Abstract
Hearing loss is associated with an increased risk of Alzheimer disease (AD). However, the mechanisms of hearing loss promoting the onset of AD are poorly understood. Here we show that hearing loss aggravates cognitive impairment in both wild-type mice and mouse models of AD. Embryonic growth/differentiation factor 1 (GDF1) is downregulated in the hippocampus of deaf mice. Knockdown of GDF1 mimics the detrimental effect of hearing loss on cognition, while overexpression of GDF1 in the hippocampus attenuates the cognitive impairment induced by deafness. Strikingly, overexpression of GDF1 also attenuates cognitive impairment in APP/PS1 transgenic mice. GDF1 activates Akt, which phosphorylates asparagine endopeptidase and inhibits asparagine endopeptidase-induced synaptic degeneration and amyloid-β production. The expression of GDF1 is downregulated by the transcription factor CCAAT-enhancer binding protein-β. These findings indicate that hearing loss could promote AD pathological changes by inhibiting the GDF1 signaling pathway; thus, GDF1 may represent a therapeutic target for AD.






Similar content being viewed by others
Data availability
Additional data that support the findings of this study are available from the corresponding authors upon reasonable request. All the RNA-seq results presented in this paper are based on the processed data generated by bioinformatics specialists at the BGI, which are presented in Supplementary Table 1. The raw reads of the RNA-seq analyses presented in Fig. 2a–d are not available because they were deleted by BGI before they could be retrieved by the authors at the time of acceptance of this manuscript. Source data are provided with this paper.
References
Lee, M. et al. Variation in population attributable fraction of dementia associated with potentially modifiable risk factors by race and ethnicity in the US. JAMA Netw. Open 5, e2219672 (2022).
Ford, A. H. et al. Hearing loss and the risk of dementia in later life. Maturitas 112, 1–11 (2018).
Liu, C.-M. & Lee, C. T.-C. Association of hearing loss with dementia. JAMA Netw. Open 2, e198112 (2019).
Loughrey, D. G., Kelly, M. E., Kelley, G. A., Brennan, S. & Lawlor, B. A. Association of age-related hearing loss with cognitive function, cognitive impairment, and dementia. JAMA Otolaryngol. Head Neck Surg. 144, 115–126 (2018).
Livingston, G. et al. Dementia prevention, intervention, and care: 2020 report of the Lancet Commission. Lancet 396, 413–446 (2020).
Scheltens, P. et al. Alzheimer’s disease. Lancet 397, 1577–1590 (2021).
Nedelec, T. et al. Identifying health conditions associated with Alzheimer’s disease up to 15 years before diagnosis: an agnostic study of French and British health records. Lancet Digit. Health 4, e169–e178 (2022).
Johnson, J. C. S. et al. Hearing and dementia: from ears to brain. Brain 144, 391–401 (2021).
Griffiths, T. D. et al. How can hearing loss cause dementia? Neuron 108, 401–412 (2020).
Swords, G. M., Nguyen, L. T., Mudar, R. A. & Llano, D. A. Auditory system dysfunction in Alzheimer disease and its prodromal states: a review. Ageing Res. Rev. 44, 49–59 (2018).
Coebergh, J. A. F. et al. Auditory agnosia for environmental sounds in Alzheimer’s disease: not hearing and not listening? J. Alzheimers Dis. 73, 1407–1419 (2020).
Salemme, S. et al. Pure word deafness: a case report of an atypical manifestation of Alzheimer’s disease. Neurol. Sci. 43, 5275–5279 (2022).
Brenowitz, W. D. et al. Association of genetic risk for Alzheimer disease and hearing impairment. Neurology 95, e2225–e2234 (2020).
Liu, Y. et al. Hearing loss is an early biomarker in APP/PS1 Alzheimer’s disease mice. Neurosci. Lett. 717, 134705 (2020).
Mei, L., Liu, L.-M., Chen, K. & Zhao, H.-B. Early functional and cognitive declines measured by auditory-evoked cortical potentials in mice with Alzheimer’s disease. Front. Aging Neurosci. 13, 710317 (2021).
