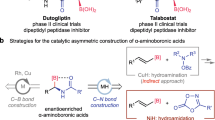
Abstract
Bioisosteric replacement has emerged as a clear strategy for drug-structure optimization. Naphthalene is the core element of many chiral pharmaceuticals and drug candidates. However, as a promising isostere of naphthalene, the chiral version of 1,2-benzazaborine has rarely been explored due to the lack of efficient synthetic methods. Here we describe a copper-catalysed enantioselective hydroboration of alkenes with 1,2-benzazaborines. The method provides a general platform for the atom-economic and efficient construction of diverse chiral 1,2-benzazaborine compounds (more than 60 examples) that bear a 2-carbon-stereogenic centre or allene skeleton in high yields and excellent enantioselectivities. Three 1,2-benzazaborine analogues of bioactive chiral naphthalene-containing molecules have been prepared, and a series of transformations around chiral 1,2-benzazaborines have also been developed. Notably, the hydroboration process of this study reveals that the identity of 1,2-benzazaborine plays an essential role in the rate-determining step and catalyst resting state.




Similar content being viewed by others
Data availability
The data that support the findings of this study are available within the Article and its Supplementary Information files. Crystallographic data for the structure reported in this Article has been deposited at the Cambridge Crystallographic Data Centre, under deposition number CCDC 2245401 (3b). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/.
References
Patani, G. A. & LaVoie, E. J. Bioisosterism: a rational approach in drug design. Chem. Rev. 96, 3147–3176 (1996).
Brown, N. (ed.) Bioisosteres in Medicinal Chemistry (Wiley, 2012).
Makar, S., Saha, T. & Singh, S. K. Naphthalene, a versatile platform in medicinal chemistry: sky-high perspective. Eur. J. Med. Chem. 161, 252–276 (2019).
Bosdet, M. J. D. & Piers, W. E. B-N as a C-C substitute in aromatic systems. Can. J. Chem. 87, 8–29 (2009).
Giustra, Z. X. & Liu, S.-Y. The state of the art in azaborine chemistry: new synthetic methods and applications. J. Am. Chem. Soc. 140, 1184–1194 (2018).
Stojanović, M. & Baranac-Stojanović, M. Mono BN-substituted analogues of naphthalene: a theoretical analysis of the effect of BN position on stability, aromaticity and frontier orbital energies. New J. Chem. 42, 12968–12976 (2018).
McConnell, C. R. & Liu, S.-Y. Late-stage functionalization of BN-heterocycles. Chem. Soc. Rev. 48, 3436–3453 (2019).
Bhattacharjee, A., Davies, G. H. M., Saeednia, B., Wisniewski, S. R. & Molander, G. A. Selectivity in the elaboration of bicyclic borazarenes. Adv. Synth. Catal. 363, 2256–2273 (2021).
Abengózar, A., García-García, P., Fernández-Rodríguez, M. A., Sucunza, D. & Vaquero, J. J. in Advances in Heterocyclic Chemistry (eds Scriven, E. F.V. & Ramsden, C. A.) Vol. 135, 197–259 (Elsevier, 2021).
Rombouts, F. J. R., Tovar, F., Austin, N., Tresadern, G. & Trabanco, A. A. Benzazaborinines as novel bioisosteric replacements of naphthalene: propranolol as an example. J. Med. Chem. 58, 9287–9295 (2015).
Vlasceanu, A., Jessing, M. & Kilburn, J. P. BN/CC isosterism in borazaronaphthalenes towards phosphodiesterase 10A (PDE10A) inhibitors. Bioorg. Med. Chem. 23, 4453–4461 (2015).
Lee, H., Fischer, M., Shoichet, B. K. & Liu, S.-Y. Hydrogen bonding of 1,2-azaborines in the binding cavity of T4 lysozyme mutants: structures and thermodynamics. J. Am. Chem. Soc. 138, 12021–12024 (2016).
Lovering, F. Escape from Flatland 2: complexity and promiscuity. Med. Chem. Commun. 4, 515–519 (2013).
