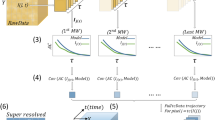
Abstract
Single-molecule localization microscopy (SMLM) relies on the blinking behavior of a fluorophore, which is the stochastic switching between fluorescent and dark states. Blinking creates multiple localizations belonging to the same fluorophore, confounding quantitative analyses and interpretations. Here we present a method, termed distance distribution correction (DDC), to eliminate blinking-caused repeat localizations without any additional calibrations. The approach relies on obtaining the true pairwise distance distribution of different fluorophores naturally from the imaging sequence by using distances between localizations separated by a time much longer than the average fluorescence survival time. We show that, using the true pairwise distribution, we can define and maximize the likelihood, obtaining a set of localizations void of blinking artifacts. DDC results in drastic improvements in obtaining the closest estimate of the true spatial organization and number of fluorescent emitters in a wide range of applications, enabling accurate reconstruction and quantification of SMLM images.






Similar content being viewed by others
Data availability
The data in this paper are shown in the main figures and Extended Data figures. All raw data for each of the simulation systems (Figs. 1 and 2) are also included at https://github.com/XiaoLabJHU/DDC. Source data are provided with this paper. All other data are available upon request. The complete package of DDC (data, code, user guide) is available for download at https://github.com/XiaoLabJHU/DDC (there are no access restrictions).
References
Betzig, E. et al. Imaging intracellular fluorescent proteins at nanometer resolution. Science 313, 1642–1645 (2006).
Rust, M. J., Bates, M. & Zhuang, X. Sub-diffraction-limit imaging by stochastic optical reconstruction microscopy (STORM). Nat. Methods 3, 793–796 (2006).
Hess, S. T., Girirajan, T. P. K. & Mason, M. D. Ultra-high resolution imaging by fluorescence photoactivation localization microscopy. Biophys. J. 91, 4258–4272 (2006).
Coltharp, C., Yang, X. & Xiao, J. Quantitative analysis of single-molecule superresolution images. Curr. Opin. Struct. Biol. 28, 112–121 (2014).
Baddeley, D. & Bewersdorf, J. Biological insight from super-resolution microscopy: what we can learn from localization-based images. Annu. Rev. Biochem. 87, 965–989 (2018).
Sauer, M. & Heilemann, M. Single-molecule localization microscopy in eukaryotes. Chem. Rev. 117, 7478–7509 (2017).
Endesfelder, U. et al. Multiscale spatial organization of RNA polymerase in Escherichia coli. Biophys. J. 105, 172–181 (2013).
Chen, X. et al. Study of RNA polymerase II clustering inside live-cell nuclei using Bayesian nanoscopy. ACS Nano 10, 2447–2454 (2016).
Weng, X. & Xiao, J. Spatial organization of transcription in bacterial cells. Trends Genet. 30, 287–297 (2014).
Lillemeier, B. F. et al. TCR and Lat are expressed on separate protein islands on T cell membranes and concatenate during activation. Nat. Immunol. 11, 90–96 (2010).
Rossy, J., Owen, D. M., Williamson, D. J., Yang, Z. & Gaus, K. Conformational states of the kinase Lck regulate clustering in early T cell signaling. Nat. Immunol. 14, 82–89 (2013).
Ehmann, N. et al. Quantitative super-resolution imaging of Bruchpilot distinguishes active zone states. Nat. Commun. 5, 4650 (2014).
Garcia-Parajo, M. F., Cambi, A., Torreno-Pina, J. A., Thompson, N. & Jacobson, K. Nanoclustering as a dominant feature of plasma membrane organization. J. Cell Sci. 127, 4995–5005 (2014).
Coltharp, C., Buss, J., Plumer, T. M. & Xiao, J. Defining the rate-limiting processes of bacterial cytokinesis. Proc. Natl Acad. Sci. USA 113, E1044–E1053 (2016).
Buss, J. et al. In vivo organization of the FtsZ-ring by ZapA and ZapB revealed by quantitative super-resolution microscopy. Mol. Microbiol. 89, 1099–1120 (2013).
