Abstract
Background
PREDICT Prostate is an endorsed prognostic model that provides individualised long-term prostate cancer-specific and overall survival estimates. The model, derived from UK data, estimates potential treatment benefit on overall survival. In this study, we externally validated the model in a large independent dataset and compared performance to existing models and within treatment groups.
Methods
Men with non-metastatic prostate cancer and prostate-specific antigen (PSA) < 100 ng/ml diagnosed between 2000 and 2010 in the nationwide population-based Prostate Cancer data Base Sweden (PCBaSe) were included. Data on age, PSA, clinical stage, grade group, biopsy involvement, primary treatment and comorbidity were retrieved. Sixty-nine thousand two hundred six men were included with 13.9 years of median follow-up. Fifteen-year survival estimates were calculated using PREDICT Prostate for prostate cancer-specific mortality (PCSM) and all-cause mortality (ACM). Discrimination was assessed using Harrell’s concordance (c)-index in R. Calibration was evaluated using cumulative available follow-up in Stata (TX, USA).
Results
Overall discrimination of PREDICT Prostate was good with c-indices of 0.85 (95% CI 0.85–0.86) for PCSM and 0.79 (95% CI 0.79–0.80) for ACM. Overall calibration of the model was excellent with 25,925 deaths predicted and 25,849 deaths observed. Within the conservative management and radical treatment groups, c-indices for 15-year PCSM were 0.81 and 0.78, respectively. Calibration also remained good within treatment groups. The discrimination of PREDICT Prostate significantly outperformed the EAU, NCCN and CAPRA scores for both PCSM and ACM within this cohort overall.
A key limitation is the use of retrospective cohort data.
Conclusions
This large external validation demonstrates that PREDICT Prostate is a robust and generalisable model to aid clinical decision-making.
Similar content being viewed by others
Background
Prostate cancer represents a growing burden on health care globally, with increasing numbers and proportions of men presenting with non-metastatic prostate cancer (PCa) [1]. Alongside this, there has been increased confidence in the use of conservative management (active surveillance and watchful waiting) [2]. Understanding disease prognosis to guide treatment decision-making is therefore of great importance. However, until recently, no high-quality individualised model for survival existed.
Using data from over 10,000 UK men, we have previously published an individualised prognostic model for cancer-specific and overall survival called ‘PREDICT Prostate’ [3]. PREDICT Prostate (available online [4]) provides cancer-specific and overall percentage survival estimates for up to 15 years and has been endorsed by the National Institute for Health and Care Excellence (NICE) [5]. To maximise usability, it uses routinely available clinico-pathological data (age, PSA, grade, stage, biopsy involvement, treatment type and comorbidity). It represents real-world data from a non-screened, primary diagnostic cohort, including a significant number of men treated conservatively. Crucially, the model also allows adjustment for competing mortalities by incorporating both cancer-specific and non-cancer survival outcomes to contextualise the diagnosis as part of a decision aid. Internal validation and accuracy within a small external population were promising during model development [3]. However, external validation in independent cohorts, ideally in a different location, is vital to demonstrate generalisability and accuracy of a multivariable prognostic model [6].
The Prostate Cancer data Base Sweden (PCBaSe) is one of the largest and most comprehensive prostate cancer cohorts world-wide and is well suited for external validation of PREDICT Prostate [7]. The aim of this study was to validate PREDICT Prostate and compare performance to existing models.
Methods
Source of data
Data from PCBaSE 3.0 were used, according to a pre-specified project outline (Additional file 2) [8,9,10]. PCBaSe was created by the combination of the National Prostate Cancer Register of Sweden with other national healthcare and demographic databases [11]. The capture rate of this register is 98% of all incident prostate cancer cases compared to the Swedish Cancer Registry—to which registration is mandated by law [12]. The cause of death information is updated from the Cause of Death Registry which captures all deaths in Sweden. The agreement between the recorded cause of death and reviewed medical records has been reported at 86% (95% CI 85–87%) [13].