Ishikawa, M. & Hashimoto, Y. Improvement in aqueous solubility in small molecule drug discovery programs by disruption of molecular planarity and symmetry. J. Med. Chem. 54, 1539–1554 (2011).
Mazzanti, A., Mercanti, E. & Mancinelli, M. Axial chirality about boron-carbon bond: atropisomeric azaborines. Org. Lett. 18, 2692–2695 (2016).
Yang, J. et al. Chiral phosphoric acid-catalyzed remote control of axial chirality at boron-carbon bond. J. Am. Chem. Soc. 143, 12924–12929 (2021).
Zhang, X. et al. Stepwise asymmetric allylic substitution-isomerization enabled mimetic synthesis of axially chiral B,N‐heterocycles. Angew. Chem. Int. Ed. 61, e202210456 (2022).
Morita, T., Murakami, H., Asawa, Y. & Nakamura, H. Enantioselective synthesis of oxazaborolidines by palladium-catalyzed N-H/B-H double activation of 1,2-azaborines. Angew. Chem. Int. Ed. 61, e202113558 (2022).
Wisniewski, S. R., Guenther, C. L., Argintaru, O. A. & Molander, G. A. A convergent, modular approach to functionalized 2,1-borazaronaphthalenes from 2-aminostyrenes and potassium organotrifluoroborates. J. Org. Chem. 79, 365–378 (2014).
Bagutski, V., Ros, A. & Aggarwal, V. K. Improved method for the conversion of pinacolboronic esters into trifluoroborate salts: facile synthesis of chiral secondary and tertiary trifluoroborates. Tetrahedron 65, 9956–9960 (2009).
Noh, D., Yoon, S. K., Won, J., Lee, J. Y. & Yun, J. An efficient copper(I)-catalyst system for the asymmetric hydroboration of β-substituted vinylarenes with pinacolborane. Chem. Asian J. 6, 1967–1969 (2011).
Jang, W. J., Song, S. M., Park, Y. & Yun, J. Asymmetric synthesis of γ-hydroxy pinacolboronates through copper-catalyzed enantioselective hydroboration of α,β-unsaturated aldehydes. J. Org. Chem. 84, 4429–4434 (2019).
Sang, H. L., Yu, S. & Ge, S. Copper-catalyzed asymmetric hydroboration of 1,3-enynes with pinacolborane to access chiral allenylboronates. Org. Chem. Front. 5, 1284–1287 (2018).
Jang, W. J., Song, S. M., Moon, J. H., Lee, J. Y. & Yun, J. Copper-catalyzed enantioselective hydroboration of unactivated 1,1-disubstituted alkenes. J. Am. Chem. Soc. 139, 13660–13663 (2017).
Xi, Y. & Hartwig, J. F. Diverse asymmetric hydrofunctionalization of aliphatic internal alkenes through catalytic regioselective hydroboration. J. Am. Chem. Soc. 138, 6703–6706 (2016).
Huang, Y., del Pozo, J., Torker, S. & Hoveyda, A. H. Enantioselective synthesis of trisubstituted allenyl-B(pin) compounds by phosphine-Cu-catalyzed 1,3-enyne hydroboration. insights regarding stereochemical integrity of Cu-allenyl intermediates. J. Am. Chem. Soc. 140, 2643–2655 (2018).
Noh, D., Chea, H., Ju, J. & Yun, J. Highly regio- and enantioselective copper-catalyzed hydroboration of styrenes. Angew. Chem. Int. Ed. 48, 6062–6064 (2009).
Feng, X., Jeon, H. & Yun, J. Regio- and enantioselective copper(I)-catalyzed hydroboration of borylalkenes: asymmetric synthesis of 1,1-diborylalkanes. Angew. Chem. Int. Ed. 52, 3989–3992 (2013).
Chen, X., Cheng, Z., Guo, J. & Lu, Z. Asymmetric remote C-H borylation of internal alkenes via alkene isomerization. Nat. Commun. 9, 3939 (2018).
