Abstract
Habitat degradation and climate change are globally acting as pivotal drivers of wildlife collapse, with mounting evidence that this erosion of biodiversity will accelerate in the following decades1,2,3. Here, we quantify the past, present and future ecological suitability of Europe for bumblebees, a threatened group of pollinators ranked among the highest contributors to crop production value in the northern hemisphere4,5,6,7,8. We demonstrate coherent declines of bumblebee populations since 1900 over most of Europe and identify future large-scale range contractions and species extirpations under all future climate and land use change scenarios. Around 38–76% of studied European bumblebee species currently classified as ‘Least Concern’ are projected to undergo losses of at least 30% of ecologically suitable territory by 2061–2080 compared to 2000–2014. All scenarios highlight that parts of Scandinavia will become potential refugia for European bumblebees; it is however uncertain whether these areas will remain clear of additional anthropogenic stressors not accounted for in present models. Our results underline the critical role of global change mitigation policies as effective levers to protect bumblebees from manmade transformation of the biosphere.


Similar content being viewed by others
Data availability
Bombus occurrence data are available on Atlas Hymenoptera: http://www.atlashymenoptera.net/page.aspx?id=169). All ISIMIP2b data used are publicly available on https://www.isimip.org/protocol/2b/.
Code availability
R scripts related to the ecological niche modelling and projections are all available at https://github.com/sdellicour/projections_bombus.
References
Brondizio, E. S., Settele, J., Diaz, S. & Ngo, H. T. Global Assessment Report on Biodiversity and Ecosystem Services (IPBES, 2019).
Wagner, D. L. Insect declines in the Anthropocene. Annu. Rev. Entomol. 65, 457–480 (2020).
Forister, M. L., Pelton, E. M. & Black, S. H. Declines in insect abundance and diversity: we know enough to act now. Conserv. Sci. Pract. 1, e80 (2019).
Rasmont, P. et al. Climatic risk and distribution atlas of European Bumblebees Biorisk 10 (Pensoft Publishers, 2015).
Rasmont, P., Ghisbain, G. & Terzo, M. Bumblebees of Europe and Neighbouring Regions (NAP Editions, 2021).
Nieto, A. et al. European Red List of Bees (Publication Office of the European Union, 2014).
Kleijn, D. et al. Delivery of crop pollination services is an insufficient argument for wild pollinator conservation. Nat. Commun. 6, 7414 (2015).
Cameron, S. A. & Sadd, B. M. Global trends in bumble bee health. Annu. Rev. Entomol. 65, 209–232 (2020).
Wagner, D. L., Grames, E. M., Forister, M. L., Berenbaum, M. R. & Stopak, D. Insect decline in the Anthropocene: death by a thousand cuts. Proc. Natl Acad. Sci. USA 118, e2023989118 (2021).
Hallmann, C. A. et al. More than 75 percent decline over 27 years in total flying insect biomass in protected areas. PLoS ONE 12, e0185809 (2017).
Warren, R., Price, J., Graham, E., Forstenhaeusler, N. & VanDerWal, J. The projected effect on insects, vertebrates and plants of limiting global warming to 1.5 °C rather than 2 °C. Science 360, 791–795 (2018).
Chowdhury, S. et al. Protected areas and the future of insect conservation. Trends Ecol. Evol. 38, 85–95 (2022).
Dicks, L. V. et al. A global-scale expert assessment of drivers and risks associated with pollinator decline. Nat. Ecol. Evol. 5, 1453–1461 (2021).
Potts, S. G. et al. Safeguarding pollinators and their values to human well-being. Nature 540, 220–229 (2016).
Gallai, N., Salles, J.-M., Settele, J. & Vaissière, B. E. Economic valuation of the vulnerability of world agriculture confronted with pollinator decline. Ecol. Econ. 68, 810–821 (2009).
Ollerton, J., Winfree, R. & Tarrant, S. How many flowering plants are pollinated by animals? Oikos 120, 321–326 (2011).
Goulson, D., Lye, G. C. & Darvill, B. Decline and conservation of bumble bees. Annu. Rev. Entomol. 53, 191–208 (2008).
Hemberger, J., Crossley, M. S. & Gratton, C. Historical decrease in agricultural landscape diversity is associated with shifts in bumble bee species occurrence. Ecol. Lett. 24, 1800–1813 (2021).
Kerr, J. T. et al. Climate change impacts on bumblebees converge across continents. Science 349, 177–180 (2015).
