Abstract
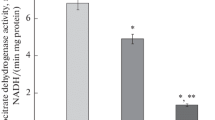
The activity of a key enzyme of the cytochrome component of the respiratory chain (cytochrome oxidase), the quantitative redistribution of mitochondrial cytochromes b, c 1, c, and aa 3, as well as the activities of the key enzymes of cytochrome heme metabolism (δ-aminolevulinate synthase and heme oxygenase) under conditions of acetaminophen-induced liver injury were studied on the background of dietary protein deprivation. Under conditions of acetaminophen-induced hepatitis that developed on the background of alimentary protein deprivation, an inhibition of cytochrome oxidase activity and a decrease in the contents of mitochondrial cytochromes on the background of an increase in the δ-aminolevulinate synthase and heme oxygenase activity were observed. In animals with a toxic liver injury that were kept under conditions of dietary protein deprivation, the contents of mitochondrial cytochromes b, c 1, c, and aa 3 progressively decreased, which was accompanied by an increase in heme oxygenase activity, whereas δ-aminolevulinate synthase activity remained at the control level. It was concluded that dietary protein deprivation is a critical factor for the development of disturbances in the structural-functional integrity of the cytochrome component of the respiratory chain. The identified changes can be considered as a possible mechanism that underlies the disturbance in the function of the energy biotransformation system under conditions of dietary protein deprivation.
Similar content being viewed by others
References
J. Borlak, B. Chatterji, K. B. Londhe, and P. B. Watkins, Genome Medicine 5 (86), 2 (2013).
E. Yu. Eremina, Gastroenterol. St. Peterb. 1, 6 (2012).
K. Somanawat, D. Thong-Ngam, and N. Klaikeaw, World J. Gastroenterol. 19 (12), 1962 (2013).
N. R. Carvalho, E. F. Rosa, M. H. Silva, et al., PLOS ONE 8 (12), e81961 (2013).
V. Sangar, A. Eddy, N. D. Price, and E. Simeonidis, Front. Physiol. 3, 404 (2012).
S. S. Kuznetsova, N. V. Azarkina, T. V. Vygodina, S. A. Siletsky, and A. A. Konstantinov, Biochemistry (Moscow) 70 (2), 128 (2005).
O. N. Voloshchuk, M. M. Marchenko, and M. S. Mudrak, Biomed. Khim. 58 (6), 684 (2012).
N. V. Kharchenko, G. A. Anokhina, and V. V. Kravchenko, Suchasna Gastroenterol. 4 (66), 76 (2012).
E. Pasini, R. Aquilani, F. S. Dioguardi, et al., Am. J. Cardiol. 101 (1), 11E–15E (2008).
G. P. Kopyl’chuk and O. M. Voloshchuk, Ukr. Biokhim. Zh. 87 (1), 20 (2015).
T. V. Barannik, N. M. Inshina, and P. A. Kaliman, Ukr. Biokhim. Zh. 77 (5), 120 (2005).
S. Bansal, G. Biswas, and N. G. Avadhan, Redox Biol. 2, 273 (2014).
O. N. Voloshchuk, G. P. Kopyl’chuk, and T. G. Kadaiskaya, Vopr. Pitaniya 3, 12 (2014).
G. Kuvandik, M. Duru, A. Nacar, et al., Toxicol. Pathol. 36, 714 (2008).
O. N. Voloshchuk and M. M. Marchenko, Byull. Eksp. Biol. Med. 156 (7), 103 (2013).
M. M. Marchenko and O. N. Voloshchuk, Radiat. Biol. Radioekol. 52 (5), 496 (2012).
A. Collins, H. Marver, and P. Tschudy, J. Biol. Chem. 241 (19), 4323 (1966).
D. P. Converso, C. Taille, M. S. Carreras, et al., FASEB J. 20 (8), 1236 (2006).
O. H. Lowry, M. J. Rosenbrough, A. L. Farr, and R. J. Rendal, J. Biol. Chem. 193 (1), 265 (1951).
H. J. Kim, O. Khalimonchuk, P. M. Smith, and D. R. Winge, Biochim. Biophys. Acta 1823 (9), 1604 (2012).
K. K. Andringa, M. L. Bajt, H. Jaeschke, and S. M. Bailey, Toxicol. Lett. 177 (3), 188 (2008).
Y. Djeridane, S. Lyoumi, H. Puy, and Y. Touitou, J. Physiol. Pharmacol. 61 (1), 115 (2010).
P. A. Kaliman and T. V. Barannik, Ukr. Biokhim. Zh. 73 (1), 5 (2001).
R. C. Scarpulla, Physiol. Rev. 88, 611 (2008).
T. Takahashi, K. Morita, R. Akagi, and S. Curr. Med. Chem. 11 (12), 1545 (2004).
W. T. Johnson and L. C. DeMars, J. Nutrit. 134, 1328 (2004).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © O.N. Voloshchuk, G.P. Kopylchuk, 2015, published in Biofizika, 2015, Vol. 60, No. 3, pp. 519–524.
Rights and permissions
About this article
Cite this article
Voloshchuk, O.N., Kopylchuk, G.P. The peculiarities of the structural and functional state of the cytochrome component of the liver mitochondrial respiratory chain under conditions of acetaminophen-induced hepatitis on the background of alimentary protein deprivation. BIOPHYSICS 60, 420–424 (2015). https://doi.org/10.1134/S0006350915030215
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006350915030215