Park, S. Y. et al. Cognitive decline and increased hippocampal p-tau expression in mice with hearing loss. Behav. Brain Res. 342, 19–26 (2018).
Mun, S.-K. et al. MicroRNAs related to cognitive impairment after hearing loss. Clin. Exp. Otorhinolaryngol. 14, 76–81 (2021).
Kim, J. S. et al. Conductive hearing loss aggravates memory decline in Alzheimer model mice. Front. Neurosci. 14, 843 (2020).
Paciello, F. et al. Auditory sensory deprivation induced by noise exposure exacerbates cognitive decline in a mouse model of Alzheimer’s disease. eLife 10, e70908 (2021).
Armstrong, N. M. et al. Association of midlife hearing impairment with late-life temporal lobe volume loss. JAMA Otolaryngol. Head Neck Surg. 145, 794–802 (2019).
Golub, J. S., Sharma, R. K., Rippon, B. Q., Brickman, A. M. & Luchsinger, J. A. The association between early age-related hearing loss and brain β-amyloid. Laryngoscope 131, 633–638 (2021).
Irace, A. L., Rippon, B. Q., Brickman, A. M., Luchsinger, J. A. & Golub, J. S. The Laterality of early age-related hearing loss and brain β-amyloid. Otol. Neurotol. 43, e382–e390 (2022).
Wang, H.-F. et al. Hearing impairment is associated with cognitive decline, brain atrophy and tau pathology. EbioMedicine 86, 104336 (2022).
Xu, W. et al. Age-related hearing loss accelerates cerebrospinal fluid tau levels and brain atrophy: a longitudinal study. Aging 11, 3156–3169 (2019).
Zheng, M. et al. Worsening hearing was associated with higher β-amyloid and tau burden in age-related hearing loss. Sci. Rep. 12, 10493 (2022).
Lin, F. R. et al. Hearing loss and incident dementia. Arch. Neurol. 68, 214–220 (2011).
Nianogo, R. A. et al. Risk factors associated with Alzheimer disease and related dementias by sex and race and ethnicity in the US. JAMA Neurol. 79, 584–591 (2022).
Adrait, A. et al. Do hearing aids influence behavioral and psychological symptoms of dementia and quality of life in hearing impaired Alzheimer’s disease patients and their caregivers? J. Alzheimers Dis. 58, 109–121 (2017).
Dillard, L. K. et al. Associations of hearing loss and hearing aid use with cognition, health-related quality of life, and depressive symptoms. J. Aging Health 35, 455–465 (2023).
Mahmoudi, E. et al. Can hearing aids delay time to diagnosis of dementia, depression, or falls in older adults? J. Am. Geriatr. Soc. 67, 2362–2369 (2019).
Neff, R. M. et al. Neuropathological findings of dementia associated with subjective hearing loss. Otol. Neurotol. 40, e883–e893 (2019).
Yang, Z. et al. Effect of hearing aids on cognitive functions in middle-aged and older adults with hearing loss: a systematic review and meta-analysis. Front. Aging Neurosci. 14, 1017882 (2022).
Derynck, R. & Budi, E. H. Specificity, versatility and control of TGF-β family signaling. Sci. Signal. 12, eaav5183 (2019).
Onishi, Y. et al. GDF1 is a novel mediator of macrophage infiltration in brown adipose tissue of obese mice. Biochem. Biophys. Rep. 5, 216–223 (2016).
Zhang, Z. et al. Delta-secretase cleaves amyloid precursor protein and regulates the pathogenesis in Alzheimer’s disease. Nat. Commun. 6, 8762 (2015).
Wang, Z.-H. et al. BDNF inhibits neurodegenerative disease-associated asparaginyl endopeptidase activity via phosphorylation by AKT. JCI Insight 3, e99007 (2018).
Heldin, C.-H. & Moustakas, A. Signaling receptors for TGF-β family members. Cold Spring Harb. Perspect. Biol. 8, a022053 (2016).
