Abstract
Background
Growth hormone (GH) is a heterogeneous protein composed of several molecular isoforms, the most abundant ones being the 22 kDa- and 20 kDa-GH. Exercise-induced secretion of GH isoforms has been extensively investigated in normal-weight individuals due to antidoping purposes, particularly recombinant human GH (rhGH) abuse. On the other hand, the evaluation of exercise-induced responses in GH isoforms has never been performed in obese subjects.
Methods
The acute effects of whole body vibration (WBV) or maximal voluntary contraction (MVC) alone and the combination of MVC with WBV (MVC + WBV) on circulating levels of 22 kDa- and 20 kDa-GH were evaluated in 8 obese male adolescents [mean age ± SD: 17.1 ± 3.3 yrs.; weight: 107.4 ± 17.8 kg; body mass index (BMI): 36.5 ± 6.6 kg/m2; BMI standard deviation score (SDS): 3.1 ± 0.6].
Results
MVC (alone or combined with WBV) significantly stimulated 22 kDa- and 20 kDa-GH secretion, while WBV alone was ineffective. In particular, 22 kDa- and 20 kDa-GH peaks were significantly higher after MVC + WBV and MVC than WBV. In addition, 22 kDa-GH (but not 20 kDa-GH) peak was significantly higher after MVC + WBV than MVC. Importantly, the ratio of circulating levels of 22 kDa- to 20 kDa-GH was constant throughout the time window of evaluation after exercise and similar among the three different protocols of exercise.
Conclusions
The results of the present study confirm the ability of MVC, alone and in combination with WBV, to stimulate both 22 kDa- and 20 kDa-GH secretion in obese patients, these responses being related to the exercise workload. Since the ratio of 22 kDa- to 20 kDa-GH is constant after exercise and independent from the protocols of exercise as in normal-weight subjects, hyposomatotropism in obesity does not seem to depend on an unbalance of circulating GH isoforms. Since the present study was carried out in a small cohort of obese sedentary adolescents, these preliminary results should be confirmed in further future studies enrolling overweight/obese subjects with a wider age range.
Similar content being viewed by others
Background
Pituitary and circulating growth hormone (GH) is a heterogeneous mix of molecules deriving from different gene expression, alternative splicing, post-translational modifications, homo- or hetero-dimerization and oligomerization, peripheral metabolism and clearance. The predominant isoform is the 1- to 191-amino acid 22 kDa-GH, while alternative splicing of the exon 3 removes the residues 32–46 from the primary structure, yielding the 176-amino acid 20 kDa-GH, which represents about 15–20% of total circulating GH [1, 2].
In the last decade. Analytical methods have been implemented to measure circulating levels of GH isoforms with adequate accuracy, specificity and sensitivity [3].
Since the biochemical heterogeneity of GH implies a different binding of the ligand to the receptor, dimerization of GH receptor, receptor activation and signal transduction [2], measurement of GH isoforms is fundamental to understand the complexity of the physiological effects of GH or, more appropriately, “somatotropic hormones”.
Exercise-induced GH secretion has been extensively investigated in the last decades, several factors being identified to affect this response, including duration, intensity and protocol of exercise, training, nutritional status, body composition, age and gender of individuals undergoing exercise and concomitant pathophysiological conditions, such as GH deficiency (GHD) [4].
In this context, morbid obesity is characterized by hyposomatotropism, which is associated with blunted GH responses to a variety of pharmacological and physiological stimuli, including exercise [5, 6].
The interest of measuring circulating levels of GH isoforms after exercise has derived from the need to validate a promising antidoping test, based on the altered ratio of circulating GH isoforms in athletes administered with recombinant human GH (rhGH), which corresponds uniquely to 22 kDa-GH [7,8,9].
Some studies, carried out in normal-weight subjects, have revealed a post-exercise increase in circulating levels of GH isoforms, mainly 22 kDa- and 20 kDa-GH, being those of non-22 kDa-GH isoforms more sustained in the recovery phase, presumably for their long half-life [10,11,12,13,14]. Since non-22 kDa-GH isoforms are more hyperglycemic than 22 kDa-GH [15], the subject should be protected by post-exercise hypoglycaemia.
