Abstract
Background
In Phase 2/3 studies of basal insulin peglispro (BIL) compared to insulin glargine, patients with type 1 or type 2 diabetes previously treated with insulin and randomized to BIL had an increase in serum triglycerides (TGs). To further understand lipoprotein changes, a lipid substudy which included liver fat content was designed to assess relationships among the measured variables for each diabetes cohort and compare the hepato-preferential insulin BIL to glargine.
Methods
In three cohorts of patients with diabetes (type 1, type 2 insulin naïve, and type 2 previously on insulin; n = 652), liver fat content (LFC) was determined by magnetic resonance imaging (MRI) and blood lipids were analyzed by nuclear magnetic resonance (NMR) spectroscopy at baseline, 26 and 52 weeks of treatment. Apolipoproteins, adiponectin, and other lipid parameters were also measured. Descriptive statistics were done, as well as correlation analyses to look for relationships among LFC and lipoproteins or other lipid measures.
Results
In patients with type 1 diabetes treated with BIL, but not glargine, small LDL and medium and large VLDL subclass concentrations increased from baseline. In patients with type 2 diabetes previously on insulin and treated with BIL, large VLDL concentration increased from baseline. In insulin naïve patients with type 2 diabetes treated with BIL, there were very few changes, while in those treated with glargine, small LDL and large VLDL decreased from baseline. Baseline LFC correlated significantly in one or more cohorts with baseline large VLDL, small LDL, VLDL size, and Apo C3. Changes in LFC by treatment showed generally weak correlations with lipoprotein changes, except for positive correlations with large VLDL and VLDL size. Adiponectin was higher in patients with type 1 diabetes compared to patients with type 2 diabetes, but decreased with treatment with both BIL and glargine.
Conclusions
The lipoprotein changes were in line with the observed changes in serum TGs; i.e., the cohorts experiencing increased TGs and LFC with BIL treatment had decreased LDL size and increased VLDL size. These data and analyses add to the currently available information on the metabolic effects of insulins in a very carefully characterized cohort of patients with diabetes.
Clinicaltrials.gov registration numbers and dates NCT01481779 (2011), NCT01435616 (2011), NCT01454284 (2011), NCT01582451 (2012)
Similar content being viewed by others
Background
In diabetes and insulin resistance, the dysregulation of lipid metabolism can be detected in altered levels of circulating lipids. The introduction of insulin therapy may improve lipid metabolism and circulating lipids, raising HDL-cholesterol and lowering triglycerides (TGs) in patients with type 2 diabetes [1, 2]. Proton nuclear magnetic resonance (NMR) spectroscopy offers a more detailed look at changes in lipid metabolism by measuring individual lipoprotein subclasses. There are limited data on the effects of insulin on lipoprotein subclasses in type 1 or type 2 diabetes.
Basal insulin peglispro (BIL) is a novel long-acting insulin analog with a large hydrodynamic size [3] and hepato-preferential action due to reduced peripheral effect [4, 5]. This reduced peripheral effect on glucose disposal suggests a reduced effect on lipogenesis; increased lipolysis in insulin-treated patients who were changed to BIL likely contributed to the observed lipid changes. In patients previously on basal insulin and randomized to BIL in Phase 2 studies, an increase from baseline in serum TGs was noted [6, 7]. In the Phase 3 program, serial lipid profiles were obtained to further characterize and understand the time course for the potential changes in lipid metabolism in response to BIL [8]. In the overall BIL program at baseline, patients with type 2 diabetes on insulin had nominally lower TG values (mean range 144–149 mg/dL) than insulin naïve patients with type 2 diabetes (mean range 159–161 mg/dL); patients with type 1 diabetes had lower TG values (mean range 83–91 mg/dL) than patients with type 2 diabetes. At 26 weeks of treatment, patients with type 2 diabetes randomized to insulin glargine had reductions in mean TG (insulin naïve: −11 mg/dL; prior insulin use: <4 mg/dL); patients with type 1 diabetes had reductions of <2.0 mg/dL. At 26 weeks of follow-up, patients randomized to BIL had increases in TG (insulin naïve: 3.6 mg/dL; all patients previously treated with insulin and then randomized to BIL: 19‒24 mg/dL).
A subset of patients from four Phase 3 studies representing different patient cohorts were recruited to participate in a lipid substudy; these patients are the focus of this report. They had blood samples analyzed by NMR for lipoprotein particle subclass concentration and size determination [9] and underwent assessment of liver fat content (LFC) by MRI [10]. Additional parameters, such as plasma fasting free fatty acids (FFA), apolipoproteins, serum total cholesterol efflux capacity (CEC), cholesterol ester transfer protein (CETP) mass and activity, and plasma adiponectin, were also measured.
The primary LFC and routine blood lipid results are reported elsewhere [8, 11]. Here we report the results of the lipoprotein particle and related lipid analyses by cohort (type 1 diabetes, type 2 insulin naïve, or type 2 previously on insulin) and treatment (insulin glargine or BIL) at baseline and follow-up. We also examine the relationships among lipoprotein particles and LFC.
The primary objective of these substudies was to compare the treatment effects of BIL vs. glargine on the change from baseline in LDL particle concentration at 52 weeks. Secondary objectives included comparing the concentration and change from baseline of LDL particles, HDL particles, intermediate density lipoprotein (IDL) particles, and very low density lipoprotein (VLDL) particles, as well as the values, changes from baseline, and associations with liver fat content of the following parameters: serum total CEC, serum FFA, CETP activity and mass, apolipoproteins [Apo A1, Apo A2, Apo B100 (calculated using Apo B total and Apo B48), Apo C3], and total adiponectin. Changes in particle size of LDL, HDL, and VLDL were also compared. The objectives and results of the LFC study have been reported elsewhere [11, 12].
