Abstract
Background, Aim and Scope
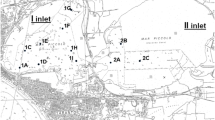
The toxicity of contaminated sediments should be evaluated considering the direct exposure of laboratory organisms to whole sediments and the indirect exposure to elutriates or extracts (Tay et al. 1992, Byrne and Halloran 1999, Nendza 2002). The alga Dunaliella tertiolecta is indicated for the use in toxicity bioassays because it is highly sensitive to several xenobiotics. Harpacticoid copepods have been already used for toxicity testing and Tigriopus fulvus is a promising Mediterranean target-species in ecotoxicology (Todaro et al. 2001, Faraponova et al. 2003, Pane et al. 2005a). In this study, the toxicity of sediments collected in harbour sites of the Northeastern Adriatic Sea was evaluated by growth inhibition test with free living and alginate-immobilized Dunaliella tertiolecta and acute toxicity test with nauplii and adult Tigriopus fulvus with the aim of pointing out the importance to utilize model organisms from different trophic levels in sediment ecotoxicology.
Methodology
Elutriates and whole sediments were tested on free living and immobilized (Pane et al. 1998) algal cells, and on laboratory reared copepods. Free-living D. tertiolecta were exposed to diluted elutriates in a static, multi-well plate system. Naalginate immobilized D. tertiolecta were placed in polystyrene inserts fitted with polyester mesh bottoms and exposed to a thin layer (2 mm) of whole sediments in multi-well plates (EPS 1992, Pane and Bertino 1999). Toxicity tests with copepods were carried out on Tigriopus fulvus nauplii (elutriates) and adults (whole sediments and elutriates). Same-aged nauplii useful for toxicity tests were obtained by egg sac detaching and consequent hatching stimulation (Pane et al. 2006). Newborn nauplii (I–II stage) were exposed to elutriates in multi-well plates provided with polystyrene inserts. Adult T. fulvus maintained in polystyrene inserts fitted with polyester mesh bottoms were placed in contact with a thin layer (2 mm) of whole sediment placed on multi-well plate bottoms. All end-points were evaluated after 96 h.
Results
In general, the effects increased with the increasing of elutriate concentration up to 50%; the stimulation or inhibition of algal growth was statistically significant in comparison to the control. The inhibiting elutriates induced EC50 variations of algal growth ranging from 66.9% to 74.3%. The mortality of T. fulvus nauplii was always < 25% after treatment with 100% elutriates and < 10% after treatment with 50% dilution; no effect was shown up with 25% dilution; therefore LC50 was not calculable. The effect of elutriates was negligible on adult copepods and LC50 values were never calculable; percent mortality always resulted in < 10% after treatment with whole sediments.
Discussion
Both experimental systems gave substantially similar results after exposition to whole sediments and elutriates. During the experiment with algal cells, the immobilization in Na-alginate and the employment of inserts which allowed the contact of organisms with sediments and their easy counting were particularly useful. Likewise, the employment of inserts of adequate mesh size in the tests with copepods allowed the contact of organisms with the sediment and made organism handling and counting easy, as well as the evaluation of mortality. The methodology here described and the utilization of the proposed test-species could have an importance also considering that the current trend in ecotoxicological research is towards finding the most appropriate organism for specific areas of concern by using indigenous species (Mariani et al. 2006) and towards the major significance of chronic and reproductive end-points.
Conclusions
Based on the above results, it can be stated that the bioassay with Dunaliella tertiolecta could be a good estimation tool for the ecotoxicological assessment of marine sediments. The immobilization of algae in Na-alginate was seen to be useful to evaluate the toxicity of whole sediments; the employment of polystyrene inserts allowed an improvement of the procedures. T. fulvus nauplii and adults, as other harpacticoids such as Tigriopus japonicus (Yoon et al. 2006), satisfy the basic criteria for the employment of a standard species in marine bioassays. To date only pelagic Acartia tonsa are utilized in the standardized procedure to evaluate the risk assessment of chemicals or wastewaters (ISO 1999). As, on the contrary, the exposure of copepods to solid-phase contaminants it is not yet standardized, the employment of polystyrene inserts improved the procedures for T. fulvus too. So, the rapidity and the possibility to solve practical problems could be the main attractive features of this technique (Pane et al. 2005a) when applied to whole sediments.