Surprisingly, to the best of our knowledge, no one has ever evaluated exercise-induced release of GH isoforms in obese subjects. Therefore, aim of the present study was to measure circulating levels of 22 kDa-GH and 20 kDa-GH in a cohort of obese subjects undergoing different protocols of exercise at increasing intensity (i.e. whole body vibration (WBV) and maximal voluntary contractions (MVC)]. For our experience, these types of exercise have demonstrated to be capable to stimulate “total” GH secretion in either normal-weight or obese subjects [16,17,18,19,20].
Methods
Since the present study represents the development of a previous research performed in healthy normal-weight and obese subjects by our group, the methodology widely replicates that adopted in the previous papers [16, 18].
Subjects
Eight obese male adolescents [mean age ± SD: 17.1 ± 3.3 years; weight: 107.4 ± 17.8 kg; body mass index (BMI): 36.5 ± 6.6 kg/m2; BMI standard deviation score (SDS): 3.1 ± 0.6; waist circumference: 118.3 ± 18.0 cm; hip circumference: 123.9 ± 15.3 cm] were recruited among patients hospitalized for a multidisciplinary integrated body weight reduction program at the Division of Auxology, Istituto Auxologico Italiano, Piancavallo (VB), Italy. In order to avoid any carry-over effect of the weight loss or modifications in diet and physical activity on GH secretion, the entire study was completed before starting the multidisciplinary integrated body weight reduction program.
All the adolescent patients voluntarily participated in this investigation after obtaining a written informed consent given by their parents. The main criteria of exclusion were any overt disease apart from morbid obesity. Diabetes mellitus, including glucose intolerance, was excluded by a 75 g oral glucose load, while Prader-Willi syndrome was excluded by clinical history, physical examination and FISH detection of chromosome 15 deletions. Due to the effects of female sex steroids on GH secretion and the difficulty to perform three exercise protocols (see below) in the same phase of the menstrual cycle, only boys were included in the study. Thyroidal (TSH and free T4 levels) and gonadal functions (LH, FSH, testosterone levels) were normal in all subjects (data not shown). All of them were habitually sedentary and were not involved in any strength or endurance training in the previous 2 weeks preceding the admission to the study protocol. No subjects had any signs of musculoskeletal disorders potentially hampering the execution of the tests.
No drugs or nutritional supplements known to interfere with GH and/or cortisol secretion were taken by the subjects in the month preceding the study.
Testing
After an overnight fast, subjects were admitted to the laboratory 1 h before the beginning of the tests.
All the participants performed one of the following exercise protocols per diem: WBV alone, MVC alone, and MVC alternated with WBV (MVC + WBV).
The three different exercise protocols were randomly carried out in separate days with an interval of at least two days in between, accordingly with a scheme of randomization generated by the web-site https://www.randomizer.org.
After a 5-min standardized warm-up on a cycloergometer (power: 50 W, cadence: 60 rpm), the three different protocols were performed as reported in Fig. 1.
During the WBV protocol, subjects initially seated in a semi-recumbent position on a horizontal leg press machine (Technogym, Gambettola, Italy) for 30 s, with the trunk-thigh and thigh-shank angles at 80°, without producing any movement. This first step was then followed by a 30-s WBV bout administered while the subject stood on a vibrating platform (Nevisys H1©, RME, Ferrara, Italy), as previously described [16]. Vertical sinusoidal vibrations were generated at a frequency of 35 Hz, the acceleration of the platform being 2.85 g at a peak-to-peak amplitude of vibration of 5 mm. These two rest-WBV cycles were repeated 15 times, resulting in a total duration of 15 min.
During the MVC protocol, subjects were initially placed on the leg press machine in the same supine position as described in the WBV protocol. The subjects performed three 5-s MVC, separated by 5-s resting periods in between. This first step was then followed by a 30 s of rest in the same static position as in the WBV protocol, but without WBV. These two MVC-rest cycles were repeated 15 times, resulting in a total duration of 15 min.
