Abstract
Background
Salmonella enterica serovar Enteritidis (SE) is one of the food-borne pathogenic bacteria, which affects poultry production and poses severe threat to human health. The correlation of immune system and metabolism in chicken after SE inoculation is important but not clear. In the current study, we identified the expression of immune and energy metabolism related genes using quantitative PCR to evaluate the correlation between immune system and energy metabolism against SE inoculation in Jining Bairi chicken.
Results
ATP5G1, ATP5G3 and ND2 were significantly up-regulated at 1 dpi (day post inoculation), and ATP5E, ATP5G1, ATP5G3 were significantly down-regulated at 7 dpi (P < 0.05). IL-8 and IL-1β were significantly down-regulated at 1 dpi, IL-8 and IL-18 were significantly down-regulated at 3 dpi, IL-8 and BCL10 were significantly up-regulated at 7 dpi (P < 0.05).
Conclusions
These findings indicate that the correlation between immune and energy metabolism related genes gradually change with time points post SE inoculation, from one homeostasis to an opposite homeostasis with 3 dpi as a turning point. These results will pave the foundation for the relationship between immune system and energy metabolism in the response to SE inoculation in chicken.
Similar content being viewed by others
Background
Salmonella enterica serovar Enteritidis (S. Enteritidis) is frequently associated with food-borne disease in the world which can infect a wide range of hosts and frequently reach the human food chain causing food-borne disease [1]. In China, Salmonella causes 70–80% of foodborne bacterial outbreaks and the primary vehicles of transmission are raw or undercooked chicken [2]. Salmonella is estimated to be responsible for approximate 1 million cases of illness and more than 450 deaths annually in the United States [3]. Poultry is considered as a primary source of foodborne diseases, especially Salmonella Enteritidis infection. S. Enteritidis is responsible for 36% of Salmonella outbreaks in the United States [4]. The losses caused by egg-related salmonellosis in Australia have reached $44 million annually [5]. S. Enteritidis causes significant economic losses to the poultry industry due to the mortality, nutrient malabsorption, retarded growth rate, and decreased egg production [1].
The response to S. Enteritidis infection is regulated through altering gene expression. Many genes have been disclosed in the response to S. Enteritidis infection [6,7,8]. Toll-like receptor (TLR)-mediated recognition of pathogens is now thought to have a crucial role in innate and adaptive immune [9]. Subsets of immune and inflammatory cells interact via interleukins (ILs) and IFNs [10]. TLRs and ILs have been reported to be associated with S. Enteritidis infection in chicken [11,12,13,14,15]. It has been reported that complex networks that intimately connect metabolism and immunity in human [16]. Wu et al. proved that the complicated interaction between the immune system and metabolism contributes to the immune responses to SE inoculation of layers at 14 dpi at the onset of lay [17].
Metabolism, a series of orderly chemical reactions, controls production, maintenance, destruction of biomolecules, and distribution of energy to organisms [16]. Energy metabolism is the process of generating energy (ATP) from nutrients. Mitochondria are the primary energy-generating system in most eukaryotic cells and participate in intermediary metabolism, oxidative phosphorylation, calcium signaling, and apoptosis [18]. Genes from the mitochondrial respiratory chain complex gene family provide instructions for proteins involved in oxidative phosphorylation. Five protein complexes (I-V) are involved in this process. Mitochondrial complex V consists of 19 genes in human. The function of genes involved in complex I and V has been reported, such as mt-ND2 [19], ATP5E [20], and ATP5G3 [21, 22]. Subunit 2 of NADH dehydrogenase (ND2) is one of genes comprising of mitochondrial complex I [23], which plays a critical role in controlling the production of the mitochondrial reactive oxygen species in chicken [24]. Mt-ND2 gene was extensively expressed in tissues, and the expression was affected by dietary fat types and chicken age [19]. The altered expression of mt-ND2 is critical to multiple disease in human [25, 26]. ATP synthase epsilon subunit (ATP5E) gene was found to encode the mitochondrial F0F1 ATP synthase subunit epsilon [27]. In addition, ATP5E was proven to be required for normal spindle orientation during embryonic divisions in Drosophila [28]. ATP5G1, a key component of complex V of the oxidative phosphorylation chain, encodes a subunit of mitochondrial ATP synthase and catalyzes ATP synthesis [29] and is involved in the biological process of oxidative phosphorylation and might be associated with oxidative stress [30]. The expression of ATP5G1 and ATP5G3 is actively regulated in response to various physiological stimuli, and maintain the basal levels of the c-Fo subunit [31]. However, little information is available on the regulation of S. Enteritidis inoculation of energy metabolism related genes and their correlation with immune related genes.