Recommendations and Perspectives
The methodology here developed being also applicable to long term and reproduction tests should be recommended because it provides relevant information in comparison with other frequently applied, standardized biotests with crustaceans (ISO 1999). The procedure has been shown to be easily applicable to selected marine organisms.
Similar content being viewed by others
References
A.P.H.A. — A.W.W. — A.W.E.F. (1995): Standard methods for the examination of water and wastewater (Eaton AD, Clesceri LS, Greenberg AE, eds) American Public Health Association, Washington DC
Antunes SC, Pereira R, Gonçalves F (2007): Evaluation of the Potential Toxicity (acute and chronic) of Sediments from Abandoned Uranium Mine Ponds. J Soils Sediments 7(6) 368–376
Apitz SE (2005): New and Recent Developments in Soil and Sediment Management, Policy and Science. Do we need a J Soils Sediments? J Soils Sediments 5, 129–133
ARPAL (2004): CTN-AIM Monitoraggio dei sedimenti e delle biocenosi marine. Raccolta, adeguamento ed integrazione delle informazioni
ASTM (1992): Designation E 1367: Standard guide for conducting 10-day static sediment toxicity tests with marine and estuarine amphipods (Vol.11.04). Philadelphia, PA: American Society for Testing and Materials
Barnett CJ, Kontogiannis JE (1975): The effect of crude oil fraction on the survival of a tidepool copepod, Tigriopus californicus. Environ Pollut 8, 45–54
Bengtsson BE (1978): Use of a harpacticoid copepod in toxicity tests. Marine Pollut Bull 9, 238–241
Bigongiari N, Braida T, Pasteris A (2001): Saggio biologico con l’anfipode Corophium orientale: Metodiche ed esempi di applicazione ai sedimenti marini. Biologia Marina Mediterranea 8(2) 60–71
Boyd D, Todd A, Jaagumagi R (2001): The influence of urban runoff on sediment quality and benthos in Toronto harbour. Ontario Ministry of Environment, Environmental Monitoring and Reporting Brunch, Ontario, 1–24
Byrne P, Halloran J (1999): Aspects of Assaying Sediment Toxicity in Irish Estuarine Ecosystems. Marine Pollut Bull 39, 97–105
Carli A, Balestra V, Pane L, Valente T (1989a): Rapporto di composizione percentuale degli acidi grassi nel Tigriopus fulvus delle pozze di scogliera della costa ligure (Copepoda Harpacticoida). Bollettino della Società Italiana di Biologia Sperimentale 55, 421–426
Carli A, Fiori A (1977): Morphological analysis of the two Tigriopus species found along the European coasts (Copepoda Harpacticoida). Natura 68, 101–110
Carli A, Mariottini GL, Pane L (1989b): Reproduction of the rockpools harpacticoid copepod Tigriopus fulvus (Fischer 1860), suitable for aquaculture. Deuxiéme Congrés International d’Aquariologie (1988), Monaco, Bulletin de l’Institut Océanographique de Monaco spécial 5, 295–300
Carli A, Mariottini GL, Pane L (1995): Influence of nutrition on fecundity and survival in Tigriopus fulvus Fischer (Copepoda: Harpacticoida). Aquaculture 134, 113–119
Carli A, Pane L, Casareto L, Bertone S, Pruzzo C (1993): Occurrence of Vibrio alginolyticus in Ligurian coast rock pools (Tyrrhenian Sea, Italy) and its association with the copepod Tigriopus fulvus (Fischer 1860). Appl Environ Microbiol 59, 1960–1962
Chapman PM (1989): Current approaches to developing sediment quality criteria. Environ Toxicol Chem 8, 589–599
Chapman PM, Long ER (1983): The use of bioassays as part of a comprehensive approach to marine pollution assessment. Marine Pollut Bull 14, 81–84
Cheung KC, Wong MH, Yung YK (2003): Toxicity assessment of sediments containing tributyltin around Hong Kong Harbour. Toxicol Letters 137, 121–131
