Abstract
Catalysis with engineered enzymes has provided more efficient routes for the production of active pharmaceutical agents. However, the potential of biocatalysis to assist in early-stage drug discovery campaigns remains largely untapped. In this study, we have developed a biocatalytic strategy for the construction of sp3-rich polycyclic compounds via the intramolecular cyclopropanation of benzothiophenes and related heterocycles. Two carbene transferases with complementary regioisomer selectivity were evolved to catalyse the stereoselective cyclization of benzothiophene substrates bearing diazo ester groups at the C2 or C3 position of the heterocycle. The detailed mechanisms of these reactions were elucidated by a combination of crystallographic and computational analyses. Leveraging these insights, the substrate scope of one of the biocatalysts could be expanded to include previously unreactive substrates, highlighting the value of integrating evolutionary and rational strategies to develop enzymes for new-to-nature transformations. The molecular scaffolds accessed here feature a combination of three-dimensional and stereochemical complexity with ‘rule-of-three’ properties, which should make them highly valuable for fragment-based drug discovery campaigns.






Similar content being viewed by others
Data availability
Protein crystal structure coordinates have been deposited with the Protein Data Bank (PDB) under accession numbers 7SLH (MbBTIC-C3) and 7SLI (MbBTIC-C2). Crystallographic data for the structures reported in this Article have been deposited at the Cambridge Crystallographic Data Centre under CCDC deposition numbers 2157009 (2a), 2157011 (2d), 2157007 (4a), 2157010 (4f) and 2157008 (4k). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/.
References
Bornsceuer, U. T. et al. Engineering the third wave of biocatalysis. Nature 485, 185–194 (2012).
Devine, P. N. et al. Extending the application of biocatalysis to meet the challenges of drug development. Nat. Rev. Chem. 2, 409–421 (2018).
Slagman, S. & Fessner, W. D. Biocatalytic routes to anti-viral agents and their synthetic intermediates. Chem. Soc. Rev. 50, 1968–2009 (2021).
Savile, C. K. et al. Biocatalytic asymmetric synthesis of chiral amines from ketones applied to sitagliptin manufacture. Science 329, 305–309 (2010).
Huffman, M. A. et al. Design of an in vitro biocatalytic cascade for the manufacture of islatravir. Science 366, 1255–1259 (2019).
McIntosh, J. A. et al. Engineered ribosyl-1-kinase enables concise synthesis of molnupiravir, an antiviral for COVID-19. ACS Cent. Sci. 7, 1980–1985 (2021).
Brandenberg, O. F., Fasan, R. & Arnold, F. H. Exploiting and engineering hemoproteins for abiological carbene and nitrene transfer reactions. Curr. Opin. Biotechnol. 47, 102–111 (2017).
Schwizer, F. et al. Artificial metalloenzymes: reaction scope and optimization strategies. Chem. Rev. 118, 142–231 (2018).
Ren, X. & Fasan, R. Engineered and artificial metalloenzymes for selective C–H functionalization. Curr. Opin. Green Sustain. Chem. 31, 100494 (2021).
Liu, Z. & Arnold, F. H. New-to-nature chemistry from old protein machinery: carbene and nitrene transferases. Curr. Opin. Biotechnol. 69, 43–51 (2021).
Ramsden, J. I., Cosgrove, S. C. & Turner, N. J. Is it time for biocatalysis in fragment-based drug discovery? Chem. Sci. 11, 11104–11112 (2020).
Erlanson, D. A., Fesik, S. W., Hubbard, R. E., Jahnke, W. & Jhoti, H. Twenty years on: the impact of fragments on drug discovery. Nat. Rev. Drug Discov. 15, 605–619 (2016).
Erlanson, D. A., Davis, B. J. & Jahnke, W. Fragment-based drug discovery: advancing fragments in the absence of crystal structures. Cell Chem. Biol. 26, 9–15 (2019).
Jhoti, H., Williams, G., Rees, D. C. & Murray, C. W. The ‘rule of three’ for fragment-based drug discovery: where are we now? Nat. Rev. Drug Discov. 12, 644 (2013).