In this study, we aim to investigate the correlation between immunity and energy metabolism in chicken in the response to SE inoculation. The mRNA expression of immune and energy metabolism related genes after SE inoculation in chicken spleen will be analyzed.
Results
The expression of energy metabolism related genes in spleen
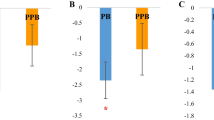
The expression of ATP5E, ATP5G1, ATP5G3 and ND2 in control and treat group was shown in Fig. 1. At 1 dpi, the expression level of ATP5G1, ATP5G3 and ND2 in the treat group was significantly higher than that in the control group with fold change of 1.29, 1.99 and 2.87, respectively (P < 0.05) (Fig. 1b, c and d). At 7dpi, the expression of ATP5E, ATP5G1 and ATP5G3 in the treat group was significantly lower than that in the control group with a fold change of 0.77, 0.54 and 0.69, respectively (P < 0.05) (Fig. 1a, b and c).
Relative mRNA expression of energy metabolism related genes in spleen between control and treat chickens. The relative expression was normalized with the control at the same time point following SE inoculation. * indicated P < 0.05, ** indicated P < 0.01. Different letters within each group indicate statistical difference (P < 0.05)
Within the treat group, ATP5G1 and ATP5G3 were significantly decreased across the different time points (P < 0.05) (Fig. 1b and c). The expression of ATP5G1 and ATP5G3 was significantly higher at 3 dpi compared to that at 7 dpi (P < 0.05) (Fig. 1b and c). Within the control group, the expression of ATP5E and ATP5G1 was significantly higher at 3 dpi than that at 1 dpi (P < 0.05) (Fig. 1a and b). The expression of ATP5G1 at 7 dpi was the lowest across 3 different time points (P < 0.05) (Fig. 1b).
The expression of immune-related genes
The expression of immune-related genes was shown in Fig. 2. At 1 and 3 dpi, the expression of IL-8 in the treat group was significantly lower compared with that in the control group with a fold change of 0.03 and 0.23, respectively (P < 0.05) (Fig. 2a). At 3 dpi, the expression of IL-18 in the treat group was significantly lower compared with that in the control group (P < 0.05) (Fig. 2b). At 7 dpi, the expression of IL-8 and BCL10 in the treat group was significantly higher compared with that in the control group with a fold change of 6.56 and 1.50, respectively (P < 0.05) (Fig. 2a and c). At 1 and 7 dpi, the expression of IL-1β in the treat group was significantly lower compared with that in the control group with a fold change of 0.42 and 0.35, respectively (P < 0.05) (Fig. 2d). At 1 dpi, the expression of TLR1LA in the treat group was significantly higher compared with that in the control group with a fold change of 2.17 (P < 0.01) (Fig. 2e).
Relative mRNA expression of immune-related genes in spleen between control and treat chickens. The relative expression was normalized with the control at the same time point following SE inoculation. *indicated P < 0.05, ∗∗ indicated P < 0.01. IL = interleukin. Different letters within each group indicate statistical difference (P < 0.05)
Within the treat group, IL-8 at 1 dpi was significantly lower compared to that at 7 dpi (P < 0.05) (Fig. 2a). IL-1β at 7 dpi was significantly lower compared to that at 1 and 3 dpi (P < 0.05) (Fig. 2d). TLR1LA at 1 dpi was significantly higher compared to that at 7 dpi (P < 0.05) (Fig. 2e).
Within the control group, IL-18 at 3 dpi was significantly higher compared to that at 1 and 7dpi (P < 0.05) (Fig. 2b). BCL10 at 1 dpi was significantly higher compared to that at 7 dpi (P < 0.05) (Fig. 2c). IL-1β at 3 dpi was significantly higher compared to that at 1 and 7 dpi (P < 0.05) (Fig. 2d). TLR1LA at 1 dpi was significantly lower than that at 3 and 7 dpi (P < 0.05) (Fig. 2e).
Correlative expression of immune-related and energy metabolism related genes following SE inoculation
At 1 dpi, the expression of ATP5E, ATP5G1, ATP5G3 and ND2 in the treat group was higher than that in the control group, however, the expression of IL-1β, IL-8, IL-18 and BCL10 in the treat group was lower than that in the control group (Fig. 3). At 3 dpi, the expression of ATP5E, ATP5G3, TLR1A and IL-1β was higher while the expression of ATP5G1, ND2, IL-8, IL-18 and BCL10 was lower in the treat group compared with that in the control group (Fig. 3). At 7 dpi, with respect to the control group, the expression of ATP5E, ATP5G1, ATP5G3 and ND2 was lower in the treat group, while the expression of IL-8, IL-18, BCL10 and TLR1LA was higher in the treat group (Fig. 3).