During the MVC + WBV protocol, subjects initially performed the three 5-s MVC (separated by 5-s resting periods in between) as in the MVC protocol. This first step was then followed by a 30-s WBV bout as in the WBV protocol. These two MVC + WBV cycles were repeated 15 times, resulting in a total duration of 15 min.
The characteristics of the different protocols, including the temporal patterns, are identical to those adopted in a previous work performed by our group [18] in healthy normal-weight subjects.
Blood sampling and measurements of GH isoforms
Blood samples (5 ml at each time point) for measurement of 22 kDa- and 20 kDa-GH levels were drawn before the start of the experiment (baseline), immediately at the end of the exercise (T0) and after 15 (T15), 30 (T30), 45 (T45) and 75 min (T75). All blood samples were drawn through an indwelling cannula inserted into an ante-cubital vein kept patent via a continuous infusion of isotonic saline.
All blood samples were allowed to clot, centrifuged for 5 min and immediately stored at − 20 °C for the next analysis.
Serum levels of 22 kDa-GH were measured using the automated IDS iSYS hGH chemiluminescence assay system. In this assay, the detection monoclonal antibody (mAb) targets an epitope in the loop connecting helix 1 and 2 of GH, which is missing in 20 kDa-GH, thereby conferring specificity of the assay for the 22kD-GH molecules. Further details concerning the methodology had been published in a previous work [21]. In our hands, functional sensitivity of the assay was 0.04 μg/L and intra- and inter-assay coefficients of variation (CVs) both were below 5%.
Serum levels of 20 kDa-GH were measured using an in-house time resolved fluorescence assay as described before [22]. The assay employs two mAbs with no cross-reactivity to 22-kDa GH; intra- and inter-assay CVs were 5.4 and 6.3% at 0.2 μg/L and the functional sensitivity was 0.025 μg/L.
All samples were run in the same assay to minimize inter-assay variability (for both GH isoforms). Some samples fell below the limits of detection of 20 kDa-GH assay and were excluded from statistical analysis.
Statistical analyses
The Sigma Stat 3.5 statistical software package was used for data analyses. GraphPad Prisma 5.0 software was used for plotting data.
In order to determine a priori the sample size, a power analysis was performed by considering a difference in the mean 22 kDa-GH levels at T0 between MVC + WVB and WBV equal to 2.5 ± 2.5 μg/L with an α error of 0.05 at two tails and a power of 0.80. The result of this calculation was n = 8 patients.
The Shapiro-Wilk test showed that all parameters were normally distributed.
Results are reported as mean ± SD (standard deviation). The responses in 22 kDa- and 20 kDa-GH were evaluated as absolute values for each experimental group (MVC + WBV, MVC and WBV). Peak values of GH isoforms represent the maximal levels reached during each experimental session.
Circulating levels of 22 kDa- and 20 kDa-GH and the related ratio (i.e., 22 kDa-GH/20 kDa-GH) were compared within each experimental group (vs. the corresponding basal value for MVC + WBV, MVC or WBV) over sampling times (intra-group analysis) and among the three experimental groups (MVC + WBV vs. MVC or WBV and MVC vs. WBV) for any sampling time (inter-group analysis) by using two-way ANOVA with repeated measures (with the two factors time and group and the interaction time × group), followed by the post hoc Bonferroni’s test. One-way ANOVA, followed by the post hoc Bonferroni’s test, was used to compare 22 kDa- and 20 kDa-GH peaks and the related ratio (i.e., 22 kDa-GH-peak/20 kDa-GH-peak) among MVC + WBV, MVC and WBV groups.
A level of significance of p < 0.05 was used for all data analyses.