Buss, J. et al. A multi-layered protein network stabilizes the Escherichia coli FtsZ-ring and modulates constriction dynamics. PLoS Genet. 11, e1005128 (2015).
Fu, G. et al. In vivo structure of the E. coli FtsZ-ring revealed by photoactivated localization microscopy (PALM). PLoS ONE 5, e12682 (2010).
Spühler, I. A., Conley, G. M., Scheffold, F. & Sprecher, S. G. Super resolution imaging of genetically labeled synapses in Drosophila brain tissue. Front. Cell. Neurosci. 10, 142 (2016).
Bar-On, D. et al. Super-resolution imaging reveals the internal architecture of nano-sized syntaxin clusters. J. Biol. Chem. 287, 27158–27167 (2012).
Xu, K., Zhong, G. & Zhuang, X. Actin, spectrin, and associated proteins form a periodic cytoskeletal structure in axons. Science 339, 452–456 (2013).
Wang, W., Li, G.-W., Chen, C., Xie, X. S. & Zhuang, X. Chromosome organization by a nucleoid-associated protein in live bacteria. Science 333, 1445–1449 (2011).
Xie, X., Cosma, M. P. & Lakadamyali, M. Super resolution imaging of chromatin in pluripotency, differentiation, and reprogramming. Curr. Opin. Genet. Dev. 46, 186–193 (2017).
Spahn, C., Endesfelder, U. & Heilemann, M. Super-resolution imaging of Escherichia coli nucleoids reveals highly structured and asymmetric segregation during fast growth. J. Struct. Biol. 185, 243–249 (2014).
Lehmann, M. et al. Quantitative multicolor super-resolution microscopy reveals tetherin HIV-1 interaction. PLoS Pathog. 7, e1002456 (2011).
Annibale, P., Scarselli, M., Kodiyan, A. & Radenovic, A. Photoactivatable fluorescent protein mEos2 displays repeated photoactivation after a long-lived dark state in the red photoconverted form. J. Phys. Chem. Lett. 1, 1506–1510 (2010).
Annibale, P., Vanni, S., Scarselli, M., Rothlisberger, U. & Radenovic, A. Quantitative photo activated localization microscopy: unraveling the effects of photoblinking. PLoS ONE 6, e22678 (2011).
Baumgart, F. et al. Varying label density allows artifact-free analysis of membrane-protein nanoclusters. Nat. Methods 13, 661–664 (2016).
Coltharp, C., Kessler, R. P. & Xiao, J. Accurate construction of photoactivated localization microscopy (PALM) images for quantitative measurements. PLoS ONE 7, e51725 (2012).
Sengupta, P. et al. Probing protein heterogeneity in the plasma membrane using PALM and pair correlation analysis. Nat. Methods 8, 969–975 (2011).
Puchner, E. M., Walter, J. M., Kasper, R., Huang, B. & Lim, W. A. Counting molecules in single organelles with superresolution microscopy allows tracking of the endosome maturation trajectory. Proc. Natl Acad. Sci. USA 110, 16015–16020 (2013).
Hartwich, T. M. P., Subach, F. V., Cooley, L., Verkhusha, V. V. & Bewersdorf, J. Determination of two-photon photoactivation rates of fluorescent proteins. Phys. Chem. Chem. Phys. 15, 14868–14872 (2013).
Lee, S.-H., Shin, J. Y., Lee, A. & Bustamante, C. Counting single photoactivatable fluorescent molecules by photoactivated localization microscopy (PALM). Proc. Natl Acad. Sci. USA 109, 17436–17441 (2012).
Rollins, G. C., Shin, J. Y., Bustamante, C. & Pressé, S. Stochastic approach to the molecular counting problem in superresolution microscopy. Proc. Natl Acad. Sci. USA 112, E110–E118 (2015).
Hummer, G., Fricke, F. & Heilemann, M. Model-independent counting of molecules in single-molecule localization microscopy. Mol. Biol. Cell 27, 3637–3644 (2016).
Nino, D., Rafiei, N., Wang, Y., Zilman, A. & Milstein, J. N. Molecular counting with localization microscopy: a Bayesian estimate based on fluorophore statistics. Biophys. J. 112, 1777–1785 (2017).