Discussion
The correlative expression of metabolism related genes and immune related genes has been studied in the current study. The mitochondrial ATP synthase activity plays a key role in energy production in mammals [32]. ATP5G1, ATP5G2 and ATP5G3 encode the mitochondrial ATP synthase c-Fo subunit [33]. ATP5G1 gene expression is actively regulated in response to various physiological stimuli (such as ontogenic development and cold acclimation), whereas the ATP5G2 and ATP5G3 maintain the basal levels of the c-Fo subunit [33, 34]. However, few studies focused on the energy metabolism in chicken inoculated with SE. In the current study, we found SE inoculation altered the expression of four energy metabolism related genes at different stages. Knockdown of ATP5G1 results in reduced ATP levels and oxidative phosphorylation activity, and increases the amount of reactive oxygen species [35]. It has been reported that ATP5G1 involves in immune response and impaired immunoproteasome assembly via PA28–20S-PA28 and PA700–20S-PA28 complexes [36,37,38,39]. The up-regulated ATP5G3 gene switches the Warburg effect to oxidative phosphorylation with slowing energy production rate and inhibiting cancer cells growth [40]. At day 1 post SE inoculation, the expression ATP5G1 36 and ATP5G3 was up-regulated in the current study (P < 0.05). Mitochondria may be coping with the SE infection through up-regulated energy metabolism related gens at the early period of inoculation. The down-regulation of ATP5E is directly related with mitochondrial injury, and leads to less energy production [41]. Additionally, ATP5E, ATP5G1, ATP5G3 and ATP5E were down-regulated in papillary thyroid carcinoma (PTC) patient and ATP5E was highly associated to PTC diagnosis [42]. In the current study, ATP5G1 (0.77-fold), ATP5G3 (0.54-fold) and ATP5E (0.6-fold) were down-regulated at 7 days post SE inoculation which indicates the SE inoculation lead to mitochondrial injury and reduce energy production at later period of inoculation.
Toll-like receptors are a family of germ line-encoded pattern recognition receptors which activate rapid inflammatory responses upon detection of their cognate ligands [43]. TLR1LA is needed for the recognition of bacterial lipoprotein and LPS and is widely expressed among chicken tissues [44, 45]. In chickens, TLR1.1 and TLR1.2, two members of the TLR1/TLR6/TLR10 family, can recognize lipoproteins and peptidoglycan, respectively [46, 47]. TLR1 was significantly up-regulated in chicken intestine at 2 and 4 days post H9N2 influenza virus [48]. It has been reported that TLR1LA was significantly up-regulated in the cecum (3-fold) and the ileum (2.6-fold) at 24 h post SE infection in 2-day-old chickens [49]. A similar increase in TLR1LA (2.7-fold) was also observed in chicken spleen at 1 day post SE inoculation in the current study. The data demonstrate that increased expression of TLR1LA in various tissues may play a role in facilitating increased recognition of lipoproteins and peptidoglycan.
BCL10 is an intracellular signaling protein which positively regulates lymphocyte proliferation by connecting antigen receptor induced signals of B and T cells to the activation of the transcription factor NF-κB [50, 51]. Interleukin 8 (IL-8) is the major chemoattractant and activator for neutrophils and a key component of innate immunity [52]. BCL10 was significantly down-regulated in cecum at 7 days post SE inoculation in White Leghorn [53]. In the current study, SE inoculation induced both BCL10 (1.5-fold) and IL-8 (6.56-fold) in spleen at 7 dpi. The increased expression of immune-related genes would facilitate limiting pathogens further proliferation. IL-8 and IL-1β are two major pro-inflammatory cytokines which play a central role in initiation of inflammatory responses against bacterial-and viral-infections [54]. In the current study, IL-1β was up-regulated at 3dpi, which was supported by the expression in chicken cecum following SE inoculation [15]. IL-8 and IL-18 in cecum were significantly increased at 3 days following S. Enteritidis infection [15], which was inconsistent with our study probably due to different genetic background and tissues. IL-18 provides an important link between the innate and adaptive immune responses [55] and is associated with immune organ damage of Avian reovirus and immune suppression [56]. BCL10 is a mediator of LPS-induced activation of IL-8 in human intestinal epithelial cells [57]. These results indicated SE inoculation could be regulated by BCL10 through mediating IL-8 at 7 dpi and induced immune damage in chicken spleen.