Results
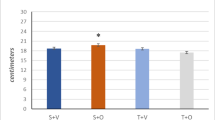
Mean basal circulating levels of 22 kDa- and 20 kDa-GH were 1.03 ± 1.52 μg/L and 0.269 ± 0.294 μg/L before MVC + WBV, 0.97 ± 1.15 μg/L and 0.234 ± 0.209 μg/L before MVC and 0.91 ± 1.05 μg/L and 0.212 ± 0.195 μg/L before WBV, respectively, without any significant difference (Fig. 2). The ratio of circulating levels of 22 kDa- to 20 kDa-GH at 0 min was similar among the three protocols of exercise (basal values: 7.382 ± 4.587 for MCV + WBV, 8.02 ± 4.73 for MCV and 8.31 ± 4.34, respectively) (Fig. 3).
Circulating levels of 22 kDa- (top panel) and 20 kDa-GH (bottom panel) after the three different protocols of exercise: maximal voluntary contractions + whole body vibration (MVC + WBV), muscle voluntary contractions (MVC) and whole body vibration (WBV). Evaluation was performed at resting after the completion of the exercise. Values are expressed as mean ± SD. *p < 0.05 vs. the corresponding basal value (T0); op < 0.05 vs. the corresponding time point of WBV; ●p < 0.05 vs. the corresponding time point of MVC
Ratio of circulating levels of 22 kDa-GH to 20 kDa-GH after the three different protocols of exercise: maximal voluntary contractions + whole body vibration (MVC + WBV), muscle voluntary contractions (MVC) and whole body vibration (WBV). Evaluation was performed at resting after the completion of the exercise. Values are expressed as mean ± SD
MVC + WBV and MCV evoked a significant increase in 22 kDa-GH levels in obese patients at 0 and 15 min and only at 0 min vs. basal level, respectively (p < 0.05). WBV was ineffective to stimulate any 22 kDa-GH secretion. The 22 kDa-GH response was significantly higher after MVC + WBV than MVC at 0 min or WBV at 0 and 15 min and after MVC than WBV at 0 min (p < 0.05) (Fig. 2).
Similarly, MVC + WBV and MCV evoked a significant increase in 20 kDa-GH levels at 0 min vs. basal level (p < 0.05), without any significant changes after WBV. This response was significantly higher after MVC + WBV than MVC at 0 min or WBV at 0 and 15 min and after MVC than WBV at 0 min (p < 0.05) (Fig. 2).
GH peaks after MVC + WBV (22 kDa-GH: 9.72 ± 5.55 μg/L and 20 kDa-GH: 0.98 ± 0.55 μg/L) and after MVC (22 kDa-GH: 6.45 ± 6.35 μg/L and 20 kDa-GH: 0.72 ± 0.68 μg/L) were significantly higher than those observed after WBV (22 kDa-GH: 2.27 ± 2.26 μg/L and 20 kDa-GH: 0.24 ± 0.25 μg/L) (p < 0.05), being only 22 kDa-GH peak after MVC + WBV higher than that after MCV (p < 0.05) (Fig. 4).
Peak values of 22 kDa- and 20 kDa-GH and the related ratio (i.e., 22 kDa-GH-peak/20 kDa-GH-peak) after the three different protocols of exercise: maximal voluntary contractions + whole body vibration (MVC + WBV), muscle voluntary contractions (MVC) and whole body vibration (WBV). Values are expressed as mean ± SD. *p < 0.05 vs. WBV; op < 0.05 vs. MVC
Importantly, the ratio of circulating levels of 22 kDa- to 20 kDa-GH in obese patients remained constant throughout the time window of evaluation and paralleled among the three protocols of exercises (Fig. 3). No significant differences were found in the ratio of peak levels of 22 kDa- to 20 kDa-GH among the three protocols of exercise (Fig. 4).
Discussion
The most important findings of the present study carried out in a cohort of obese adolescents, generally more responsive to pharmacological or physiological GH stimuli than obese adults, were: (1) circulating levels of 22 kDa- and 20 kDa-GH are increased after acute exercise; (2) 22 kDa-GH is quantitatively the main isoform secreted in circulation after exercise; (3) combination of WBV with MVC evokes the highest responses in both GH isoforms; (4) the ratio of circulating levels of 22 kDa- to 20 kDa-GH is constant throughout the time window of evaluation after exercise and is similar among three different protocols of exercise (MVC + WBV, MVC and WBV).