Diogo JB, Natal-da-Luz T, Sousa JP, Vogt C, Nowak C (2007): Tolerance of Genetically Characterized Folsomia candida Strains to Phenmedipham Exposure. A comparison between reproduction and avoidance tests. J Soils Sediments 7(6) 388–392
Ducrot V, Cognat C, Mons R, Mouthon J, Garric J (2006): Development of rearing and testing protocols for a new freshwater sediment test species: the gastropod Valvata piscinalis. Chemosphere 62, 1272–1281
EPA (1994): Methods for assessing the toxicity of sediment-associated contaminants with estuarine and marine amphipods. 600/R-94/025
EPA (2001): Method for collection, storage and manipulation of sediments for chemical and toxicological analyses. Technical manual. 823-B-01-002
EPS (1992): Biological Test Method: Growth Inhibition Test Using the Freshwater Alga Selenastrum capricornutum. Report EPS 1/RM/25
Faraponova O, Todaro MA, Onorati F, Finora MG (2003): Sensibilità sesso ed età specifica di Tigriopus fulvus (Copepoda, Harpacticoida) nei confronti di due metalli pesanti (Cadmio e Rame). Biologia Marina Mediterranea 10, 679–681
Green AS, Chandler GT, Piegorsch WW (1996): Life-stage-specific toxicity of sediment-associated Chlorpyrifos to a marine, infaunal copepod. Environ Toxicol Chem 15, 1182–1188
Guillard RRL, Ryther JH (1962): Studies of marine planktonic diatoms. I. Cyclotella nana Hustedt and Detonula confervacea Cleve. Canadian J Microbiol 8, 229–239
Hamilton MA, Russo RC, Thurston RV (1977): Trimmed Spearman-Karber method for estimating median lethal concentrations in toxicity bioassays. Environ Sci Technol 11, 714–719; Correction 12, 417 (1978)
Hoke R, Ankley G (1991): Results of dieldrin sediment spiking study conducted in support of US EPA development of sediment quality criteria. In: Sediment quality criteria for the protection of benthic organisms: Dieldrin (Draft Report). Washington, DC: US Environmental Protection Agency, pp 1–52
Hollert H, Ahlf W, Heise S, Manz W, Scrimshaw M, White S (2007): Controversies and Solutions in Environmental Sciences — Addressing toxicity of sediments and soils (Editorial). J Soils Sediments 7(6) 360
Hsu P, Matthäi A, Heise S, Ahlf W (2007): Seasonal variation of sediment toxicity in the Rivers Dommel and Elbe Environmental Pollution 148, 817–823
Hutchinson TH, Williams TD, Eales GJ (1994): Toxicity of cadmium, hexavalent chromium and copper to marine fish larvae (Cyprinodon variegatus) and copepods (Tisbe battagliai). Marine Environmental Research 38, 275–290
IRSA-CNR (1978): Metodologia di saggio algale per lo studio della contaminazione delle acque marine. Quaderni dell’Istituto di Ricerca sulle Acque, 39. IRSA-CNR Milano
ISO (1999): Water quality — Determination of acute lethal toxicity to marine copepods (Copepoda, Crustacea). International standard 14669
Kosmehl T, Krebs F, Manz W, Braunbeck T, Hollert H (2007): Differentiation Between Bioavailable and Total Hazard Potential of Sediment Induced DNA Fragmentation as Measured by the Comet Assay with Zebrafish Embryos. J Soils Sediments 7(6) 377–387
Mariani L, De Pascale D, Faraponova O, Tornambè A, Sarni A, Giuliani S, Ruggiero G, Onorati F, Magaletti E (2006): The use of a battery test in marine ecotoxicology: The acute toxicity of sodium dodecyl sulfate. Environ Toxicol, special issue 21, 373–379
Miliou H, Verriopoulos G, Maroulis D, Bouloukos D, Moraitou-Apostolopoulou M (2000): Influence of life history adaptations on the fidelity of laboratory bioassays for the impact of heavy metals (Co2+ and Cr6+) on the tolerance and population dynamics of Tisbe holothuriae. Marine Pollut Bull 40, 352–359