Wang, Z.-H. et al. C/EBPβ regulates delta-secretase expression and mediates pathogenesis in mouse models of Alzheimer’s disease. Nat. Commun. 9, 1784 (2018).
Spike, A. J. & Rosen, J. M. C/EBPß isoform specific gene regulation: it’s a lot more complicated than you think! J. Mammary Gland Biol. Neoplasia 25, 1–12 (2020).
Müller, C. et al. Reduced expression of C/EBPβ-LIP extends health and lifespan in mice. eLife 7, e34985 (2018).
Omura, J. D. et al. Modifiable risk factors for Alzheimer disease and related dementias among adults aged ≥45 years—United States, 2019. MMWR Morb. Mortal. Wkly Rep. 71, 680–685 (2022).
Cunningham, L. L. & Tucci, D. L. Hearing loss in adults. N. Engl. J. Med. 377, 2465–2473 (2017).
Nieman, C. L. & Oh, E. S. Hearing loss. Ann. Intern. Med. 173, ITC81–ITC96 (2020).
Wu, W. J. et al. Aminoglycoside ototoxicity in adult CBA, C57BL and BALB mice and the Sprague–Dawley rat. Hear. Res. 158, 165–178 (2001).
Li, Y., Liu, S., Teng, Q., Gong, S. & Liu, K. A method for constructing a mouse model of congenital hearing loss by bilateral cochlear ablation. J. Neurosci. Methods 378, 109641 (2022).
Mostafapour, S. P., Cochran, S. L., Del Puerto, N. M. & Rubel, E. W. Patterns of cell death in mouse anteroventral cochlear nucleus neurons after unilateral cochlea removal. J. Comp. Neurol. 426, 561–571 (2000).
Llano, D. A., Issa, L. K., Devanarayan, P. & Devanarayan, V. & Alzheimer’s Disease Neuroimaging Initiative (ADNI). Hearing loss in Alzheimer’s disease is associated with altered serum lipidomic biomarker profiles. Cells 9, 2556 (2020).
Llano, D. A., Kwok, S. S., Devanarayan, V. & Alzheimer’s Disease Neuroimaging Initiative (ADNI). Reported hearing loss in Alzheimer’s disease is associated with loss of brainstem and cerebellar volume. Front. Hum. Neurosci. 15, 739754 (2021).
Zhang, W.-J. et al. Clinical features and potential mechanisms relating neuropathological biomarkers and blood–brain barrier in patients with Alzheimer’s disease and hearing loss. Front. Aging Neurosci. 14, 911028 (2022).
Wang, Z.-H. et al. Neuronal ApoE4 stimulates C/EBPβ activation, promoting Alzheimer’s disease pathology in a mouse model. Prog. Neurobiol. 209, 102212 (2022).
Martorell, A. J. et al. Multi-sensory gamma stimulation ameliorates Alzheimer’s-associated pathology and improves cognition. Cell 177, 256–271 (2019).
Spongr, V. P., Flood, D. G., Frisina, R. D. & Salvi, R. J. Quantitative measures of hair cell loss in CBA and C57BL/6 mice throughout their life spans. J. Acoust. Soc. Am. 101, 3546–3553 (1997).
Henry, K. R. & Chole, R. A. Genotypic differences in behavioral, physiological and anatomical expressions of age-related hearing loss in the laboratory mouse. Audiology 19, 369–383 (1980).
Rankin, C. T., Bunton, T., Lawler, A. M. & Lee, S. J. Regulation of left-right patterning in mice by growth/differentiation factor-1. Nat. Genet. 24, 262–265 (2000).
Yang, W. et al. Epigenetic silencing of GDF1 disrupts SMAD signaling to reinforce gastric cancer development. Oncogene. 35, 2133–2144 (2016).
Cheng, W. et al. Growth differentiation factor 1-induced tumour plasticity provides a therapeutic window for immunotherapy in hepatocellular carcinoma. Nat. Commun. 12, 7142 (2021).