Gao, D.-W. et al. Cascade CuH-catalysed conversion of alkynes into enantioenriched 1,1-disubstituted products. Nat. Catal. 3, 23–29 (2019).
Rubina, M., Rubin, M. & Gevorgyan, V. Catalytic enantioselective hydroboration of cyclopropenes. J. Am. Chem. Soc. 125, 7198–7199 (2003).
Yu, S., Wu, C. & Ge, S. Cobalt-catalyzed asymmetric hydroboration/cyclization of 1,6-enynes with pinacolborane. J. Am. Chem. Soc. 139, 6526–6529 (2017).
Chen, X., Cheng, Z. & Lu, Z. Cobalt-catalyzed asymmetric Markovnikov hydroboration of styrenes. ACS Catal. 9, 4025–4029 (2019).
Jin, S. et al. Enantioselective Cu-catalyzed double hydroboration of alkynes to access chiral gem-diborylalkanes. Nat. Commun. 13, 3524 (2022).
Dong, W. et al. Enantioselective Rh-catalyzed hydroboration of silyl enol ethers. J. Am. Chem. Soc. 143, 10902–10909 (2021).
Mazet, C. & Gérard, D. Highly regio- and enantioselective catalytic asymmetric hydroboration of α-substituted styrenyl derivatives. Chem. Commun. 47, 298–300 (2011).
Bochat, A. J., Shoba, V. M. & Takacs, J. M. Ligand-controlled regiodivergent enantioselective rhodium-catalyzed alkene hydroboration. Angew. Chem. Int. Ed. 58, 9434–9438 (2019).
Bai, X.-Y., Zhao, W., Sun, X. & Li, B.-J. Rhodium-catalyzed regiodivergent and enantioselective hydroboration of enamides. J. Am. Chem. Soc. 141, 19870–19878 (2019).
Gao, T.-T., Zhang, W.-W., Sun, X., Lu, H.-X. & Li, B.-J. Stereodivergent synthesis through catalytic asymmetric reversed hydroboration. J. Am. Chem. Soc. 141, 4670–4677 (2019).
Shoba, V. M., Thacker, N. C., Bochat, A. J. & Takacs, J. M. Synthesis of chiral tertiary boronic esters by oxime-directed catalytic asymmetric hydroboration. Angew. Chem. Int. Ed. 55, 1465–1469 (2016).
Chakrabarty, S. & Takacs, J. M. Synthesis of chiral tertiary boronic esters: phosphonate-directed catalytic asymmetric hydroboration of trisubstituted alkenes. J. Am. Chem. Soc. 139, 6066–6069 (2017).
Brown, A. N., Zakharov, L. N., Mikulas, T., Dixon, D. A. & Liu, S.-Y. Rhodium-catalyzed B-H activation of 1,2-azaborines: synthesis and characterization of BN isosteres of stilbenes. Org. Lett. 16, 3340–3343 (2014).
DeFrancesco, H., Dudley, J. & Coca, A. in Boron Reagents in Synthesis (ed. Coca, A.) Ch. 1 (American Chemical Society, 2016).
Lee, J., McDonald, R. & Hall, D. Enantioselective preparation and chemoselective cross-coupling of 1,1-diboron compounds. Nat. Chem. 3, 894–899 (2011).
Sun, C., Potter, B. & Morken, J. P. A catalytic enantiotopic-group-selective Suzuki reaction for the construction of chiral organoboronates. J. Am. Chem. Soc. 136, 6534–6537 (2014).
Hong, K., Liu, X. & Morken, J. P. Simple access to elusive α-boryl carbanions and their alkylation: an umpolung construction for organic synthesis. J. Am. Chem. Soc. 136, 10581–10584 (2014).
Potter, B., Szymaniak, A. A., Edelstein, E. K. & Morken, J. P. Nonracemic allylic boronates through enantiotopic-group-selective cross-coupling of geminal bis(boronates) and vinyl halides. J. Am. Chem. Soc. 136, 17918–17921 (2014).
