Abstract
Hybridization has an important and often positive role in plant evolution. However, it can also have negative consequences for species. Two closely related species of Ornduffia are endemic to the Porongurup Range in the South West Australian Global Biodiversity Hotspot. The rare Ornduffia calthifolia is found exclusively on the summits, while O. marchantii is more widely dispersed across a greater range of elevation and is not considered threatened. Hybridisation in suitable overlapping habitat has been suspected between them for decades. Here we combine genotyping by sequencing to verify hybridisation genetically, and fine scale (2 m resolution) species distribution modelling (SDM) to test if hybrids occur in suitable intersecting habitat. From a study area of c. 4700 ha, SDM identified c. 275 ha and c. 322 ha of suitable habitat for O. calthifolia and O. marchantii, respectively. We identified range overlap between species of c. 59 ha), which enveloped 32 individuals confirmed to be hybrids. While the hybrids were at the margin of suitable habitat for O. marchantii, their preference for elevated habitat was closer to the more narrowly distributed O. calthifolia. The combination of genetic data and fine scale spatial modelling approaches enabled a better understanding of hybridisation among taxa of conservation significance. However, the level to which hybrid proliferation and competition for habitat presents as a threat to O. calthifolia is currently unknown and requires priority in conservation management given the threats from global warming and disturbance by tourism.
Similar content being viewed by others
Introduction
Hybridisation of two genetically distinguishable populations can have constructive or destructive outcomes for taxa1. For example, genetic and demographic swamping are two potential evolutionary processes that may cause extinction of one or both parents. Genetic swamping is the more frequent outcome, which occurs when hybrids replace one or both parents2,3. Demographic swamping results from outbreeding depression; where parent taxa expend reproductive energy on infertile or unfit hybrids, leading to population growth rates below what is needed for replacement4. Alternatively, hybridisation may increase diversity in previously isolated, inbred, populations (genetic rescue), increasing their viability5.
Narrow range endemic taxa are more vulnerable to extinction from the outcomes of hybridisation than common species4,6. Global biodiversity hotspots contain exceptional concentrations of endemism, including almost half of all global plant biodiversity, yet cover a fraction of the Earth’s surface7. These hotspots are therefore priorities for conservation management. For example, high levels of plant diversity and endemism occur in the South West Australian Global Biodiversity Hotspot, which includes over 2500 threatened vascular species8. This flora includes many groups of closely related taxa with narrow distributions that are geographically proximal in the relatively subdued landscapes of south-western Australia. For these taxa, understanding interactions through hybridisation have until recently been limited by available technology.
Recent advances in molecular biology (e.g. next generation sequencing) and fine scale species distribution modelling (SDM) means they can now be used together to confirm hybridisation and determine suitable habitat at unprecedented levels of detail9,10. Genotyping by sequencing can produce thousands of high-quality markers, enabling comprehensive genetic analyses even of non-model organisms11,12. Opportunities for hybridisation can be readily explored using SDM, by extrapolating the environmental correlates of the parent populations13 and delineating their overlapping niches. While there are multiple examples of SDM complementing genomic analyses (e.g.14,15), few have modelled endemic species at finer than 10 m resolution16,17.
Two closely related species of Ornduffia are endemic to a single mountain range in south-western Australia. Ornduffia calthifolia is declared rare flora under the Commonwealth Environment Protection and Biodiversity Conservation Act 199918 and is found exclusively on the summits of the Porongurup Range19. Ornduffia marchantii is found at lower elevations but is not considered threatened. Hybridisation potential has been confirmed in greenhouse experiments20 and is thought to be occurring in the intervening zone21. Keppel et al. (22) suggested that asymmetric hybridisation, with O. calthifolia as the maternal parent, could threaten the survival of O. calthifolia.
Here, we integrate genotyping by sequencing data and fine-scale SDM to confirm hybridisation between the two Ornduffia species in south-western Australia. We have three specific aims and associated hypotheses:
- (1)
verify the occurrence of hybrid individuals in the wild;
- (2)
estimate the pattern of hybridisation across species and populations; and
- (3)
examine whether hybridisation occurs in suitable overlapping habitat identified by distribution modelling. We hypothesised that the distribution of hybrid individuals would be spatially variable and occur more frequently in suitable overlapping habitat
Materials and Methods
Study area
The Porongurup Range (670 m a.s.l.), along with the Stirling Ranges (1090 m a.s.l.), provide the only significant altitudinal relief within south-western Australia23. The Porongurup Range (central point: 34°40′S, 117°52′E) is Australia’s largest granite outcrop22 consisting of multiple granite domes about 1100 million years old24. It varies in elevation from c. 230–670 m over 26 km2 and has a Mediterranean climate, with cool, wet winters and hot, dry summers. Soils at the base of this range may remain moist all year. Higher elevations receive greater orographic rainfall than the base25, though no official measurements have been produced26.
Study species
The herbaceous genus Ornduffia (Family: Menyanthaceae) includes eight semi-aquatic and wetland species with fleshy leaves, native to southern Australia22. Ornduffia calthifolia (Fig. 1A) is around 75 cm tall when in flower and has considerably larger leaves and habit, relative to Ornduffia marchantii (Fig. 1B)., which has smooth, heart-shaped leaves (Fig. 1B). Ornduffia marchantii is strongly distylous, making individuals self-incompatible20. This differs from O. calthifolia, which is self-compatible27, allowing it to produce copious fertile seed without reliance on pollinating insects20. Both species produce small (<2 mm) elliptic seeds that demonstrate hydrophobic properties, assisting dispersal by water28. Additionally, ants are thought to be an important dispersal agent for both species28.