Reportedly, the pathophysiology of hyposomatropism in obese subjects is complex, being presumably multifactorial. In particular, central (such as reduced GHRH function and/or increased somatostatin tone at the hypothalamic level) and peripheral (such as increased circulating levels of free fat acids, FFA) mechanisms have been invoked to explain the blunted GH responses to a variety of pharmacological and physiological stimuli in this special population [23,24,25,26]. Some authors have (erroneously) hypothesized the existence of altered proportions of GH isoforms in syndromic obesity (e.g., Prader-Willi syndrome) [27]. Now, we can exclude this possibility also in morbid obesity, being this the first study evaluating exercise-induced secretion of GH isoforms in an obese population and demonstrating a ratio of circulating levels of 22 kDa- to 20 kDa-GH similar to that of normal-weight individuals (i.e., about 10–15) [28]. Therefore, hyposomatotropism in morbid obesity does not derive from an alteration in the molecular mechanisms underlying the generation of GH isoforms, but seems to be a consequence of an insufficient stimulation and/or excessive inhibition of GH function (at hypothalamic and/or pituitary level). Obese state causes a “quantitative” rather than a “qualitative” alteration of GH secretion.
As shown in the present study, different protocols of exercise not surprisingly evoked responses in GH isoforms that were higher when the workload was heavier, such as that of MVC + WBV. This finding means not only the preservation of a “adequate” GH responsiveness to increasing physiological stimuli in obese subjects, but also the maintenance of a constant ratio of circulating levels of 22 kDa- to 20 kDa-GH independently from intensity, duration and protocol of exercise in both normal-weight and obese individuals.
In this context, the reliability of the antidoping test, used to detect rhGH abuse and based on an unbalance of circulating GH isoforms (particularly, the ratio of 22 kDa- tp pituitary GH, i.e. rGH/pitGH) after rhGH administration, would be ensured also in athletes, who, in some sport disciplines, are frequently overweight or obese [7, 9, 29]. In fact, as specified above the hyposomatotropism, acute exercise and execution of different protocols of exercise do not change the proportions of GH isoforms, which are similar to those present in normal-weight individuals 10,11,28]. Nevertheless, before extrapolating these data in a normative setting (e.g., WADA policy) [30], further studies in a larger population of overweight/obese athletes are mandatory, being the latter population different from ours, i.e., a small cohort of obese sedentary subjects.
A limitation of the present study could be the measurement of circulating levels of few GH isoforms, i.e. uniquely 22 kDa- and 20 kDa-GH, which have similar disappearance rates in circulation [10]. Since other GH isoforms, such as non-22 kDa-GH, persist in the recovery phase after acute exercise administered to normal-weight subjects [10], future studies should investigate the post-exercise kinetics of the remaining GH isoforms also in obese individuals. This issue is of great interest, being some GH isoforms more hyperglycemic than others [15] and exercise a well-recognized strategy to improve metabolic control [31]. In this respect, we cannot exclude that obese subjects have slower mechanisms of clearance for some GH isoforms, other than 22 kDa- and 20 kDa-GH, particularly those of higher molecular weight (e.g., dimers or oligomers).
The importance of measuring “all” GH isoforms is evident when comparing exercise-induced GH responses in studies in which different analytical methods were used. In particular, Rigamonti et al. [16] showed that WBV evoked a significant increase in (total?) GH levels, measured by a the commercially available immunometric chemiluminescence assay Immulite 2000 (DPC, Los Angeles, USA), while, in the present study, no significant changes in either 22 kDa- or 20 kDa-GH were found after WBV (alone). This might imply that some pharmacological and physiological stimuli selectively release some GH isoforms and not others or that, more likely, due to analytical reasons (e.g., sensitivity and specificity of the assays), GH stimulation may not be detectable and, erroneously, considered absent. So, before drawing any conclusion regarding the ineffectiveness of a GH stimulus, one should know what GH isoforms are measured. Since, as shown by recent biochemical and pharmacological studies [1, 2], activation of GH receptor depends on the specificity of GH isoform(s), measurement of (even very low) levels of any GH isoform is fundamental to understand signal transduction at molecular level and clinical effects in the endocrinological practice.