Participants and predictors
We included men within PCBaSe diagnosed with PCa between 1 January 2000 and 31 December 2010, with no evidence of metastatic disease and prostate-specific antigen (PSA) < 100 ng/ml. Cases were censored at death, migration or 31 December 2016, whichever event occurred first. Data were available for 82,936 men. Outcome events were ‘PCa death’ or ‘any-cause death’ from which ‘non-PCa death’ was derived. Intact data were required for variables mandatory within the model: age, PSA, T stage, histological grade group, primary treatment type and comorbidity. This led to the exclusion of 13,730 (16.6%) cases, leaving a final analysable dataset of 69,206 (Table 1). Missing data were most abundant for the histological grade group (n = 8117), as primary and secondary Gleason grades were not always registered. Data were also missing on PSA (n = 2124), T stage (n = 1364), age (n = 4) and primary treatment (n = 3960). Some men had missing data for more than one variable. All variables were determined at the time of diagnosis. Biopsy characteristics are an optional variable in the PREDICT Prostate model; therefore, missing data on proportion of positive cores ([PPC] = number of cores with any cancer/number of cores taken) were tolerated. We also re-tested the value of PPC to predict PCa death in a sub-group with intact biopsy information (n = 44,163) using the same method as previously [3]. Primary treatment was defined as the radical treatment received up to 12 months after the date of diagnosis, or conservative management. The same definition of comorbidity was used as in the model development: the combination of both Charlson Comorbidity Index of 1 or greater (excluding PCa) and a hospital admission in the 2 years preceding PCa diagnosis [3]. Up to 2008, the treatment strategies of active surveillance and watchful waiting were reported as conservative management. After 2008, these strategies were registered as separate entities. We used conservative management as a treatment strategy also for men diagnosed after 2008, although a small, well-defined active surveillance group was separately analysed.
Outcome
The model estimates prostate cancer-specific mortality (PCSM), non-PCa mortality (NPCM) and overall or all-cause mortality (ACM), counted from the time of diagnosis. It provides estimates following conservative management and radical treatment (by either radical prostatectomy or radiotherapy).
Statistical analysis methods
Beta coefficients for each prognostic factor in the model were applied to derive prognostic indices for PCSM and NPCM for each patient. These were used in combination with the model’s baseline hazard functions and time at risk to create individual estimates of unadjusted PCSM and NPCM over 15 years. These estimates were adjusted for the competing risks between the two causes of death to generate ACM estimates. To assess discrimination, 15-year estimates were generated. Harrell’s concordance index (c-index) was then applied using the ‘Hmisc’ package in R [14]. Discrimination using PREDICT Prostate was compared to the EAU and NCCN stratification systems and the UCSF CAPRA score [15,16,17]. Sub-classification of stage T2 was not available; therefore, T2 was assumed to be T2a for the sake of these classifications. When PPC was unknown, it was assumed to be < 34% in the CAPRA model. Adjusted predictions of cumulative PCSM, NPCM and ACM were generated using the available follow-up for the assessment of model calibration. Calibration was assessed using a chi-square goodness of fit (GOF) across quintiles of risk using the method of May and Hosmer [18]. Calibration was also assessed within treatment sub-groups. All data analyses were performed in Stata™ 14, unless otherwise stated above.
Results
Participants
Sixty-nine thousand two hundred six men were included with 13.9 years of median follow-up. The Swedish population attributes at baseline are compared to the UK model development cohort in Table 1. Patient characteristics were similar in both cohorts, with a larger proportion of grade group 1 disease in the Swedish cohort. A larger proportion of men underwent surgery as opposed to radiotherapy in the Swedish cohort, and smaller proportion were treated with primary androgen deprivation therapy in this time period. Breakdown of the patients by risk groups is reported in Additional file 1: Table S1.