Lee, S. J. Expression of growth/differentiation factor 1 in the nervous system: conservation of a bicistronic structure. Proc. Natl Acad. Sci. USA 88, 4250–4254 (1991).
Jaeger, P. A. et al. Network-driven plasma proteomics expose molecular changes in the Alzheimer’s brain. Mol. Neurodegener. 11, 31 (2016).
Hinck, A. P., Mueller, T. D. & Springer, T. A. Structural biology and evolution of the TGF-β family. Cold Spring Harb. Perspect. Biol. 8, a022103 (2016).
Zhang, Y. E. Non-Smad signaling pathways of the TGF-β family. Cold Spring Harb. Perspect. Biol. 9, a022129 (2017).
Derynck, R., Zhang, Y. & Feng, X. H. Transcriptional activators of TGF-β responses: Smads. Cell 95, 737–740 (1998).
Derynck, R. & Zhang, Y. E. Smad-dependent and Smad-independent pathways in TGF-β family signalling. Nature 425, 577–584 (2003).
Yu, J. et al. Akt and calcium-permeable AMPA receptor are involved in the effect of pinoresinol on amyloid β-induced synaptic plasticity and memory deficits. Biochem. Pharmacol. 184, 114366 (2021).
Rai, S. N. et al. The role of PI3K/Akt and ERK in neurodegenerative disorders. Neurotox. Res. 35, 775–795 (2019).
Singh, A. K. et al. Neuroprotection through rapamycin-induced activation of autophagy and PI3K/Akt1/mTOR/CREB signaling against amyloid-β-induced oxidative stress, synaptic/neurotransmission dysfunction, and neurodegeneration in adult rats. Mol. Neurobiol. 54, 5815–5828 (2017).
Shah, S. A., Lee, H. Y., Bressan, R. A., Yun, D. J. & Kim, M. O. Novel osmotin attenuates glutamate-induced synaptic dysfunction and neurodegeneration via the JNK/PI3K/Akt pathway in postnatal rat brain. Cell Death Dis. 5, e1026 (2014).
Beaulieu, J.-M. et al. An Akt/β-arrestin 2/PP2A signaling complex mediates dopaminergic neurotransmission and behavior. Cell 122, 261–273 (2005).
Wang, Q. et al. Control of synaptic strength, a novel function of Akt. Neuron 38, 915–928 (2003).
Wu, Z. et al. Neurotrophic signaling deficiency exacerbates environmental risks for Alzheimer’s disease pathogenesis. Proc. Natl Acad. Sci. USA 118, e2100986118 (2021).
Wang, Z.-H. et al. Deficiency in BDNF/TrkB neurotrophic activity stimulates δ-secretase by upregulating C/EBPβ in Alzheimer’s disease. Cell Rep. 28, 655–669 (2019).
Jankowsky, J. L. et al. Mutant presenilins specifically elevate the levels of the 42 residue β-amyloid peptide in vivo: evidence for augmentation of a 42-specific γ secretase. Hum. Mol. Genet. 13, 159–170 (2004).
Wu, W. J. et al. Aminoglycoside ototoxicity in adult CBA, C57BL and BALB mice and the Sprague–Dawley rat. Hear. Res. 158, 165–178 (2001).
Chen, S. et al. Developmental abnormalities in supporting cell phalangeal processes and cytoskeleton in the Gjb2 knockdown mouse model. Dis. Model. Mech. 11, dmm033019 (2018).
Tian, Y. et al. Transgenic mice expressing human α-synuclein 1–103 fragment as a novel model of Parkinson’s disease. Front. Aging Neurosci. 13, 760781 (2021).
Kaech, S. & Banker, G. Culturing hippocampal neurons. Nat. Protoc. 1, 2406–2415 (2006).
Pan, L. et al. Tau accelerates α-synuclein aggregation and spreading in Parkinson’s disease. Brain 145, 3454–3471 (2022).