The immune responses are highly energy dependent process and energy metabolism is also involved in the immune networking for self-defense and against pathophysiology [58]. Various stresses may increase energy demands for immune regulation at the expense of energy reserves, and then reduce the energy available for growth and other processes [59, 60]. Furthermore, disease and nutrition are closely interlinked [61]. The immune variations under ambient pressures may trigger energy metabolic changes in mollusks [59, 62]. It has been reported that interaction between the immune system and metabolism contributes to the immune responses of egg-type chicken to SE inoculation at the onset of lay [17]. In the current results, IL-8 was down-and up- regulated at 1 and 7 days post SE inoculation. ATP5G1 and ATP5G3 were up-and down-regulated at 1 and 7 days post SE inoculation. These results suggested there existed a negative correlation between IL-8 and ATP5G1, ATP5G3 at 1 and 7 days post SE inoculation. The correlation between immune related genes and energy metabolism related genes was multidirectional at 3 dpi. These findings indicated that the correlation between immune and energy metabolism related genes gradually change with time points post SE inoculation, from one homeostasis to an opposite homeostasis. Day 3 post SE inoculation maybe a key point to regulate the correlation between immune system and energy metabolism in the response to SE inoculation. The findings in the current study would deepen the understanding of comprehensive interaction between immune system and energy metabolism in the response to SE inoculation in chicken.
Conclusions
These findings indicated that the correlation between immune and energy metabolism related genes gradually change with time points post SE inoculation, from one homeostasis to an opposite homeostasis. Day 3 post SE inoculation maybe a key point to regulate the correlation between immune system and energy metabolism in the response to SE inoculation. These results provided the foundation for the relationship between immune system and energy metabolism in the response to SE inoculation in chicken.
Methods
Animals and sample collection
Jining Bairi Chicken, a Chinese local chicken breed used in the current study was provided by Shandong Bairi Chicken Breeding Co., Ltd. (Shandong, China). The S. Enteritidis strain (CVCC3377) used in the current study was purchased from the China Veterinary Culture Collection Center (Beijing, China). To make the inoculant, SE were enriched in LB broth at 37℃ for 16 h, pelleted at 4,000 rpm for 5 min, and diluted with sterilized PBS. The concentration of SE in the inoculant was measured by the plating method. The animal trial was performed as described previously [6]. In brief, 168 two-day-old S. Enteritidis–negative chickens were randomly into 2 groups with 84 chickens in each of treat and control groups. Chickens in treat and control groups were raised in 2 separate incubators with the same environmental conditions and free access to sterilized feed and water. Each chicken in the treat group was orally inoculated with 0.3 ml of 109 colony-forming units (cfu)/ml SE inoculant. Each chicken in the control group inoculated with 0.3 ml sterile phosphate buffer saline (PBS). Twelve chickens from each of the treat and control groups were euthanized by cervical dislocation for sample collection at 1, 3, 7, 14, 21, 28 and 35 days post-inoculation. Cecum content was collected for SE enumeration. The chicken mortality was zero in the treat group. Chickens in the control group were SE negative. Spleen samples were collected and stored at -80 °C for further utilization. Three individual spleen samples in the treat group with the highest bacterial number and three random individual spleen samples in the control group at each of 1, 3 and 7 dpi were used in the current study. All animal procedures were approved by Shandong Agricultural University Animal Care and Use Committee.
Total RNA extraction and cDNA synthesis
Total RNA was extracted from each individual spleen sample using TRIzol reagent (Invitrogen, US) following the manufacturer’s instructions and stored at -80℃ until further use. In total, 18 RNA samples were used in the current study. The quantity and quality of RNA sample was evaluated using DS-11 Spectrophotometer (DeNovix, US) and gel electrophoresis, respectively. Total RNA was digested with DNase I to eliminate the genomic DNA and reverse transcribed into the cDNA using Transcriptor First Strand cDNA Synthesis Kit (Perfect Real Time) (Takara, Dalian, China) according to the manufacturer’s instructions. The 20 µL reaction system was used and contained total RNA 1 µg, 5 × Primer Script Buffer (for Real Time) 4 µL, Primer Script RT Enzyme Mix I 1 µL, Random 6 mers (100 µM) 1 µL, and adding RNase-free ddH2O to 20 µL. The reaction condition was 37 ℃ for 15 min, 85 ℃ for 5 s.