Methods
Patients and studies
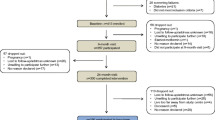
The lipid/MRI substudy was conducted as protocol addenda to four Phase 3 clinical studies of BIL compared to glargine in patients with type 1 or type 2 diabetes [13,14,15,16]. Three patient cohorts were represented (type 1 diabetes [IMAGINE 1 and IMAGINE 3], type 2 diabetes previously taking insulin (and then randomized to either glargine or BIL, described as the “basal switch” cohort [IMAGINE 5]), and type 2 diabetes insulin naïve [IMAGINE 2]). All four studies were multinational and randomized; IMAGINE 2 and IMAGINE 3 were double-blind. Common features of the Phase 3 studies included intensive insulin adjustment with similar basal (and bolus in type 1 diabetes) insulin adjustment algorithms, patient inclusion and exclusion criteria, no changes in lipid-altering medications up to week 12, and stable doses of background glucose-lowering medications before and during the study. Exclusion criteria included fasting hypertriglyceridemia (defined as serum TGs >400 mg/dL). In addition, if a patient developed elevated fasting TG >600 mg/dL at any time during the trial, the patient was discontinued from the study treatment.
Laboratory methods
For all of the analyses in the lipid substudy, blood was collected after an approximately ≥10-h fast. Lipoprotein particle concentrations and sizes were measured at Liposcience (now Laboratory Corporation of America Holdings, Morrisville, NC, USA) by NMR LipoProfile analysis using the LP2 algorithm as previously described [9]. The liver fat content was measured by MRI as previously described [11]. Global CEC was performed by Vascular Strategies LLC, Plymouth Meeting, PA, USA as previously described [17, 18]. CETP mass (ELISA) and activity (fluorometric assay) were determined by Pacific Biomarkers, Seattle, WA, USA. Apo A1, Apo B and Apo B48 were determined by nephelometry by Siemens Healthcare GmgH, Erlangen, Germany. Apo-B100 was calculated as the difference between Apo-B and Apo-B48. The following assays were performed by Covance, Princeton, NJ, USA: serum free fatty acids (enzymatic), Apo A2 and Apo C3 (turbidometric immunoassay), and adiponectin (Quantikine Human Adiponectin/Acrp30 Immunoassay, R&D Systems, Minneapolis, MN).
Liver fat content (LFC) measurements
LFC was evaluated by MRI. For each subject, all scans were obtained using the same scanning techniques, equipment, and imaging parameters as at baseline. The entire liver was scanned with 3–5 overlapping series in the axial plane. LFC was estimated using six-echo images with spectral model and T2* correction, at sequential alternating out-of-phase and in-phase echo times, as previously described [10]. To ensure consistent interpretation of scans, phantoms containing liquid of varying fat fractions were used for quality control, and study images were centrally evaluated by a qualified vendor (Virtual Scopics Inc. Rochester, NY, USA).
Statistical methods
Statistical software used was SAS version 9.1 or higher (SAS Institute Inc. Cary, NC, USA). Analyses were conducted on all randomized patients in the lipid substudy who took at least one dose of study insulin. A mixed-model repeated measures (MMRM) model was used to analyze continuous variables collected at multiple post-treatment time points with terms for treatment, baseline values of the analysis variables, stratification factors for randomization, week, and treatment by week interactions. Values are presented as least squares mean (LSM) ± standard error (SE) unless otherwise noted. All treatment differences are reported as LSM difference (BIL-glargine) with 95% confidence intervals (CI). For treatment comparison at baseline, an analysis of variance model was used for continuous variables and Fisher’s exact test for categorical outcomes. Spearman’s correlation analyses were performed to assess the relationships between NMR parameters and LFC. To adjust for multiplicity, statistical significance is defined as two-sided p value <0.001. Raw data was analyzed as it was collected without transformation or exclusion of outliers. Missing data was handled through MMRM analysis without explicit imputation.
Results
Patients
A total of 652 patients from 4 Phase 3 studies of BIL versus glargine comprised the full analysis set for the lipid substudy; 219 were from the glargine arms and 433 from the BIL arms. Baseline demographic and clinical characteristics, NMR lipoproteins, and other lipid parameters for the three patient cohorts are summarized in Table 1. Within each patient cohort there were no major differences between treatment groups at baseline. Six patients in the lipid substudy discontinued from study treatment because of a post-baseline fasting TG >600 mg/dL, per the protocol; all were in the BIL treatment group.
Comparing the type 1 cohort to the combined type 2 cohorts, the type 1 diabetes patient cohort was about 20 years younger and had numerically lower body mass index (BMI), higher HDL-C, and lower serum TGs (Table 1). The type 1 diabetes cohort also differed from the type 2 diabetes cohort at baseline in having numerically lower mean LFC, higher large and medium HDL, lower small HDL, higher large and lower small LDL, and lower VLDL subclass concentrations, and higher adiponectin levels (Table 1). Baseline values for some of the lipoprotein subclass and other lipid parameters were positively correlated with the baseline LFC (Table 2). The correlations reaching statistical significance were with large VLDL (all cohorts), Apo A2 (type 1 cohort), and Apo C3 (type 1 and type 2 basal switch cohorts).