O’Brien, C. E., Bonanno, L., Zhang, H. & Wyss-Coray, T. Beclin 1 regulates neuronal transforming growth factor-β signaling by mediating recycling of the type I receptor ALK5. Mol. Neurodegener. 10, 69 (2015).
Zhang, X. et al. Amphiphysin I cleavage by asparagine endopeptidase leads to tau hyperphosphorylation and synaptic dysfunction. eLife 10, e65301 (2021).
Sahara, M. et al. Manipulation of a VEGF-Notch signaling circuit drives formation of functional vascular endothelial progenitors from human pluripotent stem cells. Cell Res. 24, 820–841 (2014).
Zhang, Z. et al. Cleavage of tau by asparagine endopeptidase mediates the neurofibrillary pathology in Alzheimer’s disease. Nat. Med. 20, 1254–1262 (2014).
Nunez, J. Morris water maze experiment. J. Vis. Exp. (19), 897 (2008).
Zhang, G. et al. Islet amyloid polypeptide cross-seeds tau and drives the neurofibrillary pathology in Alzheimer’s disease. Mol. Neurodegener. 17, 12 (2022).
Wang, Q. et al. Activation of Wnt/β-catenin pathway mitigates blood–brain barrier dysfunction in Alzheimer’s disease. Brain 145, 4474–4488 (2022).
Acknowledgements
This work was supported by grants from the National Key Research and Development Program of China (no. 2019YFE0115900), the National Natural Science Foundation of China (nos. 82271447 and 81822016) and the Innovative Research Groups of Hubei Province (no. 2022CFA026).
Author information
Authors and Affiliations
Contributions
Z.Z. conceived the project and designed the experiments. L.P. and C.L. performed most of the experiments. L.M., G.Z., D.S., X.Z., M.X. and Z.H. helped with the cell culture and animal experiments. L.Z. and D.X. performed the electrophysiological experiments. Y.T. and T.X. helped analyze the RNA-seq data. S.C. and Y.S. helped with the ABR experiments.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Aging thanks Hongbo Zhao, Shinichi Someya, Yadong Huang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Cochlear ablation promotes AD pathologies.
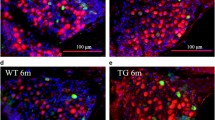
a, b, Representative images of Aβ deposition in APP/PS1 mice 6 m after cochlear ablation (n = 6 mice per group). AuC, auditory cortex; TeA, temporal association cortex. Scale bar, 200 μm. c, d, ThS staining of Aβ plaques in the hippocampus of WT and APP/PS1 mice (n = 6 mice per group). Scale bar, 50 μm. e, RT-PCR shows the mRNA levels of mAPP and HuAPP in the hippocampus (n = 6 mice per group). f, Western blot analysis of ADAM10 and BACE1 levels in the hippocampus (n = 5 mice per mouse). Right, quantification analysis. Data are presented as means ± SEM. P values were determined by two-sided Mann-Whitney test (b, d) or two-way ANOVA (e, f). *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001. n.s., not significant.
Extended Data Fig. 2 Cochlear ablation leads to synaptic dysfunction.
a, b, Morris water maze test showed no difference in escape latency (a) or swim speed (b) in the visible training (n = 13 in the WT-SO group, n = 7 in the WT-CA group, n = 15 in the APP/PS1-SO group, n = 11 in the APP/PS1-CA group). c-g, Electron microscopy of synapses. Shown are the representative images (c), synaptic density (d), the average thickness of PSD (e), length of the active zone (f), and width of synaptic clefts (g). Scale bar, 2 μm. (n = 6 fields per group). h, Western blot analysis of synaptic proteins in the hippocampus (n = 5 mice per group). Right, quantification of PSD95, synapsin 1, synaptophysin, and VAMP2. Data are presented as means ± SEM. P values were determined by two-way ANOVA (a, b, d-h). *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001. n.s., not significant.