Drossart, M. et al. Belgian Red List of Bees (Presse universitaire de l’Université de Mons, 2019).
Reemer, M. Basisrapport voor de Rode Lijst Bijen (Ministerie van Landbouw, Natuur en Voedselkwaliteit, 2018).
Frieler, K. et al. Assessing the impacts of 1.5 °C global warming—simulation protocol of the Inter-Sectoral Impact Model Intercomparison Project (ISIMIP2b). Geosci. Model Dev. 10, 4321–4345 (2017).
Thiery, W. et al. Intergenerational inequities in exposure to climate extremes. Science 374, 158–160 (2021).
Elith, J., Leathwick, J. R. & Hastie, T. A working guide to boosted regression trees. J. Anim. Ecol. 77, 802–813 (2008).
Ghisbain, G., Gérard, M., Wood, T. J., Hines, H. M. & Michez, D. Expanding insect pollinators in the Anthropocene. Biol. Rev. 96, 2755–2770 (2021).
Williams, P. H. et al. The arctic and alpine bumblebees of the subgenus Alpinobombus revised from integrative assessment of species’ gene coalescents and morphology (Hymenoptera, Apidae, Bombus). Zootaxa 4625, 1–68 (2019).
Martinet, B. et al. Global effects of extreme temperatures on wild bumblebees. Conserv. Biol. 35, 1507–1518 (2021).
Biella, P. et al. Distribution patterns of the cold adapted bumblebee Bombus alpinus in the Alps and hints of an uphill shift (Insecta: Hymenoptera: Apidae). J. Insect. Conserv. 21, 357–366 (2017).
Guidelines for Using the IUCN Red List Categories and Criteria. Version 15.1 https://www.iucnredlist.org/documents/RedListGuidelines.pdf (IUCN, 2022).
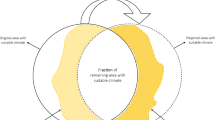
Pecl, G. T. et al. Biodiversity redistribution under climate change: impacts on ecosystems and human well-being. Science 355, eaai9214 (2017).
Myers, N., Mittermeier, R. A., Mittermeier, C. G., da Fonseca, G. A. B. & Kent, J. Biodiversity hotspots for conservation priorities. Nature 403, 853–858 (2000).
Ashcroft, M. B. Identifying refugia from climate change. J. Biogeogr. 37, 1407–1413 (2010).
Fijen, T. P. M. Mass-migrating bumblebees: an overlooked phenomenon with potential far-reaching implications for bumblebee conservation. J. Appl. Ecol. 58, 274–280 (2021).
Schmid-Hempel, R. et al. The invasion of southern South America by imported bumblebees and associated parasites. J. Anim. Ecol. 83, 823–837 (2014).
Ghisbain, G. Are bumblebees relevant models for understanding wild bee decline? Front. Conserv. Sci. 2, 752213 (2021).
Marshall, L. et al. Bumblebees moving up: shifts in elevation ranges in the Pyrenees over 115 years. Proc. R. Soc. B. 287, 20202201 (2020).
Goulson, D., Nicholls, E., Botias, C. & Rotheray, E. L. Bee declines driven by combined stress from parasites, pesticides and lack of flowers. Science 347, 1255957 (2015).
Potts, S. G. et al. Global pollinator declines: trends, impacts and drivers. Trends Ecol. Evol. 25, 345–353 (2010).
Gérard, M., Amiri, A., Cariou, B. & Baird, E. Short-term exposure to heatwave-like temperatures affects learning and memory in bumblebees. Glob. Change Biol. 28, 4251–4259 (2022).
Martinet, B. et al. Mating under climate change: Impact of simulated heatwaves on the reproduction of model pollinators. Funct. Ecol. 35, 739–752 (2020).
Hoffmann, A. A. & Sgrò, C. M. Climate change and evolutionary adaptation. Nature 470, 479–485 (2011).
Gérard, M. et al. Shift in size of bumblebee queens over the last century. Glob. Change Biol. 26, 1185–1195 (2020).
Lecocq, T. et al. An integrative taxonomic approach to assess the status of Corsican bumblebees: implications for conservation. Anim. Conserv. 18, 236–248 (2015).
Lecocq, T. et al. Methods for species delimitation in bumblebees (Hymenoptera, Apidae, Bombus): towards an integrative approach. Zool. Scr. 44, 281–297 (2015).
Brasero, N. et al. Resolving the species status of overlooked West‐Palaearctic bumblebees. Zool. Scr. 50, 616–632 (2021).