Quantitative real time-polymerase chain reaction (qRT-PCR)
ATP5E, ATP5G1, ATP5G3, ND2, BLC10, TLR1A, IL-1β, IL-8 and IL-18 were selected for qRT-PCR. Beta-actin (β-actin) was used as the reference gene to adjust relative quantification of the target genes. The specific primer sequences of all genes were designed using Primer Premier 5.0 and listed in Table 1. The qRT-PCR was performed using the Applied Biosystems 7500 Fast system (Applied Biosystems, US) with a SYBR®Premix Ex Taq™ Kit (Roche, US). A reaction mixture (20 µL) consisted of cDNA 2 µL, forward primer 0.5 µL, reverse primer 0.5 µL, SYBR Green Master (Mix) 10 µL, and ddH2O 7 µL. The qRT-PCR amplification condition was as follows: 50℃ for 2 min, 95℃ for 10 min, followed by 40 cycles at 95℃ for 15 s, 60 ℃ for 1 min, finally, a single melt cycle was 95℃ for 15 s and 65℃ for 1 min. Triplicate was performed for each cDNA sample to run the qRT-PCR.
Statistical analysis
The relative quantification of target gene was determined using the 2−ΔΔCt method [63]. The difference of expression level for each gene between treat and control groups within each time point was analyzed using student T-test, across different time points within treat or control group were analyzed using the general linear model procedure of SAS 8.1 (SAS Institute, Cary, NC). P < 0.05 was considered significant difference.
Abbreviations
- SE:
-
Salmonella enterica serovar Enteritidis
- BCL10:
-
B-cell CLL/lymphoma 10
- TLR:
-
Toll-like receptor
- IL:
-
Interleukin
- PBS:
-
Phosphate buffer saline
- qRT-PCR:
-
Quantitative real time-polymerase chain reaction
- β-actin:
-
Beta-actin
References
Betancor L, Pereira M, Martinez A, Giossa G, Fookes M, Flores K, Barrios P, Repiso V, Vignoli R, Cordeiro N. Prevalence of Salmonella enterica in poultry and eggs in Uruguay during an epidemic due to Salmonella enterica serovar Enteritidis. J Clin Microbiol. 2010;48(7):2413–23. .
Yang B, Qu D, Zhang X, Shen J, Cui S, Shi Y, Xi M, Sheng M, Zhi S, Meng J. Prevalence and characterization of Salmonella serovars in retail meats of marketplace in Shaanxi, China. Int J Food Microbiol. 2010;141(1–2):63–72. .
Scallan E, Griffin PM, Angulo FJ, Tauxe RV, Hoekstra RM. Foodborne Illness Acquired in the United States—Unspecified Agents. Emerg Infect Dis. 2011;17(1):16–22. .
Guard-Petter J. The chicken, the egg and Salmonella enteritidis. Environ Microbiol. 2010;3(7):421–30. .
Samiullah CKK, Roberts JR, Sexton M, May D, Kiermeier A. Effects of egg shell quality and washing on Salmonella Infantis penetration. Int J Food Microbiol. 2013;165(2):77–83. .
Liu L, Lin L, Zheng L, Hui T, Fan X, Xue N, Min L, Min L, Li X. Cecal microbiome profile altered by Salmonella enterica, serovar Enteritidis inoculation in chicken. Gut Pathogens. 2018;10(1):34-.
Babu US, Raybourne RB. Impact of dietary components on chicken immune system and Salmonella infection. Expert Rev Anti Infect Ther. 2008;6(1):121–35.
Matulova M, Varmuzova K, Sisak F, Havlickova H, Babak V, Stejskal K, Zdrahal Z, Rychlik I. Chicken innate immune response to oral infection with Salmonella enterica serovar Enteritidis. Vet Res. 2013;44(1):1–11.
Trinchieri G, Sher A. Cooperation of Toll-like receptor signals in innate immune defence. Nat Rev Immunol. 2007;7(3):179–90. .
Akdis M. Interleukins, from 1 to 37, and interferon-γ: receptors, functions, and roles in diseases. J Allergy Clin Immunol. 2011;127(3):701-21.e770. .
Kaiser MG, Cheeseman JH, Kaiser P, Lamont SJ. Cytokine expression in chicken peripheral blood mononuclear cells after in vitro exposure to Salmonella enterica serovar Enteritidis. Poult Sci. 2006;85(11):1907–11.
Kogut MH, Tellez GI, Mcgruder ED, Hargis BM, Deloach JR. Heterophils are decisive components in the early responses of chickens to SE infections. Microb Pathog. 1994;16(2):141–51. .