Model performance
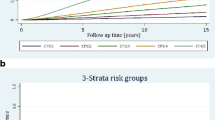
Overall discrimination of PREDICT Prostate was very good with c-indices 0.85 (95% CI 0.85–0.86) for PCSM and 0.79 (95% CI 0.79–0.79) for ACM (Table 2). Overall calibration of the model was excellent with 25,925 deaths predicted and 25,849 deaths observed in PCBaSe. This equates to an overall observed to expected (O:E) ratio of 1:1.003. Calibration across quintiles of risk is shown in Fig. 1 and Additional file 1: Table S2. Although the O:E ratio for any-cause death was very close to 1, expected numbers of PCa deaths were slightly higher than observed (O:E 0.897) and expected numbers of non-PCa deaths were lower than observed (O:E 1.060), particularly in the highest risk quintiles.
Treatment sub-groups
Overall, 20,384 men underwent conservative management and 32,842 received radical treatment. Within these groups, c-indices remained good, with c-index for 15-year PCSM 0.81 (95% CI 0.80–0.82) for conservative management and 0.78 (95% CI 0.77–0.80) for radical treatment (Table 2).
Among men on well-defined active surveillance, c-indices were further improved at 0.88 for PCSM and 0.75 for ACM (Additional file 1: Table S3). Calibration also remained good within treatment groups with differences between observed and predicted numbers of overall deaths 1.4%, 2.2% and 3.1% among men who received active surveillance, radiotherapy and prostatectomy, respectively (Table 3). The model overestimated PCSM and underestimated NPCM within the sub-group which received androgen deprivation monotherapy by as much as 8%—but remained within 2% for overall death (Table 3).
Comparison to existing models
PREDICT Prostate significantly outperformed the comparator models when predicting ACM, both overall and within every major treatment sub-group (Table 2 and Additional file 1: Table S3). Discriminatory performance was significantly better for PCSM overall (Additional file 1: Table S4). Across all treatment sub-groups, the model outperformed the 3-stratum EAU risk categories. Improvements in discrimination failed to reach significance for PCSM in some comparisons with the NCCN and CAPRA scores, but in only one incidence was the c-index better for one of these comparator models (CAPRA score for PCSM among RP patients, Additional file 1: Table S3).
Biopsy parameter sub-analysis
Biopsy parameterisation using percentage of positive cores (PPC) was re-explored within a group of 44,163 men who had this information registered (Additional file 1). Inclusion of biopsy characteristics did not significantly alter the discriminatory performance of the model (Additional file 1: Tables S4 & Table S5): either using a dichotomous 50% percentage of cores cut-off or PPC as a continuous variable. Inclusion of biopsy information did improve calibration across lower-risk quintiles of risk for PCSM. Calibration for any-cause death however was unchanged regardless of inclusion of biopsy information (Additional file 1: Table S6 & Figure S1).
Discussion
In this large external validation cohort, we demonstrated that PREDICT Prostate is a robust and generalisable long-term prognostic model. In the analysis of an independent cohort, ten times larger than the original cohort, discriminatory accuracy and calibration was good. This also remained true within treatment groups, particularly in men managed conservatively or by radical therapy.
Conveying information to an individual about their disease prognosis within their own context of competing mortality has historically been an imprecise exercise with little objective data available. The most current prognostication is based on stratification groups of the cancer itself and discussions with clinicians who may be conflicted towards a particular treatment [19,20,21]. PREDICT Prostate was conceived to address this gap in clinical need and standardise the decision-making process [3] and has shown promise to positively influence clinical decision-making [22]. It is built around long-term actual survival data and has been designed to address all AJCC criteria [6].