Finally, it is also worth mentioning that, in the present study, there was an increasing variability of circulating levels of GH isoforms during the recovery phase (i.e. starting from T30). This finding, already evidenced by Wallace et al. [10], is likely to be related to generation of GH fragments and/or dimers/oligomers in circulation, which could impair the specificity of our assays. However, despite this (potential) analytical interference, the ratio of circulating levels of 22 kDa- to 20 kDa is robustly stable.
Before closing, two other limitations should be recalled, i.e., the reduced number of subjects recruited and inclusion of only males. Therefore, the conclusions of the present study can be considered only preliminary; furthermore, we do propose a more extensive use of advanced analytical methods to measure GH isoforms in obesity and other pathophysiological conditions for better understanding the complexity of GH-IGF-I axis.
Conclusions
The results of the present study confirm the ability of MVC, alone and in combination with WBV, to stimulate both 22 kDa- and 20 kDa-GH secretion in obese patients, being these responses related to the exercise workload. Since the ratio of 22 kDa- to 20 kDa-GH is constant after exercise and independent from the protocols of exercise as in normal-weight subjects, hyposomatotropism in obesity does not seem to depend on an unbalance of circulating GH isoforms and exercise-induced GH response is unlikely to affect the reliability of direct methods for detection of rhGH abuse in overweight/obese athletes. Since the present study was carried out in a small cohort of obese sedentary adolescents, these preliminary results should be confirmed by further future studies enrolling overweight/obese subjects with a wider age range.
Abbreviations
- BMI:
-
Body mass index
- FFA:
-
Free fat acids
- GH:
-
Growth hormone
- GHD:
-
GH deficiency
- GHRH:
-
GH releasing hormone
- IGF-I:
-
Insulin-like growth factor I
- MVC:
-
Maximal voluntary contractions
- rhGH:
-
Recombinant human GH
- SDS:
-
Standard deviation score
- WBV:
-
Whole body vibration
References
Rigamonti AE, Bozzola M, Banfi G, Meazza C, Müller EE, Cella SG. Growth hormone variants: a potential avenue for a better diagnostic characterization of growth hormone deficiency in children. J Endocrinol Investig. 2012;35:937–44.
Baumann GP. Growth hormone doping in sports: a critical review of use and detection strategies. Endocr Rev. 2012;33:155–86.
Bidlingmaier M, Freda PU. Measurement of human growth hormone by immunoassays: current status, unsolved problems and clinical consequences. Growth Hormon IGF Res. 2010;20:19–25.
Godfrey RJ, Madgwick Z, Whyte GP. The exercise-induced growth hormone response in athletes. Sports Med. 2003;33:599–613.
Franco C, Bengtsson BA, Johannsson G. The GH/IGF-1 axis in obesity: physiological and pathological aspects. Metab Syndr Relat Disord. 2006;4:51–6.
Thomas GA, Kraemer WJ, Comstock BA, Dunn-Lewis C, Maresh CM, Volek JS. Obesity, growth hormone and exercise. Sports Med. 2013;43:839–49.
Rigamonti AE, Cella SG, Marazzi N, Di Luigi L, Sartorio A, Müller EE. Growth hormone abuse: methods of detection. Trends Endocrinol Metab. 2005;16:160–6.
Rigamonti AE, Locatelli L, Cella SG, Bonomo SM, Giunta M, Molinari F, Sartorio A, Müller EE. Muscle expressions of MGF, IGF-IEa, and myostatin in intact and hypophysectomized rats: effects of rhGH and testosterone alone or combined. Horm Metab Res. 2009;41:23–9.
Bidlingmaier M. New detection methods of growth hormone and growth factors. Endocr Dev. 2012;23:52–9.