Type 1 diabetes—effects of glargine and BIL
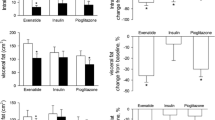
Patients treated with glargine had no change from baseline in LDL particle concentration at 52 weeks, either in total LDL or in the large or small LDL subclasses (Fig. 1; Additional file 1: Table S1). In contrast, patients treated with BIL had a significant increase from baseline in total LDL at 52 weeks (Fig. 1; Additional file 1: Table S1). Among the LDL subclasses, large LDL concentrations were not significantly different between treatments at 26 or 52 weeks, although concentrations decreased significantly from baseline with BIL treatment at 26 weeks. However, small LDL concentrations showed significant increases from baseline with BIL treatment at both 26 and 52 weeks (Fig. 1; Additional file 1: Table S1). LDL particle size did not change with glargine treatment, but decreased significantly with BIL treatment from baseline to 26 and 52 weeks (Fig. 2; Additional file 1: Table S1).
Patients treated with glargine had no change in VLDL particle concentration over time, either in total VLDL or in the large, medium, or small VLDL subclasses (Fig. 3; Additional file 1: Table S1). Patients treated with BIL had significant increases from baseline in large and medium VLDL subclasses (Fig. 3; Additional file 1: Table S1). Total HDL particle concentration was not significantly different with BIL vs. glargine treatment, but large HDL decreased with BIL (Fig. 4; Additional file 1: Table S1).
Patients treated with glargine had no change in mean LFC, while patients treated with BIL had a mean increase in LFC [LSM difference at 52 weeks: 2.20% (1.26–3.13%); p < 0.001] (Additional file 1: Table S1) [8]. Positive correlations with changes in LFC were found for changes in medium HDL, large VLDL, and VLDL size, but only large VLDL in the glargine group correlated significantly (Table 3).
Patients treated with glargine had no significant changes in the apolipoproteins measured (Fig. 5; Additional file 1: Table S1). Patients treated with BIL had significant increases from baseline in Apo A2, Apo B100, and Apo C3, with significant treatment differences at 26 and 52 weeks for Apo C3 (Fig. 5; Additional file 1: Table S1). There were no significant changes in CETP or CEC. Adiponectin concentrations decreased significantly from baseline to 52 weeks with both glargine and BIL treatment, with no significant difference between treatments (Fig. 6; Additional file 1: Table S1).
Type 2 diabetes—effects of glargine and BIL
The two cohorts of type 2 diabetes patients were similar in baseline characteristics, with the exception that LFC was numerically higher in the insulin naïve cohort (Table 1). The insulin naïve cohort had a numerically higher concentration of large VLDL compared to the cohort previously taking insulin (basal switch) (Table 1).
Insulin naïve cohort
Total LDL concentrations decreased from baseline with glargine treatment; the change was largely in the small LDL subclass (Fig. 1; Additional file 1: Table S1). Large VLDL decreased from baseline with glargine treatment and was significantly lower compared to the BIL group at 52 weeks (Fig. 3; Additional file 1: Table S1). Compared to baseline, VLDL size decreased while LDL size increased with glargine treatment (Fig. 2; Additional file 1: Table S1). Total HDL concentrations were unchanged with glargine treatment but decreased from baseline with BIL treatment (Fig. 4; Additional file 1: Table S1).
Mean LFC decreased from baseline in patients treated with glargine, while there was no significant change with BIL treatment [LSM difference at 52 weeks: 2.57% (0.94–4.21%); p = 0.002] (Additional file 1: Table S1) [8]. Stronger correlations were found for changes in lipoprotein parameters with changes in LFC in patients taking BIL in this population (Table 3). The strongest correlations were with total LDL, IDL, small LDL, total VLDL, small VLDL, Apo A2, Apo B100, and Apo C3 [all positive; five had correlation coefficients (r) > 0.3].
There were no significant changes from baseline or treatment differences in any measured apolipoproteins in either treatment group at 26 or 52 weeks (Fig. 5; Additional file 1: Table S1). FFAs decreased significantly from baseline in both treatment groups, but this decrease was significantly greater with glargine vs. BIL (Additional file 1: Table S1). There were no significant changes in CETP or CEC. Adiponectin concentrations increased with glargine and decreased with BIL, but these changes were not significant (Fig. 6).
Basal switch cohort
Patients previously on insulin that were randomized to glargine did not have any significant changes from baseline to 52 weeks in total LDL or any LDL subclass during treatment (Fig. 1; Additional file 1: Table S1), or in total VLDL or any VLDL subclass (Fig. 4). Patients assigned to BIL had mean increases from baseline in the large VLDL subclass which were significantly different from the glargine group at 26 weeks (Fig. 3; Additional file 1: Table S1). The glargine group had no changes from baseline in concentrations of HDL or HDL subclass particles. The BIL group had a mean decrease from baseline in total HDL and small HDL particles, but there were no significant differences between treatments at any endpoint and there was a difference at baseline (Fig. 4; Additional file 1: Table S1).
Mean LFC did not change in the glargine group, but increased from baseline in the BIL group [LSM difference at 52 weeks: 5.27% (3.43–7.11%); p < 0.001] (Additional file 1: Table S1) [8]. Some correlations were found for changes in lipoprotein parameters with changes in LFC in patients taking BIL (Table 3). The highest correlations were with small LDL, large VLDL, and Apo B100 (all positive), but none of r values were >0.3.
Patients treated with glargine had no significant change from baseline in any measured apolipoproteins. The BIL group had a significant increase from baseline in Apo C3 at 52 weeks (Fig. 5; Additional file 1: Table S1). There were no significant changes in CETP or CEC. Adiponectin concentrations declined with both BIL and glargine treatment, but the change from baseline was statistically significant only with BIL at 26 weeks (Fig. 6; Additional file 1: Table S1).