Ghisbain, G. et al. A worthy conservation target? Revising the status of the rarest bumblebee of Europe. Insect Conserv. Divers. 14, 661–674 (2021).
Williams, P. H. Not just cryptic a barcode bush: PTP re-analysis of global data for the bumblebee subgenus Bombus s. str. supports additional species (Apidae, genus Bombus). J. Nat. Hist. 55, 271–282 (2021).
Williams, P. H. et al. Widespread polytypic species or complexes of local species? Revising bumblebees of the subgenus Melanobombus world-wide (Hymenoptera, Apidae, Bombus). Eur. J. Taxon. 719, 1–120 (2020).
Ghisbain, G., Michez, D., Marshall, L., Rasmont, P. & Dellicour, S. Wildlife conservation strategies should incorporate both taxon identity and geographical context—further evidence with bumblebees. Divers. Distrib. 26, 1741–1751 (2020).
Soroye, P., Newbold, T. & Kerr, J. Climate change contributes to widespread declines among bumble bees across continents. Science 367, 685–688 (2020).
Williams, P. H., Cameron, S. A., Hines, H. M., Cederberg, B. & Rasmont, P. A simplified subgeneric classification of the bumblebees (genus Bombus). Apidologie 39, 46–74 (2008).
Vray, S. et al. A century of local changes in bumblebee communities and landscape composition in Belgium. J. Insect Conserv. 23, 489–501 (2019).
Christians, C. Quarante ans de politique agricole européenne commune et d’agriculture en Belgique. Bull. Soc. Géograph. Liège 35, 41–55 (1998).
Rollin, O. et al. Drastic shifts in the Belgian bumblebee community over the last century. Biodivers. Conserv. 29, 2553–2573 (2020).
Elith, J. et al. Novel methods improve prediction of species’ distributions from occurrence data. Ecography 29, 129–151 (2006).
Klitting, R. et al. Predicting the evolution of the Lassa virus endemic area and population at risk over the next decades. Nat. Commun. 13, 5596 (2022).
Marshall, L. et al. The interplay of climate and land use change affects the distribution of EU bumblebees. Glob. Change Biol. 24, 101–116 (2018).
Randin, C. F. et al. Are niche-based species distribution models transferable in space? J. Biogeogr. 33, 1689–1703 (2006).
Valavi, R., Elith, J., Lahoz-Monfort, J. J. & Guillera-Arroita, G. blockCV: an R package for generating spatially or environmentally separated folds for k-fold cross-validation of species distribution models. Methods Ecol. Evol. 10, 225–232 (2019).
Lobo, J. M., Jiménez-Valverde, A. & Real, R. AUC: a misleading measure of the performance of predictive distribution models. Glob. Ecol. Biogeogr. 17, 145–151 (2008).
Jiménez-Valverde, A. Threshold-dependence as a desirable attribute for discrimination assessment: implications for the evaluation of species distribution models. Biodivers. Conserv. 23, 369–385 (2014).
Jiménez-Valverde, A. Insights into the area under the receiver operating characteristic curve (AUC) as a discrimination measure in species distribution modelling. Glob. Ecol. Biogeogr. 21, 498–507 (2012).
Sørensen, T. A method of establishing groups of equal amplitude in plant sociology based on similarity of species and its application to analyses of the vegetation on Danish commons. K. Dansk.Vidensk. Selsk. Skr. 5, 1–34 (1948).
Leroy, B. et al. Without quality presence–absence data, discrimination metrics such as TSS can be misleading measures of model performance. J. Biogeogr. 45, 1994–2002 (2018).
Li, W. & Guo, Q. How to assess the prediction accuracy of species presence–absence models without absence data? Ecography 36, 788–799 (2013).
Lange, S. Bias correction of surface downwelling longwave and shortwave radiation for the EWEMBI dataset. Earth Syst. Dynam. 9, 627–645 (2018).
Dunne, J. P. et al. GFDL’s ESM2 global coupled climate–carbon earth system models. Part I: Physical formulation and baseline simulation characteristics. J. Clim. 25, 6646–6665 (2012).
Jones, C. D. et al. The HadGEM2-ES implementation of CMIP5 centennial simulations. Geosci. Model Dev. 4, 543–570 (2011).
Dufresne, J.-L. et al. Climate change projections using the IPSL-CM5 Earth System Model: from CMIP3 to CMIP5. Clim. Dynam. 40, 2123–2165 (2013).