In the model development study, c-indices were 0.84 for PCSM and 0.77 for ACM within the UK validation cohort [3]. In the original study, external validity was also assessed within a Singaporean cohort. However, this cohort was small (n = 2546) and follow-up was quite short (5.1 years). Here we show in a cohort of > 69,000 men with longer median follow-up that our c-indices were actually improved to 0.85 for PCSM and 0.79 for ACM with excellent calibration. We did note a marginal overestimation of PCSM, which was contrary to the slight underestimation we had observed in the Singapore external validation in the original paper [3]. Given that the model is very well calibrated for ACM, this apparent overestimation of PCSM (and corresponding underestimation of NPCM) is likely to be a result of differences in cause of death classification, reporting or recording practices. ACM is the key outcome of interest, and a more unequivocal endpoint, against which this model performs very well.
When compared to existing models, PREDICT Prostate consistently outperformed the three-stratum risk classification system used in the EAU, D’Amico and NICE stratification criteria [16, 20, 23]. We recognise that comparisons against these risk stratification criteria are limited and that they are not designed to be prognostic nomograms; however, they are widely used in clinical practice to inform treatment decisions. Benefits of PREDICT were also seen against the NCCN and CAPRA scores, which add more granularity but ultimately retain a grouping system rather than individual estimates [16, 17]. For the outcome of PCSM, the CAPRA score did perform similarly well for some treatment groups, particularly in men treated with prostatectomy. This is unsurprising, as the model was originally built around prostatectomy patients [24]. It should be noted that PREDICT Prostate is not a treatment-specific tool; therefore, by assessing discrimination within treatment sub-groups, its discriminatory performance will inevitably be reduced. Nonetheless, PREDICT Prostate performed significantly better in predicting ACM and PCSM in most treatment groups. We also confirmed that adding in biopsy data to the model improved the performance though this effect was marginal in addition to the other variables already included. Using PPC as a continuous variable maximises the use of prognostic information, and this parameterisation did lead to marginally superior discrimination for ACM.
The primary utility of PREDICT Prostate will be in men for whom conservative management and radical treatment might both be appropriate options, for whom the decision is most difficult. Abundant literature demonstrates that decision aids contribute to more knowledgeable and informed patients and that they can improve clinician-patient communication [25, 26]. Therefore, the model may have wide potential applications in informing patient, clinician and multi-disciplinary team decision-making to reduce both over- and under-treatment. Formal clinical impact assessments are also crucial to show face and functional validity, and these are underway with PREDICT Prostate [27]. Future research endeavours could assess what impact the use of the model might have on actual treatment practices and compare this model with prognostic biomarkers, or radiological prognosticators. Over time, additional parameters can be incorporated into this base model, or the model itself be updated, should new variables be shown to have independent prognostic effects [28].
More recent efforts in prognostic tools have sought to utilise novel genomic or biological markers to generate prognostic estimates. However, most established genomic tools such as Prolaris CCP and Oncotype DX GPS have predominantly been tested against shorter-term outcomes or in treatment-specific cohorts [29, 30]. Where they have been assessed against PCSM, concordance has been very similar to our model—for example, the Decipher genomic classifier alongside CAPRA showed an AUC of 0.78 (95% CI 0.68–0.87) for 10-year PCSM following prostatectomy [31]. Direct comparison with PREDICT Prostate is not possible without a head-to-head or combined study, but the value of such expensive tests do need to be re-assessed in the context of optimised clinical multivariable models [32]. In this context, we would welcome collaborations or independent studies on the value of adding genomic classifiers to future iterations of PREDICT Prostate.