Discussion
The effects of currently available insulins on lipid profiles have been summarized by Chaudhuri [2]. Data from the Phase 3 clinical program for BIL—compared to insulin glargine or NPH—have also recently been reported [8]. However, the effects of insulin therapies on NMR-determined lipoprotein subclasses is limited to selected cohorts of patients with type 1 diabetes [19,20,21,22,23,24] and very small studies in type 2 diabetes [25,26,27,28,29]. What follows is a discussion of our findings in the context of the available literature.
Lipoprotein subclasses in type 1 diabetes
Glargine treatment had little effect on LDL particles; BIL treatment led to increases from baseline in total LDL, small LDL, and Apo B100, but a decrease in large LDL. Glargine did not affect VLDL particles; BIL treatment led to increases from baseline in medium and large VLDL (similar in diameter range to the VLDL1 subfraction [30]), and an increase in Apo C3. Medium HDL was (nominally) lower with glargine, but increased with BIL treatment. These changes are consistent with the increase in serum TGs noted with BIL treatment in the Phase 2/3 clinical program [8].
In a cross-sectional study, patients with type 1 diabetes compared to healthy controls had lower medium VLDL, lower small HDL, and larger HDL size, with higher concentrations of large HDL [21]. Whether these differences were due to insulin treatment is not known. The effects of intensive insulin therapy on NMR lipoproteins were evaluated during the Diabetes Complications and Control Trial (DCCT) [24]. The results showed that intensive insulin therapy was associated with larger LDL diameter and lower levels of small LDL and small HDL.
The effects of these differences on coronary artery disease (CAD) risk were studied by Erbey et al. [20] in the Pittsburgh Epidemiology Study of Diabetes Complications (EDC). In a large cohort of patients with type 1 diabetes (n = 337), higher concentrations of small, dense LDL were associated with higher cholesterol, TGs, total LDL and lower HDL-C, and an increased risk of CAD. Soedamah et al. [22] also studied subjects with type 1 diabetes from the EDC cohort in a nested case (CAD, n = 59) control study (non-CAD, n = 59). In univariate analyses, lipid mass and particle concentrations of all three VLDL subclasses, and small and medium LDL were higher in cases than controls, while large HDL concentration was lower. Medium HDL was higher in patients with CAD and in the multivariate model was associated with CAD. Lyons et al. [23] evaluated lipoprotein subclasses and relationships with carotid intima media thickness (CIMT) in DCCT/EDIC participants. In analyses that adjusted for multiple variables, LDL particle concentrations were associated with internal CIMT in both sexes; LDL-C and Apo B were also associated with CIMT.
In our study, patients with type 1 diabetes treated with glargine did not have significant changes in any lipoprotein subclasses, presumably because the study treatment was similar to the patients’ pre-study treatment. In patients treated with BIL, lipoprotein changes were suggestive of an increased risk for CVD (although the magnitude of the change in CVD risk is uncertain). There were too few cardiovascular events in the BIL program to make assessments of any relationships with lipoprotein subclasses [31].
Lipoprotein subclasses in type 2 diabetes
In the type 2 diabetes insulin naïve cohort, glargine use was associated with nominal decreases from baseline in total and small LDL and large VLDL; these changes were not seen with BIL treatment. In contrast, patients with type 2 diabetes previously on insulin that were randomized to glargine (basal switch cohort) did not have these changes, suggesting effects of prior insulin treatment. Randomization to BIL treatment was associated with increased large VLDL in the basal switch cohort, which was concordant with the observed changes in TGs [8].
Several small, short term studies evaluated insulin effects on lipid subfractions in type 2 diabetes, which showed that insulin treatment was associated with lower concentrations of TG, VLDL, small LDL, and small HDL, as well as increased activity of adipose tissue lipoprotein lipase (LPL) and CETP, and decreased activity of hepatic lipase [26,27,28,29, 32]. These changes are consistent with the known effects of insulin in lowering circulating TG [2], and the subsequent effects of TG levels on lipoproteins [33].
Although the effects of conventional insulin on serum TGs have been previously reported [2], data for the effects of insulin on VLDL particles are limited. In the overall BIL program, TGs increased from baseline when patients who were previous taking insulin were switched to BIL, but not when insulin naïve patients were treated with BIL [8]. In the current study, changes in large VLDL most closely paralleled these observations (as did, to a lesser extent, VLDL size), suggesting that the observed differences between glargine and BIL hinged on changes in the large VLDL subclass. Glargine has a greater peripheral insulin effect than BIL and thus would be expected to increase peripheral lipogenesis, while reducing lipolysis [4]. This may be mediated via increased fatty acid uptake subsequent to increased LPL activity [25]. BIL would have less effect on peripheral lipogenesis, and would allow increased peripheral lipolysis compared to glargine, which would likely result in increased FFA levels and hepatic VLDL production. The studies noted above did not report surrogate imaging markers of CVD to assess effects of lipid subfractions on CVD risk. In the BIL Phase 3 program, there were too few clinical CV events in patients with type 2 diabetes to assess any relationship of these lipid fractions with clinical disease [31].