Ornduffia species and habitat within the Porongurup Range (non-flowering season). (A) O. calthifolia, with large fleshy leaves and distinct crenulations, found in shallow soils amongst bare rock. (B) O. marchantii, with small, smooth heart shaped leaves, found in leaf litter of forest floor. (C) Putative hybrid with intermediate morphological characteristics of both parent species, along the Devils Slide walking track. (D) Typical habitat of O. calthifolia, in protected crevasses between rock formations at high elevations (>550 m). (E) Typical habitat of O. marchantii, in karri (Eucalyptus diversicolor) forest, the dominate vegetation type at lower elevation of the Porongurup Range.
Putative hybrids (Fig. 1C) have only been identified along a long-established walking track at Devil’s Slide (Fig. 2)21,22. The two parent species have different habitat preferences; O. calthifolia occurs mostly within moist, sheltered crevices at the highest-elevation (e.g. 640 m asl) granite peaks (Fig. 1D18,20), whilst O. marchantii occurs at lower elevations (e.g. 350 m asl), often in disturbed habitat, under forest dominated by karri (Eucalyptus diversicolor) where soils are wet and loamy25,29(Fig. 1E20).
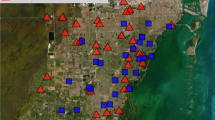
Sampling locations for O. calthifolia, O. marchantii and transect populations of putative hybrids across the Porongurup Range National Park. OC = parent populations of O. calthifolia, OM = parent populations of O. marchantii. Imagery was taken in 2011 by airplane flying 1700–2200 m above the ground using a Digital Mapping Camera from Z/I Imaging and acquiring RGB spectral bands covering 425–515, 515–550 and 600–650 nm at a spatial resolution of 0.2 m. ArcGIS v 10.5 was used to create the map.
Sample collection
We conducted a field survey in the Porongurup Range in March 2017. Three reference populations for each of O. calthifolia (‘OC Nancy Peak’, ‘OC Devils Slide’, ‘OC Castle Rock’) and O. marchantii (‘OM Tree in Rock’, ‘OM Mira Flores’, ‘OM East’) were sampled (Fig. 2), and reference populations were located across the species ranges. We performed transect sampling for putative hybrids, starting at low elevations, scaling the mountains, and collecting populations when found. Transect one was sampled adjacent to a recreational walking track to Devil’s Slide and Transect two in undisturbed habitat leading to the summit of Nancy Peak (Fig. 2).
Transect one comprised five populations (T1, 1–5) of putative hybrids (Fig. 2) between 580–630 m asl. Transect one sampling was limited to within 2 m of the walking track. We ran Transect two on the south side of Nancy Peak (Fig. 2) in forest seldom traversed. This transect comprised three populations between 360 and 560 m (T2, 1–3), with two additional individuals at 600 m comprising T2, 4.
We collected 187 samples, each population comprising 13 or 14 samples except for T2,4, which only had two individuals. The largest healthy leaf from each sample was collected for morphometric analysis, placed in an airtight bag and refrigerated in the field. Where there were ample individuals, samples were a minimum of 5 m apart. We recorded the coordinates of each sample with the aid of a GNSS receiver (Garmin Etrex 10).
Morphometric analysis
Morphometric analysis was used to identify phenological intermediates. Ornduffia marchantii is identified as having a smaller narrower leaf and shorter stem than O. calthifolia20. Three leaf dimensions (length along midrib from base to tip, maximum width perpendicular to midrib and maximum length from tip to lower lobe), and petiole length were measured on the largest healthy leaf of each plant. Sampling occurred out of flowering season and thus no reproductive variables could be obtained. We produced 2-dimensional multidimensional scaling (MDS) ordination plots in PATN V2.329 to visually assess trends in the resultant patterns. The goal of MDS is to faithfully represent the distance matrix in the lowest possible dimensional space, with level of stress measuring the resultant distortion. If stress is low, the chosen dimensional representation (in this case two) reasonably represents the objects relative positions. We used the Gower association measure30 to derive the distance matrix, due to its capacity to handle continuous data29, with 1000 random starts, a maximum 1000 iterations and a cut value of 0.938.
We ran analysis of similarities (ANOSIM) and permutational multivariate analysis of variance (PERMANOVA) to test for significant morphological differences between these a priori groups in PAST3 software31. These functions operate directly on the dissimilarity matrix and are allied with MDS ordination in that it uses the rank order of dissimilarity values. If two groups of sampling units are different, then compositional dissimilarities between the groups ought to be greater than those within groups. The ANOSIM statistic R is based on the difference of mean ranks between groups (r_B) and within groups. In both cases the Gower dissimilarity measure and 10 000 permutations were applied.
Genetic analysis
Diversity Arrays Technology Pty. Ltd extracted DNA from 0.1 g dried leaf samples with the NucleoSpin Plant II method (Macherey-Nagel). Genome wide scans by Diversity Arrays Technology (DArTSeq) sequenced the isolated DNA fragments. DARTSeq is a genome complexity reduction method combined with next generation sequencing technologies, capable of identifying tens of thousands of single nucleotide polymorphism (SNP) loci. To ensure only high quality and informative markers were included, loci with more than 10% missing data (call rate <0.9) were removed from analysis32 as were loci with a minor allele frequency <0.05 (e.g.33) or average read depth <5 (e.g.34). Loci displaying departure from Hardy-Weinberg equilibrium and linkage equilibrium were removed with the dartR package in R35.
We used BAYESCAN V2.136 to identify outlier loci, whose allele frequencies vary more between populations than expected under genetic drift. These loci may represent putatively non-neutral genetic regions and were removed from the analysis. We applied default parameters for iterations (20 pilot runs of 5000 iterations each, 100 000 total iterations with 50 000 burn-in). We set ‘Prior odds for neutral model’ to 200, as higher odds are required for larger datasets37. The inbreeding coefficient was set as ‘FIS uniform between 0 and 0.5’ (default is uniform between 0 and 1) based on FIS results calculated for each locus using the R package ADEGENET38. We applied a false discovery rate (FDR) threshold of 0.05 (q value < 0.05) to separate neutral and adaptive loci36.