Moreira SM, Guilhermino L, Ribeiro R (2006): An in situ assay with the microalga Phaeodactylum tricornutum for sediment-overlying water toxicity evaluations in estuaries. Environ Toxicol Chem 25, 2272–2279
Nendza M (2002): Inventory of marine biotest methods for the evaluation of dredged material and sediments. Chemosphere 48, 865–883
Norr C, Riepert F (2007): Bioaccumulation Studies with Eisenia fetida Using an Established Degradation Test System. J Soils Sediments 7(6) 393–397
OECD (1984): Alga Growth Inhibition Test. OECD Guideline for Testing of Chemicals 201. Paris, France
Onorati F, Bigongiari N, Pellegrini D, Giuliani S (1999): The suitability of Corophium orientale (Crustacea, Amphipoda) in harbour sediment toxicity bioassessment. Aquatic Ecosystem, Health Management 2, 465–473
Onorati F, Volpi Ghirardini A (2001): Informazioni fornite dalle diverse matrici da testare con i saggi biologici: applicabilityà di Vibrio fischeri. Biologia Marina Mediterranea 8(2) 31–40
Pane L, Bertino C (1999): Immobilizzazione di alghe fitoplanctoniche in alginato di calcio e colture in piastra. Biologi Italiani 29, 9–14
Pane L, Boccardo S, Bonfiglioli F, Mariottini GL, Priano F, Conio O (2005b): Polycyclic aromatic hydrocarbons in water, seston and copepods in a harbour area in the Western Mediterranean (Ligurian Sea). Marine Ecol 26, 89–99
Pane L, De Nuccio L, Bozzolasco M, Mariottini GL, Debbia E (2003): Studio dell’associazione zooplancton-batteri in un’area neritica del Golfo di Genova (Mar Ligure). Biologia Marina Mediterranea 10, 1022–1025
Pane L, De Nuccio L, Pruzzo C, Carli A (2000): Adhesion of bacteria and diatoms to the exoskeleton of the harpacticoid copepod Tigriopus fulvus in culture: electron and epifluorescent microscopy study. J Biol Res 76, 37–43
Pane L, Feletti M, Bertino C, Carli A (1998): Viability of the marine microalga Tetraselmis suecica grown free and immobilized in alginate beads. Aquaculture International 6, 411–420
Pane L, Giacco E, Mariottini GL (2005a): Acute and chronic heavy metal bioassay on Tigriopus fulvus Fischer (Copepoda: Harpacticoida). 13th Symposium PRIMO 13, Pollutant response in marine organisms, Alessandria, Italy. June 19–22, 2005, 113
Pane L, Giacco E, Mariottini GL (2006): Utilizzo di Tigriopus fulvus (Copepoda: Harpacticoida) in ecotossicologia. Saggi con disperdenti e tensioattivi. Biologia Marina Mediterranea 13(2) 348–349
Penttinen O-P, Kilpi-Koski J, Jokela M, Toivainen K, Väisänen A (2008): Importance of Dose Metrics for Lethal and Sublethal Sediment Metal Toxicity in the Oligochaete Worm Lumbriculus variegatus. J Soils Sediments 8(1) 59–66
Peters C, Becker S, Noack U, Pfitzner S, Bülow W, Barz K, Ahlf W, Berghahn R (2002): A marine bioassay test set to assess marine water and sediment quality. Its need, the approach and first results. Ecotoxicol 11, 379–383
Plesha PD, Stein JE, Schiewe MH, McCain BB, Varanasi U (1988): Toxicity of marine sediments supplemented with mixtures of selected chlorinated and aromatic hydrocarbons to the infaunal amphipod Rhepoxynius abronius. Marine Environ Res 25, 85–97
Raisuddin S, Kwok KWH, Leung KMY, Schlenk D, Lee J-S (2007): The copepod Tigriopus: A promising marine model organism for ecotoxicology and environmental genomics. Aquatic Toxicol 83, 161–173
Richter S, Nagel R (2007): Bioconcentration, biomagnification and metabolism of 14C-terbutryn and 14C-benzo[a]pyrene in Gammarus fossarum and Asellus aquaticus Chemosphere 66, 603–610