Adiponectin
There is extensive literature describing adiponectin as an adipokine that is associated with insulin sensitivity and vascular wall anti-inflammatory activity in type 2 diabetes, and showing that lower levels of adiponectin are associated with insulin resistance in type 2 diabetes [34,35,36,37,38]. However, there are only limited data in patients with type 1 diabetes. In the current study, the type 1 diabetes cohort had much higher mean adiponectin levels than the type 2 diabetes cohorts at baseline. Other investigators have reported that adiponectin levels are higher in patients with type 1 diabetes compared to non-diabetes controls [39,40,41,42,43]. The observations from the current study that adiponectin levels decreased from baseline with both BIL and glargine treatment are also concordant with literature showing that adiponectin levels decline with intensification of insulin treatment [42,43,44]. In summary, the current data confirm that patients with type 1 diabetes may have high adiponectin levels, but whether lower levels of adiponectin are associated with insulin resistance or clinical cardiovascular disease cannot be confirmed.
Associations with liver fat content
Increased LFC has been associated with increased risk for CVD [45,46,47]. Whether this is the result of “common soil” with other markers of insulin resistance is unclear. However, the data from the current study characterizes some of the associations between LFC and the NMR profile of lipoproteins and apolipoproteins.
Liver fat content was assessed at baseline and after insulin treatment in these diabetes cohorts [12]. In correlations of baseline LFC with baseline values of lipoproteins and other soluble biomarkers, we found that large VLDL, VLDL size, Apo C3, and small LDL had the strongest and most consistent positive correlations with LFC, with r values of 0.216‒0.460 across the diabetes cohorts. There were also positive correlations with small HDL and Apo B100, and a negative correlation with adiponectin. These parameters are all associated with features of insulin resistance.
These observations support the concept that LFC is a function of insulin resistance not only in type 2 diabetes, but also in type 1 diabetes. The negative baseline correlations of LFC with adiponectin, an insulin-sensitizing and anti-steatotic adipokine, are consistent with previous observations that lower levels of adiponectin are associated with higher LFC [48]. Of note, even in the type 1 diabetes cohort that had a much higher baseline adiponectin compared to the type 2 diabetes cohorts, the negative baseline relationship of adiponectin to LFC was present. These data suggest that even with lower LFC in type 1 diabetes than in type 2 diabetes, some of the same mechanisms of insulin resistance may be operative.
Because glargine treatment reduced LFC in insulin naïve patients, and patients randomized to BIL who had previously been on insulin had increases in LFC, we examined the correlations between changes in the various lipid parameters, change in LFC, and treatment (Table 3). In patients with type 1 diabetes treated with glargine, there was very little absolute change in LFC and few major correlations of LFC change with lipoprotein changes, except for a direct association with large VLDL. In insulin naïve patients with type 2 diabetes treated with glargine, there was a decrease in LFC that correlated positively with changes in VLDL measures; this effect was even stronger in patients with type 2 diabetes previously treated with insulin, where there was only a small decrease in LFC with glargine.
These observations suggest that no single lipoprotein subclass, apolipoprotein or even adiponectin is a major driver of change in LFC in these insulin-treated patients with diabetes, although large VLDL, VLDL size, Apo B100, and Apo C3 may be related in some scenarios.
Strengths and limitations
The strengths of this study include the carefully characterized subgroup of patients from a large Phase 3 program in which three cohorts of patients with different diabetes types were studied [5]. There was uniform collection of serial lipid profiles and LFC, measures of glycemic efficacy, insulin dosing and adverse event profiles. The MRI data for LFC and the lipid and lipoprotein measures were all collected in standardized fashion across the substudy cohort. Background glucose- and lipid-lowering medications were stable before randomization and during the study and did not differ between treatment groups.
Limitations of this study include: (1) The substudy cohort was not representative of the whole Phase 3 cohort in terms of randomization and outcomes, as the ability to perform NMR/MRI was limited to certain study sites. (2) The study cohort is too small to do extensive multivariable analyses of whether such things as sex, glycemic control, background glucose-lowering medications, lipid-lowering medications, or changes in body weight affected lipids and lipoproteins or LFC. (3) Although elevated TG (and VLDL TG) and nonalcoholic fatty liver have been associated with hepatic insulin resistance [49, 50], we did not obtain insulin levels needed to calculate insulin resistance because of limited ability to interpret plasma insulin concentrations in insulin-treated patients. (4) Although we did multiplicity adjustments, the results for individual lipoprotein parameters should be interpreted with caution. (5) Although in patients treated with BIL lipoprotein particle and apoprotein changes were suggestive of increased CVD risk (although the magnitude of the change in CVD risk is uncertain), analyses related to clinical CVD risk or events were not feasible in this study. (6) Individual studies did not capture all of the details of all potential variables that might affect lipid values, including menopausal status and hormone replacement therapy.
Conclusions
In conclusion, the data from the lipid substudy of the BIL Phase 3 program in insulin-treated cohorts of both type 1 and type 2 diabetes, with multiple measures of soluble biomarkers related to lipid/lipoprotein concentrations, provide a unique and extensive database on the effects of insulin glargine and an investigational hepato-preferential insulin (BIL). These data, particularly the demonstration of differences between insulin naïve and previously insulin-treated patients with type 2 diabetes, may be helpful toward understanding the effects of future insulin treatments [51] that may exhibit varying peripheral and hepatic effects on lipid metabolism and possibly CVD risk.
References
Chaudhuri A, Rosenstock J, DiGenio A, Meneghini L, Hollander P, McGill JB, Dandona P, Ilgenfritz J, Riddle M. Comparing the effects of insulin glargine and thiazolidinediones on plasma lipids in type 2 diabetes: a patient-level pooled analysis. Diabetes Metab Res Rev. 2012;28(3):258–67.
Chaudhuri A, Dandona P. Effects of insulin and other antihyperglycaemic agents on lipid profiles of patients with diabetes. Diabetes Obes Metab. 2011;13(10):869–79.