Expected heterozygosity (HE), observed heterozygosity (HO) and allelic richness (AR) values were produced in the R package HIERFSTAT39. Inbreeding coefficients (FIS) were calculated for each locus using R packages ADEGENET38. We grouped all samples into four populations for calculating genetic diversity; the two parent reference populations, Transect 1 and Transect 2.
We generated a pairwise Nei’s genetic distance40 matrix in the R package STAMPP41. From the distance matrix, we generated a PCoA ordination graph in GENALEX 6.542 to visualise population structure and similarity between samples43. We also used Bayesian clustering methods in STRUCTURE V2.3.444,45 to produce definitive identification of hybrid individuals. STRUCTURE implements a Markov chain Monte Carlo (MCMC) algorithm, calculating percentage admixture individuals to K genetic clusters. We tested for optimal number of clusters with 10 runs each of K = 1–6. We ran a burn-in of 50 000 iterations and 250 000 MCMC iterations, no previous population information and correlated allele frequencies (e.g.46). A subsequent analysis in STRUCTURE was performed with USEPOPINFO = 1 to allocate putative hybrid individuals to the parental populations. We used the ΔK method47 in STRUCTURE HARVESTER V0.6.9348 to delineate optimal K. Individual replicates were aligned using the ‘full search’ algorithm in CLUMPP 1.1.249. The optimal K value was then used in the delineation of hybrid individuals. Pure species were delineated by a threshold at 0.1, meaning individuals assigned a value > 0.90 to a species were categorised as pure (e.g.46).
To identify hybrid status of individuals as back-crossed or F1 hybrids, we ran analyses on NEW-HYBRIDS50 to assign individuals to one of six genealogical classes (pure OC, pure OM, F1 hybrid, F2 hybrid, back-crossed OC and back-crossed OM) (e.g.46). We validated the accuracy at which both STRUCTURE and NEW-HYBRIDS correctly classified individuals using simulated populations created in HYBRIDLAB51. From each parent population we created 200 individuals, of which we simulated the creation of the other four genealogical classes identified by NEW-HYBRIDS. We then ran simulated samples through STRUCTURE and NEW HYBRIDS with the same parameters as the real data.
Spatial patterns of hybridisation
We developed SDMs with MAXENT V3.3.313 for both parent species to predict opportunities for hybridisation in the overlapping suitable habitat. All ‘pure’ individuals (89 O. marchantii and 59 O. calthifolia samples), as delineated by STRUCTURE, were included as presence data. We supplemented this survey with 207 O. calthifolia and 55 O. marchantii samples from surveys conducted in February 2012 and November 2013. As no coordinates were duplicated over surveys, we assumed all samples were unique. To quantify model accuracy, we calculated mean area under the curve (AUC) of receiver operating characteristic (ROC) curves52 with 100 bootstrap iterations. Each iteration randomly selected 20% of the points for an independent validation dataset. We did not develop an SDM for the hybrid because its current limited geographic extent comprises of only a few clustered samples and is therefore likely to result in under prediction.
Light detection and ranging (LiDAR) data for the Porongurup Range was captured in April 2011 with an airborne Leica ALS 50-II scanner. A flight height between 1700 and 2220 m resulted in c. 0.63 last return heights per m2, which were interpolated into a 2 m resolution digital elevation model (DEM). Horizontal and vertical accuracies are <0.35 m and <0.15 m respectively. Elevation, slope (first derivative of elevation), aspect (degrees from north), curvature (second derivative of elevation), solar radiation (WH/m2) at monthly intervals and the topographic wetness index (TWI) defined by Gessler et al.53 were derived from the DEM using the Spatial Analyst Toolbox of ArcGIS v 10.554.
We calculated topographic roughness index (TRI), vertical distance to channel network (VDCN), topographic position index (TPI) and the SAGA Wetness Index (SWI) in SAGA v 2.1.455. TRI56 is a measure of terrain heterogeneity, VDCN is a measure of local ridge heights. TPI57 identifies elevation relative to its neighbourhood. The SWI was used in addition to TWI because they were not significantly correlated (r = 0.4) and the SWI has predicted potential of soil moisture for cells with small vertical distance to a channel in valley floors more realistically than TWI58. As we recognised that O. calthifolia preferentially inhabits locations at or near the base of cliff faces we used a high pass filter to identify cliff edges and modelled for proximity. As O. marchantii is associated with the shade provided by karri over storey, we calculated a canopy height model (CHM).
There is no accepted method for selecting model thresholds other than they should be data driven59. We defined a threshold to classify suitable and unsuitable habitat by minimising misclassification of pseudo-absences created nearby traversed locations60.
Results
Morphometric analysis
As expected, analysis of morphometric data using MDS showed separation of individuals from the two parent species classes with little overlap in ordination plot space (Fig. 3). These two species have long been recognised as separate based on morphology and habitat20. Samples from Transect 1 occupied broad ordination space with most individuals occurring in similar ordination space to that of O. calthifolia, yet also occurring with O. marchantii and in the space between the two species. Individuals from Transect 2 occupied ordination space between that of the two species, with some overlap with O. marchantii. Each class was significantly different (ANOSIM and PERMANOVA, P < 0.01) from every other class except for O. calthifolia with Transect 1. ANOSIM produced an overall R value of 0.2045 and PERMANOVA an F statistic of 39.76. Pairwise R values can be found in Fig. 3.
Two-dimensional ordination (Semi-strong hybrid multidimensional scaling SSH MDS, Stress = 0.129) of 187 individuals were classified into four a priori groups of O. calthifolia, O. marchantii and the two transects. R values from ANOSIM are displayed with O. calthifolia and Transect 1 the only populations not significantly dissimilar.