Huang, Z. et al. Spectral identification of specific photophysics of Cy5 by means of ensemble and single molecule measurements. J. Phys. Chem. 110, 45–50 (2005).
K L Yeow, E. et al. Characterizing the fluorescence intermittency and photobleaching kinetics of dye molecules immobilized on a glass surface. J. Phys. Chem. 110, 1726–1734 (2006).
Widengren, J., Chmyrov, A., Eggeling, C., Löfdahl, P.-Å & Seidel, C. A. M. Strategies to improve photostabilities in ultrasensitive fluorescence spectroscopy. J. Phys. Chem. 111, 429–440 (2007).
Vogelsang, J. et al. A reducing and oxidizing system minimizes photobleaching and blinking of fluorescent dyes. Angew. Chem. Int. Ed. 47, 5465–5469 (2008).
Veatch, S. L. et al. Correlation functions quantify super-resolution images and estimate apparent clustering due to over-counting. PLoS ONE 7, e31457 (2012).
Spahn, C., Herrmannsdörfer, F., Kuner, T. & Heilemann, M. Temporal accumulation analysis provides simplified artifact-free analysis of membrane-protein nanoclusters. Nat. Methods 13, 963–964 (2016).
Mo, G. C. H. et al. Genetically encoded biosensors for visualizing live-cell biochemical activity at super-resolution. Nat. Methods 14, 427–434 (2017).
Zhang, J. & Shapiro, M. S. Mechanisms and dynamics of AKAP79/150-orchestrated multi-protein signalling complexes in brain and peripheral nerve. J. Physiol. 594, 31–37 (2015).
Zhang, J., Carver, C. M., Choveau, F. S. & Shapiro, M. S. Clustering and functional coupling of diverse ion channels and signaling proteins revealed by super-resolution STORM microscopy in neurons. Neuron 92, 461–478 (2016).
Cella Zanacchi, F., Manzo, C., Magrassi, R., Derr, N. D. & Lakadamyali, M. Quantifying protein copy number in super resolution using an imaging-invariant calibration. Biophys. J. 116, 2195–2203 (2019).
Wooten, M. et al. Asymmetric histone inheritance via strand-specific incorporation and biased replication fork movement. Nat. Struct. Mol. Biol. 26, 732–743 (2019).
Habuchi, S. et al. Reversible single-molecule photoswitching in the GFP-like fluorescent protein Dronpa. Proc. Natl Acad. Sci. USA 102, 9511–9516 (2005).
Subach, F. V. et al. Photoactivatable mCherry for high-resolution two-color fluorescence microscopy. Nat. Methods 6, 153–159 (2009).
Malagon, F. RNase III is required for localization to the nucleoid of the 5′ pre-rRNA leader and for optimal induction of rRNA synthesis in E. coli. RNA 19, 1200–1207 (2013).
Bohrer, C. H., Bettridge, K. & Xiao, J. Reduction of confinement error in single-molecule tracking in live bacterial cells using SPICER. Biophys. J. 112, 568–574 (2017).
Das, R., Cairo, C. W. & Coombs, D. A hidden Markov model for single particle tracks quantifies dynamic interactions between LFA-1 and the actin cytoskeleton. PLoS Comput. Biol. 5, e1000556 (2009).
Fawcett, T. An introduction to ROC analysis. Pattern Recognit. Lett. 27, 861–874 (2006).
Thevathasan, J. V. et al. Nuclear pores as versatile reference standards for quantitative superresolution microscopy. Nat. Methods 16, 1045–1053 (2019).
Betschinger, J. & Knoblich, J. A. Dare to be different: asymmetric cell division in Drosophila, C. elegans and vertebrates. Curr. Biol. 14, R674–R685 (2004).
Tran, V., Lim, C., Xie, J. & Chen, X. Asymmetric division of Drosophila male germline stem cell shows asymmetric histone distribution. Science 338, 679–682 (2012).
Wang, Y., Maharana, S., Wang, M. D. & Shivashankar, G. V. Super-resolution microscopy reveals decondensed chromatin structure at transcription sites. Sci. Rep. 4, 4477 (2014).