This study has numerous strengths, given the large sample size, long follow-up and high completeness of data in PCBaSe [33]. However, we recognise limitations inherent to using registry data. Seventeen per cent of men were excluded due to missing data, and we cannot exclude this, introducing some bias. A large proportion of men within this validation dataset had low-grade disease, such that PCa mortality rates were relatively low which may affect discriminatory performance. Men diagnosed within the inclusion period may also not be representative of contemporary practice with changes in PCa diagnosis and treatment. For instance, we recognise that primary hormone therapy is now rarely used in the context of non-metastatic PCa; hence, we included sub-group analyses within other treatment groups. We also appreciate that multi-modal therapies are increasingly used in higher risk cases, which we were not able to assess in this study due to the inclusion dates and data availability limitations of our datasets. Another particular concern is the lack of information from magnetic resonance imaging (MRI). However, the current focus for MRI is on tumour detection rather than prognostication and it is unknown if MRI lesion characteristics (Likert or PIRAD scoring) have any bearing on survival. Our model also cannot account for subsequent transitions to different treatments. However, in our UK dataset, conversions to active treatment were less than 6% across total follow-up [3]. We also recognise the lack of T-stage sub-classification, which is a key parameter in 2 of the existing models we made comparisons to. However, it is accepted that T stage is often inaccurately assigned in localised disease [34]. We also recognise that other endpoints of interest exist, particularly the development of metastases and commencement of hormone therapy. The model is untested against these endpoints, but calibrated against the more robust endpoint of death.
A key issue going forward is the validation of this model in non-Caucasian and screened populations. Although the original paper re-tested the model in Singaporean men, PREDICT Prostate remains untested in men of African descent or other ethnicities. Independent validations within screened populations, and within other prospectively collected or randomised datasets, would also be helpful and should be encouraged. Finally, we recognise that other nomograms are available, against which direct comparisons would be very insightful. These were not possible within the design of this study, or the limitations of this data, particularly with regard to comorbidity.
Conclusions
This large external validation demonstrates the robustness of PREDICT Prostate. PREDICT Prostate, available as a free-to-use web tool [4], has the potential to significantly improve shared decision-making for PCa management, particularly the choice between conservative management and radical treatment. Further, independent external validations are encouraged, especially in populations of different ethnicities.
Availability of data and materials
The datasets used in the current study can be made available at a remote server with export of aggregated data only. The application will be considered by the Prostate Cancer data Base Sweden reference group and The Research Ethics Board in Uppsala. Please contact author PS.
Abbreviations
- ACM:
-
All-cause mortality
- AJCC:
-
American Joint Committee on Cancer
- AUC:
-
Area under the curve
- CAPRA:
-
University of California San Francisco cancer of the prostate risk assessment score
- c-index:
-
Concordance index
- EAU:
-
European Association of Urology
- NCCN:
-
National Comprehensive Cancer Network
- NICE:
-
National Institute for Health and Care Excellence (UK)
- NPCM:
-
Non-prostate cancer mortality
- O:E ratio:
-
Observed to expected ratio
- PCa:
-
Prostate cancer
- PCBaSe:
-
Prostate Cancer data Base Sweden
- PCSM:
-
Prostate cancer-specific mortality
- PIRADS:
-
Prostate imaging reporting and data system
- PPC:
-
Proportion of positive cores
- PSA:
-
Prostate-specific antigen
- T stage:
-
Tumour stage
- UK:
-
United Kingdom
- 95% CI:
-
95% confidence interval
References
NPCA. National Prostate Cancer audit - annual report 2017. 2017.
Greenberg DC, Lophatananon A, Wright KA, Muir KR, Gnanapragasam VJ. Trends and outcome from radical therapy for primary non-metastatic prostate cancer in a UK population. PLoS One. 2015;10(3):e0119494.
Thurtle DR, Greenberg DC, Lee LS, Huang HH, Pharoah PD, Gnanapragasam VJ. Individual prognosis at diagnosis in nonmetastatic prostate cancer: development and external validation of the PREDICT Prostate multivariable model. PLoS Med. 2019;16(3):e1002758.
Predict Prostate Homepage. Available from: https://prostate.predict.nhs.uk/. Accessed 29 Mar 2020.
NICE. Endorsed resource - Predict Prostate. Prostate cancer: diagnosis and management. 2019.
Kattan MW, Hess KR, Amin MB, Lu Y, Moons KG, Gershenwald JE, et al. American Joint Committee on Cancer acceptance criteria for inclusion of risk models for individualized prognosis in the practice of precision medicine. CA Cancer J Clin. 2016;66(5):370–4.