Salomons W, De Rooij NM, Kerdijk H, Bril J (1987): Sediments as a source for contaminants. Hydrobiologia, 149, 13–30
Saraiva MC (1973): Étude sur la radiosensibilité d’un copépode benthique, Tigriopus fulvus (Fischer). Journal du Conseil International pour l’Exploration de la Mer 35, 7–12
Sbrilli G, Limberti A, Caldini G, Corsini A (1998): Metodologia di saggio algale per il controllo dei corpi idrici e delle acque di scarico. ARPAT Firenze, pp 1–191
Scarlett A, Galloway TS, Rowland SJ (2007): Chronic Toxicity of Unresolved Complex Mixtures (UCM) of Hydrocarbons in Marine Sediments. J Soils Sediments 7(4) 200–206
Schulze T, Ricking M, Schröter-Kermani C, Körner A, Denner H-D, Weinfurtner K, Winkler A, Pekdeger A (2007): The German Environmental Specimen Bank. Sampling, processing, and archiving sediment and suspended particulate matter. J Soils Sediments 7(6) 361–367
Scrimshaw M, White S, Manz W, Hollert H, Ahlf W, Heise S (2006): Sediment-related Discussions at the 16th SETAC Europe Annual Meeting. J Soils Sediments 6(2) 116–117
Swartz RC, Kemp PF, Schultz DW, Lamberson JO (1988): Effects of mixtures of sediment contaminants on the marine infaunal amphipod, Rhepoxinius abronius. Environ Toxicol Chem 7, 1013–1020
Swartz RC, Schults DW, Ozretich RJ, Lamberson JO, Cole FA, De Witt TH, Redmond MS, Ferraro SP (1995): PAH: a model to predict the toxicity of polynuclear aromatic hydro-carbon mixtures in field-collected sediments. Environ Toxicol Chem 14, 1977–1987
Swartz RC, Schultz DW, De Witt TH, Ditsworth GR, Lamberson JO (1990): Toxicity of fluoranthene in sediment to marine amphipods: a test of the equilibrium partitioning approach to sediment quality criteria. Environmental Toxicology and Chemistry 9, 1071–1080
Tay KL, Doe KG, Wade SJ, Vaughan DA, Berrigan RE, Moore MJ (1992): Sediment bioassessment in Halifax Harbour. Environ Toxicol Chem 11, 1567–1581
Telli-Karakoç F, Gaines AF, Hewer A, Phillips D (2001): Differences between blood and liver aromatic DNA adduct formation. Environ International 26,143–148
Tessier A, Campbell PGC (1987): Partitioning of trace metals in sediments: relationships with bioavailability. Hydrobiologia, 149, 43–52
Thompson B, Anderson B, Hunt J, Taberski K, Phillips B (1999): Relationships between sediment contamination and toxicity in San Francisco Bay. Marine Environmental Research 48, 285–309
Todaro MA, Faraponova O, Onorati F, Pellegrini D, Tongiorgi P (2001): Tigriopus fulvus (Copepoda, Harpacticoida) una possibile specie-target nella valutazione della tossicità dei fanghi portuali: ciclo vitale e prove tossicologiche preliminari. Biologia Marina Mediterranea 8, 869–872
UNI EN ISO 10253 (2000): Saggio di inibizione della crescita di alghe marine con Skeletonema costatum e Phaeodactylum tricornutum. pp 1–14
Yoon S-J, Park G-S, Oh J-H, Kang Y-S, Park S-Y (2006): Marine ecotoxicological assessment using the harpacticoid copepod Tigriopus japonicus. SETAC Europe 16th Annual Meeting 7–11 May 2006, The Hague, The Netherlands
Author information
Authors and Affiliations
Corresponding author
Additional information
ESS-Submission Editor: Prof. Dr. Henner Hollert (henner.hollert@bio5.rwth-aachen.de)
Rights and permissions
About this article
Cite this article
Pane, L., Giacco, E., Corrà, C. et al. Ecotoxicological evaluation of Harbour sediments using marine organisms from different trophic levels. J Soils Sediments 8, 74–79 (2008). https://doi.org/10.1065/jss2008.02.272
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1065/jss2008.02.272