Genetic analyses
DArT sequencing produced 48 541 SNP loci (not including presence/absence data), with 8 489 loci retained after data cleaning. BAYESCAN identified 263 outlier loci that were removed from the analysis. The more common and dispersed taxon, O. marchantii, had greater genetic diversity than O. calthifolia (HE = 0.235 v. 0.083; Table 1). Further, O. calthifolia demonstrated departure from random mating with a much higher inbreeding coefficient than O. marchantii (0.651 v 0.268; Table 1). Genetic diversity estimates for samples from Transect 1 exhibited highest genetic diversity and allelic richness (Table 1). Genetic diversity estimates for Transect 2 were consistent with those for O. marchantii, except for a lower inbreeding coefficient (Table 1).
There was greater differentiation between populations based on genetic distance analysis than morphometric analysis (Fig. 4). The PCoA showed O. calthifolia populations clustering separately from those of O. marchantii on the axis for coordinate 1. There was some variation within both species that was predominantly identified by coordinate 2, with the more isolated eastern populations (‘OC Castle Rock’ and ‘OM East’) exhibiting greatest separation from other populations within each species. Thirty-six individuals from Transect 1 were clustered with O. calthifolia from the Devil’s slide population but the majority occurred in intermediate space between the two species (Fig. 4). All samples from Transect 2 clustered with samples from O. marchantii, except for one that was between the two species (Fig. 4).
The ΔK method identified K = 2 to be optimal. Variation between replicate runs on STRUCTURE did not result in any changes to species assignment when K = 2. Admixture modelling in STRUCTURE detected hybridisation between O. calthifolia and O. marchantii (Fig. 5) and individuals identified as hybrids based on morphology. Hybridisation was more frequent on Transect 1 with 31 hybrid individuals identified, with the remaining individuals identified as pure individuals of both parent species, except for a single hybrid found on Transect 2. All other individuals on Transect 2 were assigned to O. marchantii. Ninety-nine per cent of private alleles (alleles found only in one species) were shared with hybrids.
Population genetic structure and its spatial variation for two species of Ornduffia (O. calthifolia and O. marchantii) and their hybrids in the Porongurup Range, south-western Australia. (A) Population genetic structure with genetic assignment of individuals to either O. calthifolia or to O. marchantii designated from Bayesian techniques implemented in STRUCTURE. Blue is likelihood of assignment to O. calthifolia, orange is assignment to O. marchantii. Populations are O. marchantii East (OME), Mira Flores (OMM) and Tree in Rock (OMT), O. calthifolia Castle Rock (OCC), Devil’s Slide (OCD) and Nancy Peak (OCN), Transect 1 with populations 1–4 (T1–5) and Transect 2 with populations 1–4 (T1–5). (B) Disturbed and undisturbed transect sampling locations with admixture among samples, as calculated by STRUCTURE, overlayed on an aerial photograph. Contour interval = 10 m. Imagery was taken in 2011 by airplane flying 1700–2200 m above the ground using a Digital Mapping Camera from Z/I Imaging and acquiring RGB spectral bands covering 425–515, 515–550 and 600–650 nm at a spatial resolution of 0.2 m. ArcGIS v 10.5 was used to create the map.
All samples from identified O. calthifolia populations were above the threshold of 0.9 and classified as pure. Most of the samples from populations of O. marchantii were also identified as pure, except for three samples of O. marchantii from the ‘OM East’ population, which were below the 0.9 threshold. We sampled this population at two slightly disparate locations (Fig. 2), with all seven samples from one location assigned a membership of approximately 0.9 to O. marchantii. Morphometric analysis suggested these samples were consistent as being from O. marchantii and these samples were considered as pure O. marchantii for SDMs.
Samples from Transect 1 showed a range of genetic affinities with some individuals assigned to each of the two parent species and the remainder showing a range of admixture. Samples assigned to either of the two parent species were located primarily at lower elevation, with assignment to O. calthifolia tending to increase with elevation along Transect 1 (Fig. 5). Further, each population along Transect 1 consisted of at least one pure O. calthifolia. The T1, 2 population was comprised of pure O. calthifolia, and occurred near a large cliff, which is habitat suitable only for O. calthifolia (as identified by SDM).
Results from NEW-HYBRIDS closely matched those from STRUCTURE. F1 and F2 Hybrids were more prevalent at higher altitudes along Transect 1. Across the five populations of increasing elevation, the number of F1 and F2 hybrids were 0, 0, 2, 7 and 7. Perfect classification of simulated individuals from HYBRIDLAB was achieved in both STRUCTURE and NEW-HYBRIDS with 100% of the simulated individuals correctly classified.
Spatial patterns of hybridisation
Average area under the curve (AUC) statistics across 100 bootstrap runs based on a 20% subset for validation were 0.98 and 0.92 for O. calthifolia and O. marchantii, respectively. With elevation as the highest contributor (Table 2), distribution models of O. calthifolia suggest limitation to the summits or peaks, with more than 90% of the 275 ha of suitable area being above 500 m (Fig. 6).
Species distribution model (SDM) identifying suitable habitat for two species of Ornduffia (O. calthifolia and O. marchantii) in the Porongurup Range, south-western Australia. Potential for natural hybridisation exists in the area of overlap (magenta). Threshold of suitability threshold at value producing highest overall accuracy. Area of overlap = 0.11 km2. Base imagery is from a Leica ALS 50-II scanner flown at 1700–2200 m, in 2011, and scanning approximately 1.5–2 km wide swaths. Triangulation was used to interpolate to a resolution of 2 m. ArcGIS v 10.5 was used to create the map.
Analysis of explanatory variables showed elevation had less impact on the distribution of O. marchantii, and suitable areas of habitat can be found across the extent of the range. The extent of suitable habitat for O. marchantii (322 ha) was greater and far more widespread and fragmented than that of O. calthifolia (Fig. 6). The area of estimated suitable habitat intersecting both species was limited to approximately 59 ha, near mountain summits due to the elevation dependency of O. calthifolia. All hybrid individuals existed or were contained within the area apparently suitable for both species.