Moons KG, Altman DG, Vergouwe Y, Royston P. Prognosis and prognostic research: application and impact of prognostic models in clinical practice. BMJ. 2009;338:b606.
Justice AC, Covinsky KE, Berlin JA. Assessing the generalizability of prognostic information. Ann Intern Med. 1999;130(6):515–24.
Altman DG, Vergouwe Y, Royston P, Moons KG. Prognosis and prognostic research: validating a prognostic model. BMJ. 2009;338:b605.
DeLong ER, DeLong DM, Clarke-Pearson DL. Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics. 1988;44(3):837–45.
Van Hemelrijck M, Wigertz A, Sandin F, Garmo H, Hellström K, Fransson P, et al. Cohort profile: the National Prostate Cancer Register of Sweden and Prostate Cancer data Base Sweden 2.0. Int J Epidemiol. 2013;42(4):956–67.
Tomic K, Berglund A, Robinson D, Hjälm-Eriksson M, Carlsson S, Lambe M, et al. Capture rate and representativity of the National Prostate Cancer Register of Sweden. Acta Oncol. 2015;54(2):158–63.
Fall K, Strömberg F, Rosell J, Andrèn O, Varenhorst E, Group S-ERPC. Reliability of death certificates in prostate cancer patients. Scand J Urol Nephrol. 2008;42(4):352–7.
Harrell F. Package ‘Hmisc’. In: Dupont C, editor. CRAN2018. p. 235–6.
Heidenreich A, Bastian PJ, Bellmunt J, Bolla M, Joniau S, van der Kwast T, et al. EAU guidelines on prostate cancer. Part 1: screening, diagnosis, and local treatment with curative intent-update 2013. Eur Urol. 2014;65(1):124–37.
NCCN. National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology: Prostate cancer. Version 2. 2018. Available from: https://www.nccn.org/professionals/physician_gls/pdf/prostate.pdf. Accessed 13 Mar 2020.
Cooperberg MR, Pasta DJ, Elkin EP, Litwin MS, Latini DM, Du Chane J, et al. The University of California, San Francisco Cancer of the Prostate Risk Assessment score: a straightforward and reliable preoperative predictor of disease recurrence after radical prostatectomy. J Urol. 2005;173(6):1938–42.
May S, Hosmer DW. A simplified method of calculating an overall goodness-of-fit test for the Cox proportional hazards model. Lifetime Data Anal. 1998;4(2):109–20.
AUA/ASTRO/SUO. Clinically localized prostate cancer: AUA/ASTRO/SUO guideline 2017.
NICE. National Institute for Health and Care Excellence NICE Guidelines [CG175] Prostate cancer: diagnosis and treatment. 2014.
Kim SP, Gross CP, Nguyen PL, Nguyen PY, Smaldone MC, Thompson RH, et al. Specialty bias in treatment recommendations and quality of life among radiation oncologists and urologists for localized prostate cancer. Prostate Cancer Prostatic Dis. 2014;17(2):163–9.
Thurtle DR, Jenkins V, Pharoah PD, Gnanapragasam VJ. Understanding of prognosis in non-metastatic prostate cancer: a randomised comparative study of clinician estimates measured against the PREDICT prostate prognostic model. Br J Cancer. 2019;121(8):715–8.
D'Amico AV, Whittington R, Malkowicz SB, Schultz D, Blank K, Broderick GA, et al. Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA. 1998;280(11):969–74.
Cooperberg MR, Broering JM, Carroll PR. The UCSF cancer of the prostate risk assessment (CAPRA) score accurately predicts metastasis, prostate cancer mortality, and all-cause mortality regardless of treatment type. J Urol. 2008;179(4):114–5.
O'Connor AM, Rostom A, Fiset V, Tetroe J, Entwistle V, Llewellyn-Thomas H, et al. Decision aids for patients facing health treatment or screening decisions: systematic review. BMJ. 1999;319(7212):731–4.