Extended Data Fig. 3 Administration of kanamycin promotes AD pathologies.
a, Schematic diagram of the experimental procedure and timeline. Subcutaneous injection of normal saline (NS) or kanamycin (Kana) was conducted on WT and APP/PS1 mice at 3 months of age. b, ABR threshold results. The left panel shows click-evoked ABR thresholds at 1 month after saline or kanamycin injection. The right panel shows pure tone-evoked ABR thresholds 1 month and 6 months after modeling (n = 13 in the WT-NS group, n = 14 in the WT-Kana group, n = 10 in the APP/PS1-NS group, n = 8 in the APP/PS1-Kana group). c, Immunofluorescence (red) and ThS staining (green) of Aβ deposition. AuC, auditory cortex. Scale bar, 50 μm. d, Quantitative analysis of immunofluorescence (n = 8 mice per group). e, Western blot detection of APP and CTF fragments in hippocampal lysates from WT and APP/PS1 mice at 6 months after kanamycin injection (n = 5 mice per group). f, Quantification of Western blots. g, RT-PCR shows the mRNA levels of mAPP and HuAPP in the hippocampus (n = 6 mice per group). h, Western blot analysis of ADAM10 and BACE1 levels in the hippocampus (n = 5 mice per group). i, Quantification analysis of (h). Data are presented as means ± SEM. P values were determined by two-sided Mann-Whitney test (d), or two-way ANOVA (b, f, g, i). *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001, n.s., not significant.
Extended Data Fig. 4 Kanamycin-induced hearing loss leads to cognitive impairment.
a-f, Spatial memory was determined in the Morris water maze test. Shown are the escape latency (a) and swim speed (b) in the visible train, escape latency in the hidden training (c-e), and time in the target zone in the probe trial (f). AUC, area under the curve. g, h, Working memory was assessed in the Y-maze test 6 months after kanamycin injection in WT and APP/PS1 mice. n = 13 in the WT-NS group, n = 14 in the WT-Kana group, n = 10 in the APP/PS1-NS group, n = 8 in the APP/PS1-Kana group. i, Western blot analysis of synaptic proteins in the hippocampus. j, Quantification of PSD95, synapsin 1, synaptophysin, and VAMP2 (n = 5 mice per group). Data are presented as means ± SEM. P values were determined by two-way ANOVA (a, b, d, e, g, j) or Kruskal-Wallis tests (f, h). *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001, n.s., not significant.
Extended Data Fig. 5 KEGG pathway enrichment analysis in WT mice.
a, Top 20 KEGG pathway terms gathering most DEGs in WT mice as determined by enrichment analysis. b, KEGG interaction network illustrating the relationship among the most significant DEGs in WT mice ranked by q-values.
Extended Data Fig. 6 GDF1 levels in mouse brains injected with AAV-GDF1 or AAV-shGDF1.
a, Levels of GDF1 in hippocampal tissue from WT or APP/PS1 mice injected with AAV-GDF1 or control AAVs. b, Quantification analysis of (a). n = 5 mice per group. c, Double-labeling immunofluorescence of hippocampal sections from control, GDF1-overexpressing (GDF1 OE), and GDF1-knockdown (GDF1 KD) mice. GDF1 (red) and MAP2, Iba1, GFAP (green). Regions in the box are enlarged at right. Arrowheads indicate colocalization. Intensity trace are plotted below. The upper right panel shows the quantification of the fraction of GDF1 colocalizing with MAP2, Iba1, or GFAP. The bottom right panel shows the quantification of GDF1 fluorescence intensity in control, GDF1 OE, and GDF1 KD mice. AFU, Arbitrary fluorescence intensity. n = 5 fields per group. Scale bar, 20 μm. Data are presented as means ± SEM. P values were determined by two-way ANOVA (b) or one-way ANOVA (c). *P < 0.05, **P < 0.01.