Wallace JD, Cuneo RC, Bidlingmaier M, Lundberg PA, Carlsson L, Boguszewski CL, Hay J, Boroujerdi M, Cittadini A, Dall R, Rosén T, Strasburger CJ. Changes in non-22-kilodalton (kDa) isoforms of growth hormone (GH) after administration of 22-kDa recombinant human GH in trained adult males. J Clin Endocrinol Metab. 2001;86:1731–7.
Wallace JD, Cuneo RC, Bidlingmaier M, Lundberg PA, Carlsson L, Boguszewski CL, Hay J, Healy ML, Napoli R, Dall R, Rosén T, Strasburger CJ. The response of molecular isoforms of growth hormone to acute exercise in trained adult males. J Clin Endocrinol Metab. 2001;86:200–6.
Pagani S, Cappa M, Meazza C, Ubertini G, Travaglino P, Bozzola E, Bozzola M. Growth hormone isoforms release in response to physiological and pharmacological stimuli. J Endocrinol Investig. 2008;31:520–4.
Tuckow AP, Rarick KR, Kraemer WJ, Marx JO, Hymer WC, Nindl BC. Nocturnal growth hormone secretory dynamics are altered after resistance exercise: deconvolution analysis of 12-hour immunofunctional and immunoreactive isoforms. Am J Physiol Regul Integr Comp Physiol. 2006;291:R1749–55.
Nindl BC, Kraemer WJ, Marx JO, Tuckow AP, Hymer WC. Growth hormone molecular heterogeneity and exercise. Exerc Sport Sci Rev. 2003;31:161–6.
Vickers MH, Gilmour S, Gertler A, Breier BH, Tunny K, Waters MJ, Gluckman PD. 20-kDa placental hGH-V has diminished diabetogenic and lactogenic activities compared with 22-kDa hGH-N while retaining antilipogenic activity. Am J Physiol Endocrinol Metab. 2009;297:E629–37.
Rigamonti AE, De Col A, Tamini S, Tringali G, De Micheli R, Abbruzzese L, Goncalves da Cruz CR, Bernardo-Filho M, Cella SG, Sartorio A. GH responses to whole body vibration alone or in combination with maximal voluntary contractions in obese male adolescents. Growth Horm IGF Res. 2018;42–43:22–7.
Sartorio A, Lafortuna CL, Maffiuletti NA, Agosti F, Marazzi N, Rastelli F, Rigamonti AE, Muller EE. GH responses to two consecutive bouts of whole body vibration, maximal voluntary contractions or vibration alternated with maximal voluntary contractions administered at 2-h intervals in healthy adults. Growth Hormon IGF Res. 2010;20:416–21.
Sartorio A, Agosti F, De Col A, Marazzi N, Rastelli F, Chiavaroli S, Lafortuna CL, Cella SG, Rigamonti AE. Growth hormone and lactate responses induced by maximal isometric voluntary contractions and whole-body vibrations in healthy subjects. J Endocrinol Investig. 2011;34:216–21.
Giunta M, Cardinale M, Agosti F, Patrizi A, Compri E, Rigamonti AE, Sartorio A. Growth hormone-releasing effects of whole body vibration alone or combined with squatting plus external load in severely obese female subjects. Obes Facts. 2012;5:567–74.
Giunta M, Rigamonti AE, Agosti F, Patrizi A, Compri E, Cardinale M, Sartorio A. Combination of external load and whole body vibration potentiates the GH-releasing effect of squatting in healthy females. Horm Metab Res. 2013;45:611–6.
Manolopoulou J, Alami Y, Petersenn S, Schopohl J, Wu Z, Strasburger CJ, Bidlingmaier M. Automated 22-kD growth hormone-specific assay without interference from Pegvisomant. Clin Chem. 2012;58:1446–56.
Keller A, Wu Z, Kratzsch J, Keller E, Blum WF, Kniess A, Preiss R, Teichert J, Strasburger CJ, Bidlingmaier M. Pharmacokinetics and pharmacodynamics of GH: dependence on route and dosage of administration. Eur J Endocrinol. 2007;156:647–53.
