids in rainbow trout after feeding. Bull Freshwater Fish Res Lab. 1972;22:137–44.
Ogino C. Requirement of carp and rainbow trout for essential amino acids. Bull Jap Soc Sci Fish. 1980;46:171–4.
Ok IH, Bai SC, Park, et al. The patterns of plasma free amino acids after force-feeding in rainbow trout Oncorhynchus mykiss with and without dorsal aorta cannulation. Aquacult Res. 2001;32:70–5.
Ok IH. Determination of essential amino acid requirements by using plasma free amino acid concentrations in rainbow trout Oncorhynchus mykiss. Ph. D. Thesis. Busan, Korea: Pukyong National University; 2002.
Park GJ, Bai SC, Ok, et al. Post prandial plasma free arginine concentrations increase in rainbow trout fed arginine deficient diets. Asian-Australas J Anim Sci. 2005;18:396–402.
Plakas SM, Katayama T, Tanaka Y, Deshimaru O. Changes in the levels of circulating plasma free amino acids of carp Cyprinus carpio after feeding a protein and an amino acid diet of similar composition. Aquaculture. 1980;21:307–22.
Robbins KR, Norton HW, Baker DH. Estimation of nutrient requirements from growth data. J Nutr. 1979;109:1710–4.
Rodehutscord M, Becker A, Pack M, et al. Response of rainbow trout Oncorhynchus mykiss to supplements of individual essential amino acids in a semipurified diet, including an estimate of the maintenance requirement of essential amino acids. J Nutr. 1997;126:1166–75.
Ruchimat T, Masumoto T, Hosokawa, et al. Quantitative lysine requirement of yellowtail Seriola quinqueradiata. Aquaculture. 1997;158:331–9.
Schuhmacher A, Wax C, Gropp JM. Plasma amino acids in rainbow trout Oncorhynchus mykiss fed intact protein or a crystalline amino acid. Aquaculture. 1997;151:15–28.
Simmons L, Moccia RD, Bureau, et al. Dietary methionine requirement of juvenile Arctic charr Salvelinus alpinus. Aquac Nutr. 1999;5:93–100.
Small BC, Soares Jr JH. Quantitative dietary lysine requirement of juvenile striped bass Morones axatilis. Aquacult Nutr. 2000;6:207–12.
Sunde J, Kiessling A, Higgs. Evaluation of feed protein quality by measuring plasma free amino acids in Atlantic salmon Salmo salara fterdorsal aorta cannulation. Aquacult Nutr. 2003;9:351–60.
Thebault H, Alliota E, Pastoureauda A. Quantitative methionine requirement of juvenile sea-bass Dicentrarchus labrax. Aquaculture. 1985;50:75–87.
Tibaldi E, Tulli F. Dietary threonine requirement of juvenile Europeansea bass Dicentrarchus labrax. Aquaculture. 1999;175:155–66.
Vermeirssen ELM, Scott AP, Liley NR. Female rainbow trout urine contains a pheromone which causes a rapid rise in plasma 17α, 20β-dihydroxy-4-pregnen-3-one levels and milt amounts in males. J Fish Biol. 1997;50:107–19.
Walton MJ, Wilson R. Postprandial changes in plasma and liver free amino acids of rainbow trout fed complete diets containing casein. Aquaculture. 1986;51:105–15.
Walton MJ, Cowey CB, Adron JW. The effect of dietary lysine levels on growth and metabolism of rainbow trout Salmo gairdneri. Br J Nutr. 1984;52:115–22.
Wang S, Liua YJ, Tianet, et al. Quantitative dietary lysine requirement of juvenile grass carp Ctenopharyngodon idella. Aquaculture. 2005;249:419–29.
Wilson RP, Poe WE, Robinson EH. Leucine, isoleucine, valine and histidine requirements of fingerling channel catfish. J Nutr. 1980;110:627–33.
Wilson RP. Amino acids and proteins. In: Halver JE, Hardy RW, editors. Fish nutrition. 3rd ed. New York, USA: Academic; 2002. p. 144–75.
Zhou QC, Wu ZH, Chi SY, Yang QH. Dietary lysine requirement of juvenile cobia Rachycentron canadum. Aquaculture. 2007;273:634–40.
Zicker SC, Rogers QR. Use of plasma amino acid and metabolic diseases in veterinary medicine. In: Kaneko JJ, ed. Proceedings of IVth Congress of the International Society for Animal Clinical Biochemistry. Davis, USA: University of California; 1990.
Acknowledgements
This research was supported by the funds of the Feeds and Foods Nutrition Research Center, Korea Sea Grant Program and Brain Korea 21 Program at Pukyoung National University, Busan, Korea.
Author information
Authors and Affiliations
Corresponding author
Additional information
Competing interests
The authors declare that they have no competing interests.
Authors’ contribution
HY carried out the study, sample analyses and writing draft of the manuscript. GP carried out the experiment and statistical analysis. IO participated in design of the experiment, making diet and conducted experiment. KK involved in writing draft of the manuscript and amino acid analysis of samples. SH participated in the design of the study and helped to prepare draft manuscript. SB conceived of the study, and participated in its design and helped to review and finalized the manuscript. All authors read and approved the final manuscript.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Yun, H., Park, G., Ok, I. et al. Determination of the dietary lysine requirement by measuring plasma free lysine concentrations in rainbow trout Oncorhynchus mykiss after dorsal aorta cannulation. Fish Aquatic Sci 19, 4 (2016). https://doi.org/10.1186/s41240-016-0004-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s41240-016-0004-1