Ma, S. Some typical advances in the synthetic applications of allenes. Chem. Rev. 105, 2829–2872 (2005).
Rivera-Fuentes, P. & Diederich, F. Allenes in molecular materials. Angew. Chem. Int. Ed. 51, 2818–2828 (2012).
Neff, R. K. & Frantz, D. E. Recent advances in the catalytic syntheses of allenes: a critical assessment. ACS Catal. 4, 519–528 (2014).
Huang, X. & Ma, S. Allenation of terminal alkynes with aldehydes and ketones. Acc. Chem. Res. 52, 1301–1312 (2019).
Yu, K.-L. et al. Retinoic acid receptor β,γ-selective ligands: synthesis and biological activity of 6-substituted 2-naphthoic acid retinoids. J. Med. Chem. 39, 2411–2421 (1996).
Yonova, I. M. et al. Stereospecific nickel-catalyzed cross-coupling reactions of alkyl Grignard reagents and identification of selective anti-breast-cancer agents. Angew. Chem. Int. Ed. 53, 2422–2427 (2014).
Taylor, B. L. H., Swift, E. C., Waetzig, J. D. & Jarvo, E. R. Stereospecific nickel-catalyzed cross-coupling reactions of alkyl ethers: enantioselective synthesis of diarylethanes. J. Am. Chem. Soc. 133, 389–391 (2011).
Larouche-Gauthier, R., Elford, T. G. & Aggarwal, V. K. Ate complexes of secondary boronic esters as chiral organometallic-type nucleophiles for asymmetric synthesis. J. Am. Chem. Soc. 133, 16794–16797 (2011).
Xi, Y. & Hartwig, J. F. Mechanistic studies of copper-catalyzed asymmetric hydroboration of alkenes. J. Am. Chem. Soc. 139, 12758–12772 (2017).
Acknowledgements
Financial support from the National Natural Science Foundation of China (22271048 and 22001038 to K.Y.; 21931013 and 22271105 to Q.S.), the Natural Science Foundation of Fujian Province (2022J05016 to K.Y.; 2022J02009 to Q.S.), Fuzhou University (510578 to Q.S.), Guangdong Provincial Key Laboratory of Catalysis (2020B121201002 to P.Y.) and Shenzhen Science and Technology Program (KQTD20210811090112004 to P.Y.) is gratefully acknowledged. Computational work was supported by the Center for Computational Science and Engineering and the CHEM High-Performance Supercomputer Cluster (CHEM-HPC) of the Department of Chemistry, Southern University of Science and Technology.
Author information
Authors and Affiliations
Contributions
Q.S. and K.Y. conceived and directed the project. W.S., J.Z., X.Z. and W.Q. performed experiments. W.S. prepared the Supplementary Information. P.Y. and Y.C. performed the DFT calculations and drafted the DFT parts. Q.S., P.Y. and K.Y. wrote the paper. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemistry thanks Bi-Jie Li, Ying He and the other, anonymous, reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Tables 1–13, Figs. 1–12 and starting material preparation, experimental procedures, synthetic transformations, mechanistic studies and product characterization.
Supplementary Data 1
Crystallographic data of compound 3b, CCDC reference 2245401.
Supplementary Data 2
Contains the many computational structures used in this study to generate potential energy surfaces. The ‘Opt_Freq’ folder includes the key sections in the output files of structure optimizations and frequency calculations. The ‘Quasiharmonic’ folder includes the output files formed by Truhlar’s quasiharmonic correction. The ‘Solvation_SPE’ folder includes output files obtained by performing single point energy calculations using the solvation model. All these files are included in the zipped file.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Su, W., Zhu, J., Chen, Y. et al. Copper-catalysed asymmetric hydroboration of alkenes with 1,2-benzazaborines to access chiral naphthalene isosteres. Nat. Chem. (2024). https://doi.org/10.1038/s41557-024-01505-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41557-024-01505-0
- Springer Nature Limited