Hansen RJ, Cutler GB, Vick A, Koester A, Li S, Siesky AM, Beals JM. LY2605541: leveraging hydrodynamic size to develop a novel basal insulin. Diabetes. 2012;61(Suppl 1):A228.
Mudaliar S, Henry RR, Ciaraldi TP, Armstrong DA, Burke PM, Pettus JH, Garhyan P, Choi SL, Knadler MP, Lam EC, et al. Reduced peripheral activity leading to hepato-preferential action of basal insulin peglispro compared with insulin glargine in patients with type 1 diabetes. Diabetes Obes Metab. 2016;18(Suppl 2):17–24.
Jacober SJ, Prince MJ, Beals JM, Hartman ML, Qu Y, Linnebjerg H, Garhyan P, Haupt A. Basal insulin peglispro: overview of a novel long-acting insulin with reduced peripheral effect resulting in a hepato-preferential action. Diabetes Obes Metab. 2016;18(Suppl 2):3–16.
Bergenstal RM, Rosenstock J, Arakaki RF, Prince MJ, Qu Y, Sinha VP, Howey DC, Jacober SJ. A randomized, controlled study of once-daily LY2605541, a novel long-acting basal insulin, versus insulin glargine in basal insulin-treated patients with type 2 diabetes. Diabetes Care. 2012;35(11):2140–7.
Rosenstock J, Bergenstal RM, Blevins TC, Morrow LA, Prince MJ, Qu Y, Sinha VP, Howey DC, Jacober SJ. Better glycemic control and weight loss with the novel long-acting basal insulin LY2605541 compared with insulin glargine in type 1 diabetes: a randomized, crossover study. Diabetes Care. 2013;36(3):522–8.
Ginsberg H, Cariou B, Orchard T, Chen L, Luo J, Bastyr EJ 3rd, Bue-Valleskey J, Chang AM, Ivanyi T, Jacober SJ, et al. Lipid changes during basal insulin peglispro, insulin glargine, or NPH treatment in six IMAGINE trials. Diabetes Obes Metab. 2016;18(11):1089–92.
Jeyarajah EJ, Cromwell WC, Otvos JD. Lipoprotein particle analysis by nuclear magnetic resonance spectroscopy. Clin Lab Med. 2006;26(4):847–70.
Mashhood A, Railkar R, Yokoo T, Levin Y, Clark L, Fox-Bosetti S, Middleton MS, Riek J, Kauh E, Dardzinski BJ, et al. Reproducibility of hepatic fat fraction measurement by magnetic resonance imaging. J Magn Reson Imaging. 2013;37(6):1359–70.
Cusi K, Sanyal AJ, Zhang S, Hoogwerf BJ, Chang AM, Jacober SJ, Bue-Valleskey JM, Higdon AN, Bastyr EJ 3rd, Haupt A, et al. Different effects of basal insulin peglispro and insulin glargine on liver enzymes and liver fat content in patients with type 1 and type 2 diabetes. Diabetes Obes Metab. 2016;18(Suppl 2):50–8.
Cusi K, Sanyal A, Zhang S, Hartman ML, Bue-Valleskey J, Hoogwerf BJ, Haupt A. NAFLD prevalence and its metabolic associations in patients with type 1 diabetes and type 2 diabetes. Diabetes Obes Metab. 2017. doi:10.1111/dom.12973.
Davies MJ, Russell-Jones D, Selam JL, Bailey TS, Kerenyi Z, Luo J, Bue-Valleskey J, Ivanyi T, Hartman ML, Jacobson JG, et al. Basal insulin peglispro versus insulin glargine in insulin-naive type 2 diabetes: IMAGINE 2 randomized trial. Diabetes Obes Metab. 2016;18(11):1055–64.
Garg S, Dreyer M, Jinnouchi H, Mou J, Qu Y, Hartman ML, Rosilio M, Jacober SJ, Bastyr EJ 3rd, Investigators IT. A randomized clinical trial comparing basal insulin peglispro and insulin glargine, in combination with prandial insulin lispro, in patients with type 1 diabetes: IMAGINE 1. Diabetes Obes Metab. 2016;18(Suppl 2):25–33.
Bergenstal RM, Lunt H, Franek E, Travert F, Mou J, Qu Y, Antalis CJ, Hartman ML, Rosilio M, Jacober SJ, et al. Randomized, double-blind clinical trial comparing basal insulin peglispro and insulin glargine, in combination with prandial insulin lispro, in patients with type 1 diabetes: IMAGINE 3. Diabetes Obes Metab. 2016;18(11):1081–8.
Buse JB, Rodbard HW, Trescoli Serrano C, Luo J, Ivanyi T, Bue-Valleskey J, Hartman ML, Carey MA, Chang AM. Randomized clinical trial comparing basal insulin peglispro and insulin glargine in patients with type 2 diabetes previously treated with basal insulin: IMAGINE 5. Diabetes Care. 2016;39(1):92–100.
de la Llera-Moya M, Drazul-Schrader D, Asztalos BF, Cuchel M, Rader DJ, Rothblat GH. The ability to promote efflux via ABCA1 determines the capacity of serum specimens with similar high-density lipoprotein cholesterol to remove cholesterol from macrophages. Arterioscler Thromb Vasc Biol. 2010;30(4):796–801.
Kempen HJ, Gomaraschi M, Bellibas SE, Plassmann S, Zerler B, Collins HL, Adelman SJ, Calabresi L, Wijngaard PL. Effect of repeated apoA-IMilano/POPC infusion on lipids, (apo)lipoproteins, and serum cholesterol efflux capacity in cynomolgus monkeys. J Lipid Res. 2013;54(9):2341–53.