Dempsey, G. T., Vaughan, J. C., Chen, K. H., Bates, M. & Zhuang, X. Evaluation of fluorophores for optimal performance in localization-based super-resolution imaging. Nat. Methods 8, 1027–1036 (2011).
Goossen-Schmidt, N. C., Schnieder, M., Hüve, J. & Klingauf, J. Switching behaviour of dSTORM dyes in glycerol-containing buffer. Sci. Rep. 10, 13746 (2020).
Hirvonen, L. M. & Cox, S. STORM without enzymatic oxygen scavenging for correlative atomic force and fluorescence superresolution microscopy. Methods Appl. Fluoresc. 6, 045002 (2018).
Shivanandan, A., Unnikrishnan, J. & Radenovic, A. Accounting for limited detection efficiency and localization precision in cluster analysis in single molecule localization microscopy. PLoS ONE 10, e0118767 (2015).
Lee, S. H., Shin, J. Y., Lee, A. & Bustamante, C. Counting single photoactivatable fluorescent molecules by photoactivated localization microscopy (PALM). Proc. Natl Acad. Sci. USA 109, 17436–17441 (2012).
Datsenko, K. A. & Wanner, B. L. One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc. Natl Acad. Sci. USA 97, 6640–6645 (2000).
Hensel, Z., Fang, X. & Xiao, J. Single-molecule imaging of gene regulation in vivo using cotranslational activation by cleavage (CoTrAC). J. Vis. Exp. e50042 https://doi.org/10.3791/50042 (2013).
Skinner, S. O., Sepúlveda, L. A., Xu, H. & Golding, I. Measuring mRNA copy number in individual Escherichia coli cells using single-molecule fluorescent in situ hybridization. Nat. Protoc. 8, 1100–1113 (2013).
Malagon, F. RNase III is required for localization to the nucleoid of the 5′ pre-rRNA leader and for optimal induction of rRNA synthesis in E. coli. RNA 19, 1200–1207 (2013).
Hensel, Z., Weng, X., Lagda, A. C. & Xiao, J. Transcription-factor-mediated DNA looping probed by high-resolution, single-molecule imaging in live E. coli cells. PLoS Biol. 11, e1001591 (2013).
Sage, D. et al. Quantitative evaluation of software packages for single-molecule localization microscopy. Nat. Methods 12, 717–724 (2015).
McKnight, S. L. & Miller Jr., O. L. Electron microscopic analysis of chromatin replication in the cellular blastoderm Drosophila melanogaster embryo. Cell 12, 795–804 (1977).
Lyu, Z., Coltharp, C., Yang, X. & Xiao, J. Influence of FtsZ GTPase activity and concentration on nanoscale Z-ring structure in vivo revealed by three-dimensional superresolution imaging. Biopolymers 105, 725–734 (2016).
Yang, X. et al. GTPase activity-coupled treadmilling of the bacterial tubulin FtsZ organizes septal cell wall synthesis. Science 355, 744–747 (2017).
Nahidiazar, L., Agronskaia, A. V., Broertjes, J., van den Broek, B. & Jalink, K. Optimizing imaging conditions for demanding multi-color super resolution localization microscopy. PLoS ONE 11, e0158884 (2016).
Schneider, C. A., Rasband, W. S. & Eliceiri, K. W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671–675 (2012).
Ovesný, M., Křížek, P., Borkovec, J., Svindrych, Z. & Hagen, G. M. ThunderSTORM: a comprehensive ImageJ plug-in for PALM and STORM data analysis and super-resolution imaging. Bioinformatics 30, 2389–2390 (2014).
Weng, X. et al. Spatial organization of RNA polymerase and its relationship with transcription in Escherichia coli. Proc. Natl Acad. Sci. USA 116, 20115–20123 (2019).
Flors, C., Ravarani, C. N. J. & Dryden, D. T. F. Super-resolution imaging of DNA labelled with intercalating dyes. Chemphyschem 10, 2201–2204 (2009).
Nahidiazar, L., Agronskaia, A. V., Broertjes, B., van den Broek, B. & Jalink, K. Optimizing imaging conditions for demanding multi-color super resolution localization microscopy. PLoS ONE 11, e0158884 (2016).