Discussion
This study provides an exemplar of combining genetic data and spatial modelling approaches to enable more nuanced understanding of hybridisation among taxa of conservation significance. Genetic analysis clearly demonstrated presence of individuals from both species and individuals with a range of admixture. SDM identified the differences in suitable habitat, as well as an area of overlap suitable for both parents that is concurrent with the location of hybrids detected in genetic analyses.
Genetic and morphological evidence of hybridisation
Hybridisation between O. calthifolia and O. marchantii was confirmed by combining morphological and genotyping approaches, supporting previous claims of putative hybrids25,28. Morphometric analysis identified phenotypic intermediates between the two species. However, high overlap with parental morphology made it difficult to definitively identify hybrid individuals and pure individuals along Transect 1. Nevertheless, admixture modelling in STRUCTURE based on genotyping conclusively identified 32 hybrid individuals, along with pure individuals of the two species.
An area of 59 ha suitable for both species was derived by intersecting SDMs of the parents to highlight the potential for co-occurrence and natural hybridization. All the Transect 1 sites and Transect 2, 3–4 were located within this overlap area. Therefore, the greater rate of hybridization may be due to greater overlap of the parent species in transect 1. However, given transect 1 is adjacent to a walking track it is possible that disturbance is opening new corridors for species contact and altering plant phenology, but this theory needs further exploration60,61. This is consistent with studies that show disturbance can affect the phenology of plants to induce the hybridisation (e.g.61), as opposed to merely bringing the taxa into contact (e.g.62). Plastic responses to habitat change (e.g. flowering time), are known to vary under conditions of anthropogenic disturbance63,64. Ornduff20 noted that there is some natural overlap in flowering and pollination times and was able to produce hybrids under laboratory conditions.
Whilst hybrid individuals were largely restricted to Transect 1, which is located within relatively disturbed habitat, our sampling was not designed to determine whether disturbance drives hybridization. However, given that the study site is a prominent tourist destination, future studies should investigate the role of disturbance in hybridization. Such studies could be complicated by a marked drop in the abundance of O. calthifolia since a 2013 study22. The rarity of this species therefore makes rigid sampling along elevational transect difficult. Limitations in study design mean we cannot rule out subtle differences in microhabitat between the two transects or that Transect 1 is located on a historic, geographically stable hybrid zone.
Spatially variable introgression
We found evidence of introgression and backcrossing, as hybrids exhibited a range of affinity to each species. Without introgression, all hybrids would be expected to be F1 hybrids, and would display approximately 50% affinity to each parent65. Affinity to O. calthifolia increased with elevation in Transect 1 from the lowest population (median admixture = 0.07 O. calthifolia, 0.93 O. marchantii), to the highest (median admixture = 0.59 O. calthifolia, 0.41 O. marchantii). This change in admixture is consistent with the bounded hybrid superiority model66, whereby a zone of hybridisation occurs between two geographically separated species, creating a gradient of admixture. In this model, hybrids possess superior adaptability to the intermediate conditions, but parent species dominate in their niche habitats66.
Numerous ‘pure’ individuals persisted within the hybrid zone of Transect 1, particularly in population 2, which contained almost exclusively ‘pure’ O. calthifolia individuals. Persistence of O. calthifolia amongst hybrids in this population is noteworthy considering the proximity of hybrids. The site was adjacent to a sheer rock face (Fig. 5), a key habitat property of O. calthifolia22. However, such formations were also present at populations comprised of hybrid individuals. SDM analysis showed that habitat in this area was no more suitable than other locations along the transect suggesting the influence of other factors such as soil depth.
Implications of hybridisation
There are several potential positive and negative implications of hybridisation to the parent populations, but all are uncertain. For example, hybridisation may provide O. calthifolia with new genetic variation, alleviating inbreeding or facilitating response to changing abiotic conditions67,68,69. Alternatively, hybridisation may lead to genetic swamping of O. calthifolia2,3. In this study, hybrids occurred at elevations spatially associated with the restricted O. calthifolia, and as we found evidence for introgression, this species could potentially be at risk of genetic swamping2,70. Early generation hybrids identified at higher altitudes suggest invasion of O. marchantii into the higher elevation habitat of O. calthifolia. The expansion of O. marchantii into these higher elevations fits the climate induced altitudinal migration of species, which can result in swamping1,3,71. However, putative hybridisation has been suspected at this location for decades18,20, yet little progression in extent has been recorded. Laboratory tests found pollen viability of hybrids was approximately 50%, compared to >90% for pure specimens20. Therefore, it is possible that reduced fertility of hybrid individuals could slow or negate any swamping process. Moreover, the presence of pure O. calthifolia at population 2 on Transect 2 population demonstrates its ability to persist in niche habitat amongst hybrid populations.
The combination of genetic data and spatial modelling approaches enabled a better understanding of hybridisation among taxa of conservation significance. Although the hybrids were at the margin of suitable habitat for O. marchantii, their preferred habitat was closer to that of the more narrowly distributed O. calthifolia. The extent to which hybrid spread and competition for habitat presents as a threat to O. calthifolia is unknown, as is the role of disturbance in hybridisation. However, given the restricted distribution of O. calthifolia (endemic to high elevations in the Porongorups) and the threats posed by global warming and disturbance through tourism, understanding the dynamics of hybridisation should be a priority in monitoring and conservation management.
Data availability
All genotype data will be available from the Dryad data repository.
References
Todesco, M. et al. Hybridization and extinction. Evol. Appl. 9, 892–908, https://doi.org/10.1111/eva.12367 (2016).
Rhymer, J. M. & Simberloff, D. Extinction by hybridization and introgression. Annu. Rev. Ecol. Syst. 27, 83–109, https://doi.org/10.1146/annurev.ecolsys.27.1.83 (1996).
Muhlfeld, C. C. et al. Invasive hybridization in a threatened species is accelerated by climate change. Nat. Clim. Change 4, 620–624, https://doi.org/10.1038/nclimate2252 (2014).