Purnell JQ, Marcovina SM, Hokanson JE, Kennedy H, Cleary PA, Steffes MW, Brunzell JD. Levels of lipoprotein(a), apolipoprotein B, and lipoprotein cholesterol distribution in IDDM. Results from follow-up in the diabetes control and complications trial. Diabetes. 1995;44(10):1218–26.
Erbey JR, Robbins D, Forrest KY, Orchard TJ. Low-density lipoprotein particle size and coronary artery disease in a childhood-onset type 1 diabetes population. Metabolism. 1999;48(4):531–4.
Colhoun HM, Otvos JD, Rubens MB, Taskinen MR, Underwood SR, Fuller JH. Lipoprotein subclasses and particle sizes and their relationship with coronary artery calcification in men and women with and without type 1 diabetes. Diabetes. 2002;51(6):1949–56.
Soedamah-Muthu SS, Chang YF, Otvos J, Evans RW, Orchard TJ, Pittsburgh Epidemiology of Diabetes Complications S. Lipoprotein subclass measurements by nuclear magnetic resonance spectroscopy improve the prediction of coronary artery disease in Type 1 diabetes. A prospective report from the Pittsburgh Epidemiology of Diabetes Complications Study. Diabetologia. 2003;46(5):674–82.
Lyons TJ, Jenkins AJ, Zheng D, Klein RL, Otvos JD, Yu Y, Lackland DT, McGee D, McHenry MB, Lopes-Virella M, et al. Nuclear magnetic resonance-determined lipoprotein subclass profile in the DCCT/EDIC cohort: associations with carotid intima-media thickness. Diabet Med. 2006;23(9):955–66.
Zhang Y, Jenkins AJ, Basu A, Stoner JA, Lopes-Virella MF, Klein RL, Group DER, Lyons TJ. Associations between intensive diabetes therapy and NMR-determined lipoprotein subclass profiles in type 1 diabetes. J Lipid Res. 2016;57(2):310–7.
Taskinen MR, Kuusi T, Helve E, Nikkila EA, Yki-Jarvinen H. Insulin therapy induces antiatherogenic changes of serum lipoproteins in noninsulin-dependent diabetes. Arteriosclerosis. 1988;8(2):168–77.
Romano G, Patti L, Innelli F, Di Marino L, Annuzzi G, Iavicoli M, Coronel GA, Riccardi G, Rivellese AA. Insulin and sulfonylurea therapy in NIDDM patients. Are the effects on lipoprotein metabolism different even with similar blood glucose control? Diabetes. 1997;46(10):1601–6.
Rivellese AA, Patti L, Romano G, Innelli F, Di Marino L, Annuzzi G, Iavicoli M, Coronel GA, Riccardi G. Effect of insulin and sulfonylurea therapy, at the same level of blood glucose control, on low density lipoprotein subfractions in type 2 diabetic patients. J Clin Endocrinol Metab. 2000;85(11):4188–92.
Koska J, Saremi A, Bahn G, Yamashita S, Reaven PD, Veterans Affairs Diabetes Trial I. The effect of intensive glucose lowering on lipoprotein particle profiles and inflammatory markers in the Veterans Affairs Diabetes Trial (VADT). Diabetes Care. 2013;36(8):2408–14.
Aslan I, Kucuksayan E, Aslan M. Effect of insulin analog initiation therapy on LDL/HDL subfraction profile and HDL associated enzymes in type 2 diabetic patients. Lipids Health Dis. 2013;12:54.
Adiels M, Olofsson SO, Taskinen MR, Boren J. Overproduction of very low-density lipoproteins is the hallmark of the dyslipidemia in the metabolic syndrome. Arterioscler Thromb Vasc Biol. 2008;28(7):1225–36.
Hoogwerf BJ, Lincoff AM, Rodriguez A, Chen L, Qu Y. Major adverse cardiovascular events with basal insulin peglispro versus comparator insulins in patients with type 1 or type 2 diabetes: a meta-analysis. Cardiovasc Diabetol. 2016;15(1):78.
Taskinen MR, Boren J. New insights into the pathophysiology of dyslipidemia in type 2 diabetes. Atherosclerosis. 2015;239(2):483–95.
Ginsberg HN. Insulin resistance and cardiovascular disease. J Clin Invest. 2000;106(4):453–8.
Whitehead JP, Richards AA, Hickman IJ, Macdonald GA, Prins JB. Adiponectin—a key adipokine in the metabolic syndrome. Diabetes Obes Metab. 2006;8(3):264–80.
Tishinsky JM, Robinson LE, Dyck DJ. Insulin-sensitizing properties of adiponectin. Biochimie. 2012;94(10):2131–6.
Cui J, Panse S, Falkner B. The role of adiponectin in metabolic and vascular disease: a review. Clin Nephrol. 2011;75(1):26–33.
Wu Z, Cheng Y, Aung LH, Li B. Association between adiponectin concentrations and cardiovascular disease in diabetic patients: a systematic review and meta-analysis. PLoS ONE. 2013;8(11):e78485.
Weyer C, Funahashi T, Tanaka S, Hotta K, Matsuzawa Y, Pratley RE, Tataranni PA. Hypoadiponectinemia in obesity and type 2 diabetes: close association with insulin resistance and hyperinsulinemia. J Clin Endocrinol Metab. 2001;86(5):1930–5.
Galler A, Gelbrich G, Kratzsch J, Noack N, Kapellen T, Kiess W. Elevated serum levels of adiponectin in children, adolescents and young adults with type 1 diabetes and the impact of age, gender, body mass index and metabolic control: a longitudinal study. Eur J Endocrinol. 2007;157(4):481–9.