Acknowledgements
This work was supported by the NIH (5T32GM007231, C.H.B.; F31GM115149-01A1, M.W.; R01 GM086447; and R01 DK073368 and R35CA197622, J.Z.) and the NSF (MCB1817551), a Johns Hopkins Discovery Award, a Hamilton Innovation Research Award (J.X.), NIGMS/NIH (R01GM112008, J.X. and X.C.; R35GM127075, X.C.; and R01GM133842, M.L.) and the Howard Hughes Medical Institute (55108512, X.C.).
Author information
Authors and Affiliations
Contributions
C.H.B., conceptualization, discovery and development of theory, software, data collection, data analysis, writing (original draft, review and editing); X.Y., experimental data collection, data analysis, writing (review and editing), project guidance; S.T., experimental data collection, data analysis; X.W., experimental data collection, data analysis; B.T., experimental data collection, data analysis, writing (review and editing); R.M., development of the user guide, software, writing (review and editing); B.R., experimental data collection, data analysis; M.W., experimental data collection, writing (review and editing); X.C., project guidance, funding acquisition, writing (review); J.Z., project guidance, funding acquisition, writing (review); E.R., project guidance, writing (review); M.L., experimental conceptualization, funding acquisition, writing (review and editing); J.X., conceptualization, project guidance, writing (original draft), supervision, funding acquisition, project administration, writing (review and editing).
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Rita Strack was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team. Nature Methods thanks Paolo Annibale, Sebastian Malkusch, and the other, anonymous reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Photokinetic Models.
The two kinetic models used to simulate blinking, (a) 2 dark state and (b) 1 dark state. The transition probabilities per frame are shown in the figure.
Extended Data Fig. 2 Converging of Pairwise distance distributions.
Extended Data Fig. 3 Matching the true pairwise distance distribution.
An illustration of the pairwise distance distributions at a certain frame difference, Δn, before and after being corrected with DDC. When the likelihood is maximized all of the pairwise distance distributions will match the true pairwise distance distribution. [The true pairwise distance distribution is shown as black dots.].
Extended Data Fig. 4 Toy model illustration for inner workings of DDC.
Toy model illustration for inner workings of DDC (See text within SI for in depth description): a, Simple toy model with 4 true localizations and 2 repeats (color coded), with the number showing the frame of each localization (can also be used to identify each localization for this example). b, The true pairwise distance distribution (PT (Δr)) and the distribution of distances between loci given that at least one is a repeat (PR1(Δr∣Δn = 1)) for the localizations within (a) The number (and probability) for ‘small’ distances and ‘large’ distances for each distribution is above each bar, with an assigned variable (a, b, c, d) used in the calculation of the Likelihood (Lik). We also show the specific pairs of loci under the bars to illustrate how assigning a particular loci to a certain set influences the likelihood calculation. Note: for this specific example blinks only appear with Δn = 1, and hence we ignore the distributions with Δn > 1 (See text). c, Simplified illustration of how Alg. 1 and Alg. 2 work together and assign localizations as a true localization or repeat localization. Multiple steps of the MCMC are shown with different rows (1 to 3) (See Text). Alg. 1 essentially calculates the probability that a localization is a repeat (green bars), if this value is above .5 it is assigned to that set. Alg. 2 varies this calculation by a small amount each step, generating new sets d, The sets assigned in (c) lead to different likelihoods (due to the particular distribution the distance between each pair is assigned (changing (a,b,c,d), note how the specific distances between each pair change with each assigned set), when the distributions of the assigned sets match the correct distributions (those in (B)) Lik is maximized. (See text for further details).
Extended Data Fig. 5 Maximization of Likelihood Results in Correct Conformation of Localizations.
Maximization of Likelihood Results in Correct Conformation of Localizations: For 6 systems investigated within this work, we randomly varied the percentage of true localizations and calculated the log(Lik) and the image error for each conformation (See Text).
Extended Data Fig. 6 Overcounting and undercounting in individual pixels.