Watanabe, M. et al. Improved climate simulation by MIROC5: mean states, variability and climate sensitivity. J. Clim. 23, 6312–6335 (2010).
Jones, B. & O’Neill, B. C. Spatially explicit global population scenarios consistent with the Shared Socioeconomic Pathways. Environ. Res. Lett. 11, 084003 (2016).
Acknowledgements
We are grateful to A. Dejins for his useful comments on the analytical approach. G.G., F.M. and S.D. acknowledge support from the Fonds National de la Recherche Scientifique (FNRS, Belgium). G.G. and D.M. acknowledge support from the European DG ENV projects ORBIT (contract no. 09.029901/2021/848268/SER/ENV.D.2), PULSE (contract no. 07.027755/2020/840209/SER/ENV.D.2), SPRING (contract no. 09.02001/2021/847887/SER/ENV.D.2) and the H2020 project SAFEGUARD (grant agreement no. 101003476). S.D. acknowledges support from the FNRS (grant no. F.4515.22), the Research Foundation—Flanders (Fonds voor Wetenschappelijk Onderzoek—Vlaanderen, grant no. G098321N) and from the European Union Horizon 2020 project MOOD (grant agreement no. 874850). For the production and coordination of the input data and impact model output of ISIMIP (www.isimip.org), we are grateful to the modelling groups, the ISIMIP sector coordinators and the ISIMIP cross-sectoral science team.
Author information
Authors and Affiliations
Contributions
G.G. and S.D. designed the research and wrote the manuscript. S.D. conducted the analyses with the assistance of W.T., F.M. and D.E. P.R. and D.M. managed the database. All authors contributed to editing the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature thanks Nicole Miller-Struttmann, Daniel Silva and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Assessment of the predictive performance of species-specific ecological niche models trained on past (1901–1970) and present-day (2000–2014) occurrence and environmental data.
Specifically, we computed the prevalence-pseudoabsence-calibrated Sørensen index (SIppc) while performing an optimization of the ecological suitability threshold in the range [0, 1] with a 0.01 step increment. This threshold value was used to generate binary versions of the ecological suitability maps necessary for the computation of this index and we eventually selected the threshold value maximizing the SIppc (see Extended Data Table 1 for the optimized SIppc values and associated threshold). We here report the evolution of the SIppc according to the considered ecological suitability threshold value. We report this relationship for each species and for each ecological niche model trained on past (light grey curves) and present-day (dark grey curves) data.
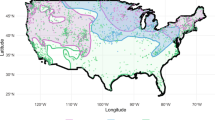
Extended Data Fig. 2 Evolution of the ecological suitability map of European bumblebees as measured by the species richness index (SRI).
The SRI is here defined as the local number of species associated with an ecological suitability higher than the median threshold value retrieved from the optimization step of the prevalence-pseudoabsence-calibrated Sørensen index (SIppc) computed for each of ten ecological niche models trained on present-day data (see the Methods for further detail). We here report the SRI estimates for the past, present (‘t0’) and future projections, as well as the differences between past, present (‘t0’) and future projections.
Extended Data Fig. 3 Occurrence data available for the 46 selected European bumblebee species for the first time period (1901–1974).
Duplicate records were removed to reflect species presence rather than species density and to further decrease potential sampling biases. All species distributions are represented by at least 30 unique data records. A total of 125,283 unique records were retained for this time period.
Extended Data Fig. 4 Occurrence data available for the 46 selected European bumblebee species for the second time period (2000–2014).
Duplicate records were removed to reflect species presence rather than species density and to further decrease potential sampling biases. All species distributions are represented by at least 30 unique data records. A total of 275,763 unique records were retained for this time period.
Extended Data Fig. 5 Response curves associated with the different environmental variables included in the ecological niche models.
Each curve was retrieved from a boosted regression tree (BRT) model trained for a distinct species on present-day (2000–2014) occurrence data.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ghisbain, G., Thiery, W., Massonnet, F. et al. Projected decline in European bumblebee populations in the twenty-first century. Nature 628, 337–341 (2024). https://doi.org/10.1038/s41586-023-06471-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-023-06471-0
- Springer Nature Limited
This article is cited by
-
Climate change predicted to exacerbate declines in bee populations
Nature (2024)
-
Contribution of climate change to the spatial expansion of West Nile virus in Europe
Nature Communications (2024)
-
Unveiling of climate change-driven decline of suitable habitat for Himalayan bumblebees
Scientific Reports (2024)
-
Heat resistance variability in the Lebanese bee fauna
Apidologie (2024)