Nerren JR, Swaggerty CL, Mackinnon KM, Genovese KJ, He H, Pevzner I, Kogut MH. Differential mRNA expression of the avian-specific toll-like receptor 15 between heterophils from Salmonella-susceptible and-resistant chickens. Immunogenetics. 2009;61(1):71–7.
Gary L, Vincenzo F, Shaun M, Smith AL, Nat B, Paul B, Loredo-Osti JC, Kenneth M, Danielle M. Allelic variation in TLR4 is linked to susceptibility to Salmonella enterica serovar Typhimurium infection in chickens. Infect Immun. 2003;71(3):1116–24. .
Magdalena C, Helena H, Marcela F, Marta M, Hana H, Frantisek S, Ivan R. Immune response of chicken gut to natural colonization by gut microflora and to Salmonella enterica serovar enteritidis infection. Infect Immun. 2011;79(7):2755–63. .
Giuseppe M, Antonio LC. The intricate interface between immune system and metabolism. Trends Immunol. 2004;25(4):193–200. .
Wu G, Liu L, Qi Y, Sun Y, Yang N, Xu G, Zhou H, Li X. Splenic gene expression profiling in White Leghorn layer inoculated with the Salmonella enterica serovar Enteritidis. Anim Genet. 2016;46(6):617–26. .
Chan DC. Mitochondria: dynamic organelles in disease, aging, and development. Cell. 2006;125(7):1241–52.
Zhang W, Hou L, Wang T, Lu W, Tao Y, Chen W, Du X, Huang Y. The expression characteristics of mt-ND2 gene in chicken. Mitochondrial Dna. 2015;27(5):3787–92.
Havlíčková V, Kaplanová V, Nůsková H, Drahota Z, Houštěk J. Knockdown of F 1 epsilon subunit decreases mitochondrial content of ATP synthase and leads to accumulation of subunit c. BBA. 2010;1797(6–7):1124–9. .
Huang Y, Wang L, Bennette B, Williams RW, Wang YJ, Gu WK, Jiao Y. Potential role of Atp5g3 in epigenetic regulation of alcohol preference or obesity from a mouse genomic perspective. Genet Mol Res. 2013;12(3):3662–74. .
Li HS, Zhang JY, Thompson BS, Deng XY, Ford ME, Wood PG, Stolz DB, Eagon PK, Whitcomb DC. Rat mitochondrial ATP synthase ATP5G3: cloning and upregulation in pancreas after chronic ethanol feeding. Physiol Genom. 2001;6(2):91–8. .
Lan D, Hu Y, Zhu Q, Liu Y. Mitochondrial DNA study in domestic chicken. Mitochondrial Dna. 2015;28(1):25–9.
Lu WW, Hou LL, Zhang WW, Zhang PF, Huang Y. Study on heteroplasmic variation and the effect of chicken mitochondrial ND2. Mitochondrial DNA A DNA Mapp Seq Anal. 2014;27(4):1–7. .
Feng S, Xiong L, Ji Z, Cheng W, Yang H. Correlation between increased ND2 expression and demethylated displacement loop of mtDNA in colorectal cancer. Mol Med Rep. 2012;6(1):125–30. .
Chen X, He XY, Zhu C, Zhang Y, Li Z, Liu Y, Zhang Y, Yin T, Li Y. Interaction between mitochondrial NADH dehydrogenase subunit-2 5178 C > A and clinical risk factors on the susceptibility of essential hypertension in Chinese population. BMC Med Genet. 2019;20(1):121.
Huang Y-J, Jan Y-H, Chang Y-C, Tsai H-F, Hsiao M. ATP Synthase Subunit Epsilon Overexpression Promotes Metastasis by Modulating AMPK Signaling to Induce Epithelial-to-Mesenchymal Transition and Is a Poor Prognostic Marker in Colorectal Cancer Patients. J Clin Med. 2019;8(7):1070. .
Kidd T. The ε-Subunit of Mitochondrial ATP Synthase Is Required for Normal Spindle Orientation During the Drosophila Embryonic Divisions. Genetics. 2005;170(2):697–708.
Vives-Bauza C, Magrane J, Andreu AL, Manfredi G. Novel Role of ATPase Subunit C Targeting Peptides Beyond Mitochondrial Protein Import. Mol Biol Cell. 2010;21(1):131–9. .
Hu Y, Chen X, Lin H, Hu Y, Mu X. Study on the antiendotoxin action of Pulsatillae Decoction using an Affymetrix rat genome array. Cell Immunol. 2009;257(1–2):32–7. .
Hou W, Hou YL, Ding X, Wang T. cDNA, genomic sequence cloning and overexpression of giant panda (Ailuropoda melanoleuca) mitochondrial ATP synthase ATP5G1. Genet Mol Res. 2012;11(3):3164–74. .