Heilman K, Zilmer M, Zilmer K, Kool P, Tillmann V. Elevated plasma adiponectin and decreased plasma homocysteine and asymmetric dimethylarginine in children with type 1 diabetes. Scand J Clin Lab Invest. 2009;69(1):85–91.
Imagawa A, Funahashi T, Nakamura T, Moriwaki M, Tanaka S, Nishizawa H, Sayama K, Uno S, Iwahashi H, Yamagata K, et al. Elevated serum concentration of adipose-derived factor, adiponectin, in patients with type 1 diabetes. Diabetes Care. 2002;25(9):1665–6.
Maahs DM, Ogden LG, Snell-Bergeon JK, Kinney GL, Wadwa RP, Hokanson JE, Dabelea D, Kretowski A, Eckel RH, Rewers M. Determinants of serum adiponectin in persons with and without type 1 diabetes. Am J Epidemiol. 2007;166(6):731–40.
Perseghin G, Lattuada G, Danna M, Sereni LP, Maffi P, De Cobelli F, Battezzati A, Secchi A, Del Maschio A, Luzi L. Insulin resistance, intramyocellular lipid content, and plasma adiponectin in patients with type 1 diabetes. Am J Physiol Endocrinol Metab. 2003;285(6):E1174–81.
Timar R, Timar B, Degeratu D, Serafinceanu C, Oancea C. Metabolic syndrome, adiponectin and proinflammatory status in patients with type 1 diabetes mellitus. J Int Med Res. 2014;42(5):1131–8.
Wolff L, Bos D, Murad SD, Franco OH, Krestin GP, Hofman A, Vernooij MW, van der Lugt A. Liver fat is related to cardiovascular risk factors and subclinical vascular disease: the Rotterdam Study. Eur Heart J Cardiovasc Imaging. 2016;17(12):1361–7.
Arulanandan A, Ang B, Bettencourt R, Hooker J, Behling C, Lin GY, Valasek MA, Ix JH, Schnabl B, Sirlin CB, et al. Association between quantity of liver fat and cardiovascular risk in patients with nonalcoholic fatty liver disease independent of nonalcoholic steatohepatitis. Clin Gastroenterol Hepatol. 2015;13(8):1513–20.
Lonardo A, Ballestri S, Guaraldi G, Nascimbeni F, Romagnoli D, Zona S, Targher G. Fatty liver is associated with an increased risk of diabetes and cardiovascular disease—Evidence from three different disease models: NAFLD, HCV and HIV. World J Gastroenterol. 2016;22(44):9674–93.
Targher G, Bertolini L, Scala L, Poli F, Zenari L, Falezza G. Decreased plasma adiponectin concentrations are closely associated with nonalcoholic hepatic steatosis in obese individuals. Clin Endocrinol (Oxf). 2004;61(6):700–3.
Bril F, Sninsky JJ, Baca AM, Superko HR, Portillo Sanchez P, Biernacki D, Maximos M, Lomonaco R, Orsak B, Suman A, et al. Hepatic steatosis and insulin resistance, but not steatohepatitis, promote atherogenic dyslipidemia in NAFLD. J Clin Endocrinol Metab. 2016;101(2):644–52.
Leon-Acuna A, Alcala-Diaz JF, Delgado-Lista J, Torres-Pena JD, Lopez-Moreno J, Camargo A, Garcia-Rios A, Marin C, Gomez-Delgado F, Caballero J, et al. Hepatic insulin resistance both in prediabetic and diabetic patients determines postprandial lipoprotein metabolism: from the CORDIOPREV study. Cardiovasc Diabetol. 2016;15:68.
Russell-Jones DL. Hepato-preferential insulins: Is this the end, or the end of the beginning? Diabetes Obes Metab. 2016;18(11):1053–4.
Authors’ contributions
BH, TI, and SZ conceived and designed the study, and analyzed and interpreted the data. TO and BC were involved in the collection and interpretation of the data. MC and JO contributed to the interpretation of the data. BH and CA drafted the article. All authors revised the article critically for important intellectual content. All authors read and approved the final manuscript.
Acknowledgements
Not applicable.
Competing interests
TO received an honorarium from Eli Lilly and Company for advisory board membership. BC has received research funding from Sanofi and Regeneron Pharmaceuticals, Inc. and Pfizer; and honoraria from Amgen, AstraZeneca, Pierre Fabre, Janssen, Eli Lilly and Company, MSD Merck & Co. Novo Nordisk, Regeneron, Sanofi, and Takeda. MC and JO are employees of LabCorp. SZ, CA, TI and BH are employees and minor shareholders of Eli Lilly and Company.
Ethics approval and consent to participate
To participate in the lipid substudy, patients enrolled in the Phase 3 studies provided additional written informed consent. Protocols and informed consent documents were approved by local ethical review boards.
Funding
This study was funded by Eli Lilly and Company.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Author information
Authors and Affiliations
Corresponding author
Additional file
12933_2017_555_MOESM1_ESM.docx
Additional file 1: Table S1. Change in LFC, NMR lipoproteins, and other parameters from baseline to 26 and 52 weeks of treatment.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Orchard, T.J., Cariou, B., Connelly, M.A. et al. The effects of basal insulin peglispro vs. insulin glargine on lipoprotein particles by NMR and liver fat content by MRI in patients with diabetes. Cardiovasc Diabetol 16, 73 (2017). https://doi.org/10.1186/s12933-017-0555-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12933-017-0555-1