Wolf, D. E., Takebayashi, N. & Rieseberg, L. H. Predicting the risk of extinction through hybridization. Conserv. Biol. 15, 1039–1053, https://doi.org/10.1046/j.1523-1739.2001.0150041039.x (2001).
Whiteley, A. R., Fitzpatrick, S. W., Funk, W. C. & Tallmon, D. A. Genetic rescue to the rescue. Trends Ecol. Evol. 30, 42–49, https://doi.org/10.1016/j.tree.2014.10.009 (2015).
Prentis, P. J., White, E. M., Radford, I. J., Lowe, A. J. & Clarke, A. R. Can hybridization cause local extinction: a case for demographic swamping of the Australian native Senecio pinnatifolius by the invasive Senecio madagascariensis? N. Phytol. 176, 902–912, https://doi.org/10.1111/j.1469-8137.2007.02217.x (2007).
Myers, N., Mittermeier, R., Mittermeier, C. G., Da Fonseca, G. A. & Kent, J. Biodiversity hotspots for conservation priorities. Nat. 403, 853–858, https://doi.org/10.1038/35002501 (2000).
Hopper, S. D. & Gioia, P. The southwest Australian floristic region: evolution and conservation of a global hot spot of biodiversity. Annu. Rev. Ecol. Syst. 35, 623–650, https://doi.org/10.1146/annurev.ecolsys.35.112202.130201 (2004).
Robinson, T. P., Di Virgilio, G., Temple-Smith, D., Hesford, J. & Wardell-Johnson, G. W. Characterisation of range restriction amongst the rare flora of Banded Ironstone Formation ranges in semiarid south-western Australia. Aust. J. Bot. 67, 234–247, https://doi.org/10.1071/BT18111 (2018).
Goulet, B. E., Roda, F. & Hopkins, R. Hybridization in plants: old ideas, new techniques. Plant. Physiol. 173, 65–78, https://doi.org/10.1104/pp.16.01340 (2017).
Allendorf, F. W., Hohenlohe, P. A. & Luikart, G. Genomics and the future of conservation genetics. Nat. Rev. Genet. 11, 697–709, https://doi.org/10.1038/nrg2844 (2010).
Sansaloni, C. P. et al. A high-density Diversity Arrays Technology (DArT) microarray for genome-wide genotyping in Eucalyptus. Plant. Methods 6, 16, https://doi.org/10.1186/1746-4811-6-16 (2010).
Phillips, S. J., Anderson, R. P. & Schapire, R. E. Maximum entropy modelling of species geographic distributions. Ecol. Model. 190, 231–259, https://doi.org/10.1016/j.ecolmodel.2005.03.026. (2006).
Tarroso, P., Pereira, R. J., Martínez-Freiría, F., Godinho, R. & Brito, J. C. Hybridization at an ecotone: ecological and genetic barriers between three Iberian vipers. Mol. Ecol. 23, 1108–1123, https://doi.org/10.1111/mec.12671 (2014).
Hudson, C. J., Freeman, J. S., Myburg, A. A., Potts, B. M. & Vaillancourt, R. E. Genomic patterns of species diversity and divergence in Eucalyptus. N. Phytol. 206, 1378–1390, https://doi.org/10.1111/nph.13316 (2015).
Riordan, E. C. et al. Association of genetic and phenotypic variability with geography and climate in three southern California oaks. Am. J. Bot. 103, 73–85, https://doi.org/10.3732/ajb.1500135 (2016).
Nezer, O., Bar-David, S., Gueta, T. & Carmel, Y. High-resolution species-distribution model based on systematic sampling and indirect observations. Biodivers. Conserv. 26, 421–437, https://doi.org/10.1007/s10531-016-1251-2 (2017).
Gilfillan, S. & Barrett, S. Mountain Villarsia (Villarsia calthifolia) Interim recovery plan 2004–2009. Department of Conservation and Land Management, Albany WA. Retrieved from, http://www.environment.gov.au/system/files/resources/9188b047-2805-4c42-a00b-22c033155095/files/v-calthifolia.pdf (2004).
Brown, A., Thompson-Dans, C. & Marchant, N. (eds). Western Australia’s threatened flora. Perth, Western Australia: Department of Conservation and Land Management (1998).
Ornduff, R. A new species of Villarsia (Menyanthaceae) from the Porongurup Range, Western Australia. Sys Bot. 15, 216–220, https://doi.org/10.2307/2419175 (1990).
Robinson, C. J. & Coates, D. J. Declared rare and poorly known plants in the Albany District. Wildlife Management Program No. 20. Perth, Western Australia: Department of Conservation and Land Management (1995).
Keppel, G. et al. A low-elevation mountain range as an important refugium for two narrow endemics in the Southwest Australian Floristic Region biodiversity hotspot. Ann. Bot-London 119, 289–300, https://doi.org/10.1093/aob/mcw182 (2017).
Barrett, S. & Yates, C. J. Risks to a mountain summit ecosystem with endemic biota in southwestern Australia. Austral Ecol. 40, 423–432, https://doi.org/10.1111/aec.12199 (2015).
Abbott, I. The vascular flora of the Porongurup Range south-western Australia. West. Australian Herbarium Res. Notes 7, 1–16 (1982).
Tippery, N. P. & Les, D. H. A new genus and new combinations in Australian Villarsia (Menyanthaceae). Novon 19, 404–411, https://doi.org/10.3417/2007181 (2009).
Wills, A. & Abbott, I. Landscape-scale species richness of earthworms in the Porongurup Range, Western Australia: influence of aspect, soil fertility, and vegetation type. Biol. Fert. Soils 39, 94–102, https://doi.org/10.1007/s00374-003-0683-5 (2003).
Ornduff, R. Comparative fecundity and population composition of heterostylous and non-heterostylous species of Villarsia (Menyanthaceae) in Western Australia. Am. J. Bot. 73, 282–286, https://doi.org/10.2307/2444182 (1986).