Lin GA, Aaronson DS, Knight SJ, Carroll PR, Dudley RA. Patient decision aids for prostate cancer treatment: a systematic review of the literature. CA Cancer J Clin. 2009;59(6):379–90.
Registry I. ISRCTN 28468474 PREDICT Prostate Patient Study Evaluation of a new tool, PREDICT Prostate, to aid treatment decision-making for men with newly diagnosed non-metastatic prostate cancer; 2018.
Wishart GC, Bajdik CD, Dicks E, Provenzano E, Schmidt MK, Sherman M, et al. PREDICT Plus: development and validation of a prognostic model for early breast cancer that includes HER2. Br J Cancer. 2012;107(5):800–7.
Ontario HQ. Prolaris cell cycle progression test for localized prostate cancer: a health technology assessment. Ont Health Technol Assess Ser. 2017;17(6):1–75.
Cucchiara V, Cooperberg MR, Dall'Era M, Lin DW, Montorsi F, Schalken JA, et al. Genomic markers in prostate cancer decision making. Eur Urol. 2018;73(4):572–82.
Cooperberg MR, Davicioni E, Crisan A, Jenkins RB, Ghadessi M, Karnes RJ. Combined value of validated clinical and genomic risk stratification tools for predicting prostate cancer mortality in a high-risk prostatectomy cohort. Eur Urol. 2015;67(2):326–33.
Herlemann A, Washington SL, Eapen RS, Cooperberg MR. Whom to treat: postdiagnostic risk assessment with Gleason score, risk models, and genomic classifier. Urol Clin North Am. 2017;44(4):547–55.
Tomic K, Westerberg M, Robinson D, Garmo H, Stattin P. Proportion and characteristics of men with unknown risk category in the National Prostate Cancer Register of Sweden. Acta Oncol. 2016;55(12):1461–6.
Reese AC, Sadetsky N, Carroll PR, Cooperberg MR. Inaccuracies in assignment of clinical stage for localized prostate cancer. Cancer. 2011;117(2):283–9.
Acknowledgements
Collection of data in the National Prostate Cancer Register of Sweden was made possible by the continuous work of the NPCR steering group: Pär Stattin (chairman), Ingela Franck Lissbrant (deputy chair,) Camilla Thellenberg, Magnus Törnblom, Stefan Carlsson, Marie Hjälm-Eriksson, David Robinson, Mats Andén, Jonas Hugosson,, Maria Nyberg, Ola Bratt, Olof Akre, Per Fransson, Eva Johansson, Johan Stranne, Fredrik Sandin and Karin Hellström.
Funding
This work was supported by a Research Scholarship from The Urology Foundation and Infrastructure support from Cancer Research UK Cambridge Institute and the Cambridge Biomedical Campus. The work within the PCBaSe was funded by the Swedish Research Council and the Swedish Cancer Society.
Author information
Authors and Affiliations
Contributions
DT, PP and VJG conceived, designed and analysed the study collaboratively. OB and PS reviewed the project design and oversaw the collection, curation and release of data. All authors were involved in the critical review of the results and have contributed to, read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The release of these data was approved by the Prostate Cancer data Base Sweden committee after peer review.
Consent for publication
N/A.
Competing interests
The authors have read the BMC Medicine editorial policy on competing interests and declare they have no relevant conflicts of interest to declare.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Additional file 1.
Supplementary files.
Additional file 2.
Data request and study outline form to PCBaSe.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Thurtle, D., Bratt, O., Stattin, P. et al. Comparative performance and external validation of the multivariable PREDICT Prostate tool for non-metastatic prostate cancer: a study in 69,206 men from Prostate Cancer data Base Sweden (PCBaSe). BMC Med 18, 139 (2020). https://doi.org/10.1186/s12916-020-01606-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12916-020-01606-w