Dieguez C, Carro E, Seoane LM, Garcia M, Camina JP, Senaris R, Popovic V, Casanueva FF. Regulation of somatotroph cell function by the adipose tissue. Int J Obes Relat Metab Disord. 2000;24(Suppl 2):S100–3.
Pombo M, Maccario M, Seoane LM, Tovar S, Micic D, Ghigo E, Casanueva FF, Dieguez C. Control and function of the GH-IGF-I axis in obesity. Eat Weight Disord. 2001;6(Suppl 3):22–7.
Rigamonti AE, Resnik M, Compri E, Agosti F, De Col A, Monteleone P, Marazzi N, Bonomo SM, Müller EE, Sartorio A. The cholestyramine-induced decrease of PYY postprandial response is negatively correlated with fat mass in obese women. Horm Metab Res. 2011;43:569–73.
Rigamonti AE, Piscitelli F, Aveta T, Agosti F, De Col A, Bini S, Cella SG, Di Marzo V, Sartorio A. Anticipatory and consummatory effects of (hedonic) chocolate intake are associated with increased circulating levels of the orexigenic peptide ghrelin and endocannabinoids in obese adults. Food Nutr Res. 2015;59:29678.
Rigamonti AE, Grugni G, Marazzi N, Bini S, Bidlingmaier M, Sartorio A. Unaltered ratio of circulating levels of growth hormone/GH isoforms in adults with Prader-Willi syndrome after GHRH plus arginine administration. Growth Hormon IGF Res. 2015;25:168–73.
Tong J, D'Alessio D, Ramisch J, Davis HW, Stambrook E, Tschöp MH, Bidlingmaier M. Ghrelin stimulation of growth hormone isoforms: parallel secretion of total and 20-kDa growth hormone and relation to insulin sensitivity in healthy humans. J Clin Endocrinol Metab. 2012;97:3366–74.
Di Luigi L, Rigamonti AE, Agosti F, Mencarelli M, Sgrò P, Marazzi N, Cella SG, Müller EE, Sartorio A. Combined evaluation of resting IGF1, N-terminal propeptide of type III procollagen and C-terminal cross-linked telopeptide of type I collagen levels might be useful for detecting inappropriate GH administration in female athletes. Eur J Endocrinol. 2009;160:753–8.
Holt RI. Detecting growth hormone misuse in athletes. Indian J Endocrinol Metab. 2013;17(Suppl 1):S18–22.
Smith JK. Exercise, obesity and CNS control of metabolic homeostasis: a review. Front Physiol. 2018;9:574.
Acknowledgements
The authors thank the nursing staff at the Division of Auxology, Istituto Auxologico Italiano, Piancavallo, VB, Italy. Our special thanks go to the subjects and their families for their willingness to participate in this research.
Funding
The study was supported by Progetti di Ricerca Corrente, Istituto Auxologico Italiano, IRCCS, Milan, Italy.
Availability of data and materials
The datasets used and/or analysed in the present study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Contributions
AER, MBF, CRGdC and AS designed the study. CRGdC, ADC, GT, RDM, LA and ST enrolled the subjects and performed the tests. MB and MH performed the biochemical determinations. MH and ST elaborated the database. AER analyzed the data and, together with AS, wrote the manuscript. MB, SGC and MBF contributed to data interpretation and discussion writing. All authors contributed to the manuscript revision. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The present study was approved by the Ethical Committee of Istituto Auxologico Italiano. Written informed consent was obtained from all patients.
Consent for publication
Not applicable.
Competing interests
The authors declare that there is no conflict of interest that could be perceived as prejudicing the impartiality of the research reported.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Rigamonti, A.E., Haenelt, M., Bidlingmaier, M. et al. Obese adolescents exhibit a constant ratio of GH isoforms after whole body vibration and maximal voluntary contractions. BMC Endocr Disord 18, 96 (2018). https://doi.org/10.1186/s12902-018-0323-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12902-018-0323-6