Chuang, T. I. & Ornduff, R. Seed morphology and systematics of Menyanthaceae. Am. J. Bot. 12, 1396–1406, https://doi.org/10.1002/j.1537-2197.1992.tb13750.x (1992).
Belbin, L. PATN: pattern analysis package: Technical reference. Division of Wildlife and Ecology. CSIRO (1994).
Gower, J. C. A general coefficient of similarity and some of its properties. Biometrics 27, 857–871, https://doi.org/10.2307/2528823 (1971).
Hammer, Ø., Harper, D. A. & Ryan, P. D. PAST: paleontological statistics software package for education and data analysis. Palaeontol. electron. 22, 1–9 (2001).
Steane, D. A. et al. Genome‐wide scans detect adaptation to aridity in a widespread forest tree species. Mol. Ecol. 23, 2500–2513, https://doi.org/10.1111/mec.12751 (2014).
Jordan, R., Dillon, S. K., Prober, S. M. & Hoffmann, A. A. Landscape genomics reveals altered genome wide diversity within revegetated stands of Eucalyptus microcarpa (Grey Box). N. Phytol. 212, 992–1006, https://doi.org/10.1111/nph.14084 (2016).
Pazmiño, D. A., Maes, G. E., Simpfendorfer, C. A., Salinas-de-León, P. & van Herwerden, L. Genome-wide SNPs reveal low effective population size within confined management units of the highly vagile Galapagos shark (Carcharhinus galapagensis). Conserv. Genet. 5, 1151–1163, https://doi.org/10.1007/s10592-017-0967-1 (2017).
Gruber, B., Unmack, P. J., Berry, O. F. & Georges, A. dartr: An r package to facilitate analysis of SNP data generated from reduced representation genome sequencing. Mol. Ecol. Resour. 18, 691–699, https://doi.org/10.1111/1755-0998.12745 (2018).
Foll, M. & Gaggiotti, O. A genome-scan method to identify selected loci appropriate for both dominant and codominant markers: a Bayesian perspective. Genet. 180, 977–993, https://doi.org/10.1534/genetics.108.092221 (2008).
Steane, D. A. et al. Genome-wide scans reveal cryptic population structure in a dry-adapted eucalypt. Tree Genet. Genomes 11, 33, https://doi.org/10.1007/s11295-015-0864-z (2015).
Jombart, T. adegenet: a R package for the multivariate analysis of genetic markers. Bioinforma. 24, 1403–1405, https://doi.org/10.1093/bioinformatics/btn129 (2008).
Goudet, J. Hierfstat, a package for R to compute and test hierarchical F‐statistics. Mol. Ecol. Resour. 5, 184–186, https://doi.org/10.1111/j.1471-8286.2004.00828.x (2005).
Nei, M. Genetic distance between populations. Am. Nat. 106, 283–292, https://doi.org/10.1086/282771 (1972).
Pembleton, L. W., Cogan, N. O. & Forster, J. W. StAMPP: an R package for calculation of genetic differentiation and structure of mixed‐ploidy level populations. Mol. Ecol. Resour. 13, 946–952, https://doi.org/10.1111/1755-0998.12129 (2013).
Peakall, R. & Smouse, P. E. GENALEX 6: genetic analysis in Excel. Population genetic software for teaching and research. Mol. Ecol. Resour. 6, 288–295, https://doi.org/10.1093/bioinformatics/bts460 (2006).
Payseur, B. A. & Rieseberg, L. H. A genomic perspective on hybridization and speciation. Mol. Ecol. 25, 2337–2360, https://doi.org/10.1111/mec.13557 (2016).
Pritchard, J. K., Stephens, M. & Donnelly, P. Inference of population structure using multilocus genotype data. Genet. 155, 945–959 (2000).
Falush, D., Stephens, M. & Pritchard, J. K. Inference of population structure using multi-locus genotype data: linked loci and correlated allele frequencies. Genet. 164, 1567–1587 (2003).
Field, D. L., Ayre, D. J., Whelan, R. J. & Young, A. G. Patterns of hybridization and asymmetrical gene flow in hybrid zones of the rare Eucalyptus aggregata and common E. rubida. Heredity 106, 841–853, https://doi.org/10.1038/hdy.2010.127 (2011).
Evanno, G., Regnaut, S. & Goudet, J. Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol. Ecol. 14, 2611–2620, https://doi.org/10.1111/j.1365-294X.2005.02553.x (2005).
Earl, D. A. & vonHoldt, B. M. STRUCTURE HARVESTER: a website and program for visualizing STRUCTURE output and implementing the Evanno method. Conserv. Genet. Resour. 4, 359–361, https://doi.org/10.1007/s12686-011-9548-7 (2012).
Jakobsson, M. & Rosenberg, N. A. CLUMPP: a cluster matching and permutation program for dealing with label switching and multimodality in analysis of population structure. Bioinforma. 23, 1801–1806, https://doi.org/10.1093/bioinformatics/btm233 (2007).
Anderson, E. C. & Thompson, E. A. A model-based method for identifying species hybrids using multi-locus genetic data. Genet. 160, 1217–1229 (2002).
Nielsen, E. E., Bach, L. A. & Kotlicki, P. HYBRIDLAB (version 1.0): a program for generating simulated hybrids from population samples. Mol. Ecol. Notes 6, 971–973, https://doi.org/10.1111/j.1471-8286.2006.01433.x (2006).
Fielding, A. H. & Bell, J. F. A review of methods for the assessment of prediction errors in conservation presence/absence models. Env. Conserv. 24, 38–49, https://doi.org/10.1017/S0016774600023726 (1997).
Gessler, P. E., Moore, I. D., McKenzie, N. J. & Ryan, P. J. Soil-landscape modelling and spatial prediction of soil attributes. Int. J. Geogr. Inf. Syst. 9, 421–432, https://doi.org/10.1080/02693799508902047 (1995).