Das AM. Regulation of the mitochondrial ATP-synthase in health and disease. Mol Genet Metab. 2003;79(2):71–82. .
Anna DG, Cecilia L, Cecilia S. Evolution of ATP synthase subunit c and cytochrome c gene families in selected Metazoan classes. Gene. 2006;371(2):224–33.
Andersson U, Houstek J. B. ATP synthase subunit c expression: Physiological regulation of the P1 and P2 genes. Biochem J. 1997;323(2):379–85. .
Seth R, Keeley J, Abu-Ali G, Crook S, Jackson D, Ilyas M. The putative tumour modifier gene ATP5A1 is not mutated in human colorectal cancer cell lines but expression levels correlate with TP53 mutations and chromosomal instability. J Clin Pathol. 2009;62(7):598–603. .
Kloetzel PM. Generation of major histocompatibility complex class I antigens: functional interplay between proteasomes and TPPII. Nat Immunol. 2004;5(7):661–9. .
Preckel T, Fung-Leung WP, Cai Z, Vitiello A, Salter-Cid L, Winqvist O, Wolfe TG, Herrath M, Von,, Angulo A, Ghazal P. Impaired immunoproteasome assembly and immune responses in PA28-/- mice. Science. 1999;286(5447):2162–5.
Soza A, Knuehl C, Groettrup M, Henklein P, Tanaka K, Kloetzel PM. Expression and subcellular localization of mouse 20S proteasome activator complex PA28. Febs Letters. 1997;413(1):27–34.
Tanahashi N, Murakami Y, Minami Y, Shimbara N, Hendil KB, Tanaka K. Hybrid proteasomes. Induction by interferon-gamma and contribution to ATP-dependent proteolysis. J Biol Chem. 2000;275(19):14336–45. .
Huang YP, Chang NW. PPARα modulates gene expression profiles of mitochondrial energy metabolism in oral tumorigenesis. Biomedicine. 2016;6(1):1–6.
Xia W, Wan Y, Li Y, Zeng H, Lv Z, Li G, Wei Z, Xu S. PFOS prenatal exposure induce mitochondrial injury and gene expression change in hearts of weaned SD rats. Toxicology. 2011;282(1):23–9.
Hurtado-López LM, Fernández-Ramírez F, Martínez-Peñafiel E, Ruiz JDC, González NEH. Molecular Analysis by Gene Expression of Mitochondrial ATPase Subunits in Papillary Thyroid Cancer: Is ATP5E Transcript a Possible Early Tumor Marker? Med Sci Monit. 2015;21:1745–51. .
Fink IR, Forlenza M, Pietretti D, Wiegertjes GF. Facing the Challenge of a Functional Characterazation of Toll-like Receptor (TLR)1 and TLR2 in Common Carp. Fish Shellfish Immunol. 2016;53:70–83. .
Fukui A, Inoue N, Matsumoto M, Nomura M, Yamada K, Matsuda Y, Toyoshima K, Seya T. Molecular cloning and functional characterization of chicken toll-like receptors. A single chicken toll covers multiple molecular patterns. J Biol Chem. 2001;276(50):47143–9. .
Iqbal M, Philbin VJ, Smith AL. Expression patterns of chicken Toll-like receptor mRNA in tissues, immune cell subsets and cell lines. Vet Immunol Immunopathol. 2005;104(1):117–27.
Boyd A, Philbin VJ, Smith AL. Conserved and distinct aspects of the avian Toll-like receptor (TLR) system: implications for transmission and control of bird-borne zoonoses. Biochem Soc Transact. 2007;35(Pt 6):1504–7. .
Higuchi M, Matsuo A, Shingai M, Shida K, Ishii A, Funami K, Suzuki Y, Oshiumi H, Matsumoto M, Seya T. Combinational recognition of bacterial lipoproteins and peptidoglycan by chicken Toll-like receptor 2 subfamily. Dev Comp Immunol. 2008;32(2):147–55. .
Nang NT, Lee JS, Song BM, Kang YM, Kim HS, Seo SH. Induction of inflammatory cytokines and Toll-like receptors in chickens infected with avian H9N2 influenza virus. Vet Res. 2011;42(1):64. .
Mackinnon KM, He H, Nerren JR, Swaggerty CL, Genovese KJ, Kogut MH. Expression profile of toll-like receptors within the gastrointestinal tract of 2-day-old Salmonella enteriditis-infected broiler chickens. Vet Microbiol. 2009;137(3):313–9. .