Extended Data Fig. 7 GDF1 knockdown inhibits AKT signaling pathway.
a, Levels of GDF1 in hippocampal tissues from WT or APP/PS1 mice injected with AAV-shGDF1 or control virus. b, Quantitative analysis of (a). n = 5 mice per group. c, Western blot analysis of AKT, p-AKT, AEP, and APP N585 in the hippocampus. d, Quantitative analysis of (c) (n = 5 mice per group). e, AEP activity assay. n = 6 mice per group. Data are presented as means ± SEM. P values were determined by two-way ANOVA (b, d, e). *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001. n.s., not significant.
Extended Data Fig. 8 GDF1 overexpression attenuates AD pathogenesis.
a, Schematic diagram of the experimental procedure and timeline. b, Levels of GDF1 in hippocampal tissue of WT or APP/PS1 mice injected with GFP-GDF1 virus or control virus. c, Quantification analysis of (b) (n = 5 mice per group). d, The expression of GFP in mice injected with GFP-vector or GFP-GDF1 virus. Scale bar, 200 μm. e, Pure tone-evoked ABR thresholds 1 month and 5 months after AAV injection (n = 12 in the WT-Vector group, n = 12 in the WT-GDF1 group, n = 8 in the APP/PS1-Vector group, n = 8 in the APP/PS1-GDF1 group). f-k, Spatial memory was determined in the Morris water maze test. Shown are the escape latency (f) and swim speed (g) in the visible train, the escape latency in the hidden training (h-j), and time in the target zone in the probe trial (k). AUC, area under the curve. n = 12 in the WT-Vector group, n = 12 in the WT-GDF1 group, n = 8 in the APP/PS1-Vector group, n = 8 in the APP/PS1-GDF1 group. l, m, IHC analysis of Aβ deposition in the hippocampus and retrosplenial area (RSP) of APP/PS1 mice. Brain slices were stained with human Aβ antibody. Scale bar, 50 μm. n = 6 mice per group. n, o, Effect of GDF1 on APP processing in the hippocampus (n = 5 mice per group). Data are presented as means ± SEM. P values were determined by two-sided Kruskal-Wallis tests (k), two-sided Mann-Whitney test (m), or two-way ANOVA test (c, f, g, i, j, o). *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001. n.s., not significant.
Supplementary information
Supplementary Table 1
RNA-seq data.
Supplementary Table 2
Sequences of oligonucleotides.
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 1
Unprocessed immunoblots.
Source Data Fig. 2
Statistical source data.
Source Data Fig. 2
Unprocessed immunoblots.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 3
Unprocessed immunoblots.
Source Data Fig. 4
Statistical source data.
Source Data Fig. 4
Unprocessed immunoblots.
Source Data Fig. 5
Statistical source data.
Source Data Fig. 5
Unprocessed immunoblots.
Source Data Fig. 6
Statistical source data.
Source Data Fig. 6
Unprocessed immunoblots.
Source Data Extended Data Fig. 1
Statistical source data.
Source Data Extended Data Fig. 1
Unprocessed immunoblots.
Source Data Extended Data Fig. 2
Statistical source data.
Source Data Extended Data Fig. 2
Unprocessed immunoblots.
Source Data Extended Data Fig. 3
Statistical source data.
Source Data Extended Data Fig. 3
Unprocessed immunoblots.
Source Data Extended Data Fig. 4
Statistical source data.
Source Data Extended Data Fig. 4
Unprocessed immunoblots.
Source Data Extended Data Fig. 6
Statistical source data.
Source Data Extended Data Fig. 6
Unprocessed immunoblots.
Source Data Extended Data Fig. 7
Statistical source data.
Source Data Extended Data Fig. 7
Unprocessed immunoblots.
Source Data Extended Data Fig. 8
Statistical source data.
Source Data Extended Data Fig. 8
Unprocessed immunoblots.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pan, L., Li, C., Meng, L. et al. GDF1 ameliorates cognitive impairment induced by hearing loss. Nat Aging 4, 568–583 (2024). https://doi.org/10.1038/s43587-024-00592-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s43587-024-00592-5
- Springer Nature America, Inc.
This article is cited by
-
Hearing loss promotes Alzheimer’s disease
Nature Aging (2024)