ESRI. ArcGIS Desktop: Release 10.4. Redlands, CA: Environmental Systems Research Institute (2015).
Conrad, O. et al. System for Automated Geoscientific Analyses (SAGA) v. 2.1.4, Geoscience Model Development 8, 1991–2007, https://doi.org/10.5194/gmd-8-1991-2015 (2015).
Riley, S. J. Index that quantifies topographic heterogeneity. Intermt. J. Sci. 5, 23–27 (1999).
Guisan, A., Weiss, S. B. & Weiss, A. D. GLM versus CCA spatial modelling of plant species distribution. Plant. Ecol. 143, 107–122, https://doi.org/10.1023/A:1009841519580 (1999).
Boehner, J. et al. Soil regionalisation by means of terrain analysis and process parameterisation. In ‘Soil classification 2001. European Soil Bureau, Research Report No. 7’. (eds. Micheli, E., Nachtergaele, F. & Montanarella, L.) pp. 213–222. (European Soil Bureau: Luxembourg) (2002).
Worth, J. R. P., Harrison, P. A., Williamson, G. J. & Jordan, G. J. Whole range and regional-based ecological niche models predict differing exposure to 21st century climate change in the key cool temperate rainforest tree southern beech (Nothofagus cunninghamii). Austral Ecol. 40, 126–138, https://doi.org/10.1111/aec.12184 (2015).
Engler, R., Guisan, A. & Rechsteiner, L. An improved approach for predicting the distribution of rare and endangered species from occurrence and pseudo‐absence data. J. Appl. Ecol. 41, 263–274, https://doi.org/10.1111/j.0021-8901.2004.00881.x (2004).
Lamont, B. B., He, T., Enright, N. J., Krauss, S. L. & Miller, B. P. Anthropogenic disturbance promotes hybridization between Banksia species by altering their biology. J. Evolution Biol. 16, 551–557, https://doi.org/10.1046/j.1420-9101.2003.00548.x (2003).
Parsons, K. & Hermanutz, L. Conservation of rare, endemic braya species (Brassicaceae): Breeding system variation, potential hybridization and human disturbance. Biol. Conserv. 128, 201–214, https://doi.org/10.1016/j.biocon.2005.09.029 (2006).
Neil, K. & Wu, J. Effects of urbanization on plant flowering phenology: a review. Urban. Ecosyst. 9, 243–257, https://doi.org/10.1007/s11252-006-9354-2 (2006).
Levin, D. A. Flowering‐time plasticity facilitates niche shifts in adjacent populations. N. Phytol. 183, 661–666, https://doi.org/10.1111/j.1469-8137.2009.02889.x (2009).
Xu, S., Tauer, C. G. & Nelson, C. D. Natural hybridization within seed sources of shortleaf pine (Pinus echinata Mill.) and loblolly pine (Pinus taeda L.). Tree Genet. Genomes 4, 849–858, https://doi.org/10.1007/s11295-008-0157-x (2008).
Abbott, R. J. & Brennan, A. C. Altitudinal gradients, plant hybrid zones and evolutionary novelty. Philos. T R. Soc. B 5, 369, https://doi.org/10.1098/rstb.2013.0346 (2014).
Janes, J. & Hamilton, J. Mixing it up: the role of hybridization in forest management and conservation under climate change. For. 8, 237, https://doi.org/10.3390/f8070237 (2017).
Ortego, J., Gugger, P. F., Riordan, E. C., Sork, V. L. & Emerson, B. Influence of climatic niche suitability and geographical overlap on hybridization patterns among southern Californian oaks. J. Biogeogr. 41, 1895–1908, https://doi.org/10.1111/jbi.12334 (2014).
Nevill, P. G., Després, T., Bayly, M. J., Bossinger, G. & Ades, P. K. Shared phylogeographic patterns and widespread chloroplast haplotype sharing in Eucalyptus species with different ecological tolerances. Tree Genet. Genomes 10, 1079–92, https://doi.org/10.1007/s11295-014-0744-y (2014).
Allendorf, F. W., Leary, R. F., Spruell, P. & Wenburg, J. K. The problems with hybrids: setting conservation guidelines. Trends Ecol. Evol. 16, 613–622, https://doi.org/10.1016/S0169-5347(01)02290-X (2001).
Gómez, J. M., González-Megías, A., Lorite, J., Abdelaziz, M. & Perfectti, F. The silent extinction: climate change and the potential hybridization-mediated extinction of endemic high-mountain plants. Biodivers. Conserv. 24, 1843–1857, https://doi.org/10.1007/s10531-015-0909-5 (2015).
Acknowledgements
J.B., G.W.J. and P.G.N. were supported partly by the Australian Government through the Australian Research Council Industrial Transformation Training Centre for Mine Site Restoration (Project Number ICI150100041). The support of the Friends of the Porongurup Range is also acknowledged.
Author information
Authors and Affiliations
Contributions
J.W.B., T.P.R., G.W.J. and P.G.N. conceived and designed the study. J.W.B. and G.W.J. conducted field work. J.W.B. and J.B. analysed the data. J.W.B. and P.G.N. wrote the manuscript with contributions and editorial comment from T.P.R., G.W.J., J.B. and M.B. All authors have reviewed and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ball, J.W., Robinson, T.P., Wardell-Johnson, G.W. et al. Fine-scale species distribution modelling and genotyping by sequencing to examine hybridisation between two narrow endemic plant species. Sci Rep 10, 1562 (2020). https://doi.org/10.1038/s41598-020-58525-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-58525-2
- Springer Nature Limited
This article is cited by
-
Potential distribution modeling based on machine learning of Sechium edule (Jacq.) Sw. in Japan
Genetic Resources and Crop Evolution (2024)
-
Identification of hybridization and introgression between Cinnamomum kanehirae Hayata and C. camphora (L.) Presl using genotyping-by-sequencing
Scientific Reports (2020)