Overcounting and undercounting in individual pixels: Comparison of four different thresholding methods with DDC in counting the number of true localizations in individual pixels on five spatial distributions as depicted and simulated in main text Fig. 2. The y axis is the difference between the true count and the method-identified count expressed as Count-[True Count], with positive values indicating the degree of over-counting and negative values the degree of under-counting. The x-axis is the number of true counts in individual pixels. The pixel size was set to 50 nm. Note that only DDC shows consistent distributions of y values near zero at different true count values and across all five spatial patterns. [Each scatter point is colored to illustrate the estimated probability density - allowing one to visualize the regions of high density (red) and regions of low density (blue).].
Extended Data Fig. 7 AKAP scatter plots through time.
Scatter plots for a section of a cell with the localizations from AKAP79 with the color indicating the frame of the localization (Blue is early and Red is late) for the three different methodologies.
Extended Data Fig. 8 Computationally varying the label density.
a, The results of computationally varying the label density on some of the simulation systems. b, The results of computationally varying the label density on AKAP79 and AKA150. (Values greater than 1 indicate significant clustering.).
Extended Data Fig. 9 Experimental Concerns.
Image Error at different densities of localizations (a) and activation probability per frame (b). The raw data points are shown as gray points and the moving average is shown in black (Supporting Material). c, An intensity trajectory of a single mEos3.2 molecule with labels showing the definitions of Ton and Toff. d, The average Ton, Toff (per frame, frame rate 33Hz), and number of blinks for Alexa647 and mEos3.2 at different UV activation intensities (405 Power).
Extended Data Fig. 10 Varying Raw Image Error.
The raw Image Error (Not Normalized) for the uncorrected SMLM images for varying the density of the localizations and the activation energy.
Supplementary information
Supplementary Information
Supplementary Table 1, Discussion and Figs. 1–12.
Source data
Source Data Fig. 1
Data used to generate plots in Fig. 1.
Source Data Fig. 2
Data used to generate plots in Fig. 2.
Source Data Fig. 3
Data used to generate plots in Fig. 3.
Source Data Fig. 4
Data used to generate plots in Fig. 4.
Source Data Fig. 5
Data used to generate plots in Fig. 5.
Source Data Fig. 6
Data used to generate plots in Fig. 6.
Source Data Extended Data Fig. 1
Data used to generate plots in Extended Data Fig. 1.
Source Data Extended Data Fig. 2
Data used to generate plots in Extended Data Fig. 2.
Source Data Extended Data Fig. 3
Data used to generate plots in Extended Data Fig. 3.
Source Data Extended Data Fig. 4
Data used to generate plots in Extended Data Fig. 4.
Source Data Extended Data Fig. 5
Data used to generate plots in Extended Data Fig. 5.
Source Data Extended Data Fig. 6
Data used to generate plots in Extended Data Fig. 6.
Source Data Extended Data Fig. 7
Data used to generate plots in Extended Data Fig. 7.
Source Data Extended Data Fig. 8
Data used to generate plots in Extended Data Fig. 8.
Source Data Extended Data Fig. 9
Data used to generate plots in Extended Data Fig. 9.
Source Data Extended Data Fig. 10
Data used to generate plots in Extended Data Fig. 10.
Rights and permissions
About this article
Cite this article
Bohrer, C.H., Yang, X., Thakur, S. et al. A pairwise distance distribution correction (DDC) algorithm to eliminate blinking-caused artifacts in SMLM. Nat Methods 18, 669–677 (2021). https://doi.org/10.1038/s41592-021-01154-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41592-021-01154-y
- Springer Nature America, Inc.
This article is cited by
-
Disordered C-terminal domain drives spatiotemporal confinement of RNAPII to enhance search for chromatin targets
Nature Cell Biology (2024)
-
Temporal analysis of relative distances (TARDIS) is a robust, parameter-free alternative to single-particle tracking
Nature Methods (2024)
-
A framework for evaluating the performance of SMLM cluster analysis algorithms
Nature Methods (2023)
-
Unbiased choice of global clustering parameters for single-molecule localization microscopy
Scientific Reports (2022)
-
High-precision estimation of emitter positions using Bayesian grouping of localizations
Nature Communications (2022)