Thome M, Tschopp J. Bcl10. Current Biology Cb. 2002;12(2):R45.
Donghai W, Yun Y, Pei-Chun L, Liquan X, Morris SW, Hu Z, Renren W, Xin L. Bcl10 plays a critical role in NF-kappaB activation induced by G protein-coupled receptors. Proc Natl Acad Sci USA. 2007;104(1):145–50. .
Wang X, Wang L, Zhang H, Ji Q, Song L, Qiu L, Zhou Z, Wang M, Wang L. Immune response and energy metabolism of Chlamys farreri under Vibrio anguillarum challenge and high temperature exposure. Fish Shellfish Immunol. 2012;33(4):1016–26. .
Wu G, Qi Y, Liu X, Yang N, Xu G, Liu L, Li X. Cecal MicroRNAome response to Salmonella enterica serovar Enteritidis infection in White Leghorn Layer. Bmc Genomics. 2017;18(1):1–13.
Herath HMLPB, Elvitigala DAS, Godahewa GI, Umasuthan N, Whang I, Noh JK, Lee J. Molecular characterization and comparative expression analysis of two teleostean pro-inflammatory cytokines, IL-1β and IL-8, from Sebastes schlegeli. Gene. 2016;575(2):732–42.
Hung LH, Li HP, Lien YY, Wu ML, Chaung HC. Adjuvant effects of chicken interleukin-18 in avian Newcastle disease vaccine. Vaccine. 2010;28(5):1148–55.
Zhang KL, Xie ZX, Huang L, Xie LJ, Liu JB, Deng XW, Xie ZQ, Fan Q, Luo SS. Quantitative detection of IL-18 in immune organs of SPF chicken infected by Avian reovirus. J Southern Agric. 2015;82:134–42. .
Sumit B, Alip B, Nitika P, Dudeja PK, Tobacman JK. Bcl10 mediates LPS-induced activation of NF-kappaB and IL-8 in human intestinal epithelial cells. Am J Physiol Gastrointest Liver Physiol. 2007;293(2):G429. .
Gomes A, Sengupta J, Datta P, Ghosh S, Gomes A. Physiological Interactions of Nanoparticles in Energy Metabolism, Immune Function and Their Biosafety: A Review. J Nanosci Nanotechnol. 2016;16(1):92–116.
Lochmiller RL, Deerenberg C. Trade-offs in evolutionary immunology: just what is the cost of immunity? Oikos. 2000;88(1):87–98.
Adamo SA. How should behavioural ecologists interpret measurements of immunity. Anim Behav. 2004;68(6):1443–9. .
Das S, Palai TK, Mishra SR, Das D, Jena B. Nutrition in Relation to Diseases and Heat stress in Poultry. Vet World. 2011;4(9):429–32. .
Demas GE. The energetics of immunity: a neuroendocrine link between energy balance and immune function. Horm Behav. 2004;45(3):173–80. .
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001;25(4):402–8.
Acknowledgements
We thank Hao Zhang for providing the Jining Bairi Chicken used in the current study.
Funding
This project was supported by the National Natural Science Foundation of China (31872343, 31601980), Natural Science Foundation of Shandong Province (ZR2018MC026), Shandong Modern Agricultural Industry & Technology System (SDAIT-11-02), Shandong Province Agricultural Seed Project (2017LZN007) and Funds of Shandong “Double Tops” Program (SYL2017YSTD12). The funding agencies did not participate in study design, data collection, analysis, interpretation, and writing of the manuscript.
Author information
Authors and Affiliations
Contributions
LL2 and XL designed the study. XM, HL and LL1 performed the animal inoculation trial. YW performed the laboratory procedures and bioinformatics analysis. YW, XM, and HL analyzed and interpreted the data. YW drafted the manuscript. XL, LL2 and PS critically revised the manuscript. All author(s) have read and approved the manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
This study was approved by Shandong Agricultural University Animal Care and Use Committee. All procedures were performed in accordance with the guidelines for the care and use of animals of Shandong Agricultural University and with current Chinese legislation.
Consent for publication
Not applicable.
Availability of data and materials
The data used and/or analyzed in the current study are available from the corresponding author on reasonable request.
Competing Interests
We certify that there is no competing of interest with any financial organization regarding the material discussed in the manuscript.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Wang, Y., Miao, X., Li, H. et al. The correlated expression of immune and energy metabolism related genes in the response to Salmonella enterica serovar Enteritidis inoculation in chicken. BMC Vet Res 16, 257 (2020). https://doi.org/10.1186/s12917-020-02474-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12917-020-02474-5