Murray, C. W. & Rees, D. C. Opportunity knocks: organic chemistry for fragment-based drug discovery (FBDD). Angew. Chem. Int. Ed. 55, 488–492 (2016).
Morley, A. D. et al. Fragment-based hit identification: thinking in 3D. Drug Discov. Today 18, 1221–1227 (2013).
Over, B. et al. Natural-product-derived fragments for fragment-based ligand discovery. Nat. Chem. 5, 21–28 (2013).
Yu, B., Zheng, Y. C., Shi, X. J., Qi, P. P. & Liu, H. M. Natural product-derived spirooxindole fragments serve as privileged substructures for discovery of new anticancer agents. Anticancer Agents Med. Chem. 16, 1315–1324 (2016).
Coelho, P. S., Brustad, E. M., Kannan, A. & Arnold, F. H. Olefin cyclopropanation via carbene transfer catalyzed by engineered cytochrome P450 enzymes. Science 339, 307–310 (2013).
Bordeaux, M., Tyagi, V. & Fasan, R. Highly diastereoselective and enantioselective olefin cyclopropanation using engineered myoglobin-based catalysts. Angew. Chem. Int. Ed. 54, 1744–1748 (2015).
Tinoco, A., Steck, V., Tyagi, V. & Fasan, R. Highly diastereo- and enantioselective synthesis of trifluoromethyl-substituted cyclopropanes via myoglobin-catalyzed transfer of trifluoromethylcarbene. J. Am. Chem. Soc. 139, 5293–5296 (2017).
Key, H. M. et al. Beyond iron: iridium-containing P450 enzymes for selective cyclopropanations of structurally diverse alkenes. ACS Cent. Sci. 3, 302–308 (2017).
Chandgude, A. L. & Fasan, R. Highly diastereo- and enantioselective synthesis of nitrile-substituted cyclopropanes by myoglobin-mediated carbene transfer catalysis. Angew. Chem. Int. Ed. 57, 15852–15856 (2018).
Knight, A. M. et al. Diverse engineered heme proteins enable stereodivergent cyclopropanation of unactivated alkenes. ACS Cent. Sci. 4, 372–377 (2018).
Wittmann, B. J. et al. Diversity-oriented enzymatic synthesis of cyclopropane building blocks. ACS Catal. 10, 7112–7116 (2020).
Nam, D., Steck, V., Potenzino, R. J. & Fasan, R. A diverse library of chiral cyclopropane scaffolds via chemoenzymatic assembly and diversification of cyclopropyl ketones. J. Am. Chem. Soc. 143, 2221–2231 (2021).
Vargas, D. A., Tinoco, A., Tyagi, V. & Fasan, R. Myoglobin-catalyzed C–H functionalization of unprotected indoles. Angew. Chem. Int. Ed. 57, 9911–9915 (2018).
Brandenberg, O. F., Chen, K. & Arnold, F. H. Directed evolution of a cytochrome P450 carbene transferase for selective functionalization of cyclic compounds. J. Am. Chem. Soc. 141, 8989–8995 (2019).
Srivastava, P., Yang, H., Ellis-Guardiola, K. & Lewis, J. C. Engineering a dirhodium artificial metalloenzyme for selective olefin cyclopropanation. Nat. Commun. 6, 7789 (2015).
Sreenilayam, G., Moore, E. J., Steck, V. & Fasan, R. Metal substitution modulates the reactivity and extends the reaction scope of myoglobin carbene transfer catalysts. Adv. Synth. Catal. 359, 2076–2089 (2017).
Dydio, P. et al. An artificial metalloenzyme with the kinetics of native enzymes. Science 354, 102–106 (2016).
Ohora, K. et al. Catalytic cyclopropanation by myoglobin reconstituted with iron porphycene: acceleration of catalysis due to rapid formation of the carbene species. J. Am. Chem. Soc. 139, 17265–17268 (2017).
Villarino, L. et al. An artificial heme enzyme for cyclopropanation reactions. Angew. Chem. Int. Ed. 57, 7785–7789 (2018).
Zhao, J. M., Bachmann, D. G., Lenz, M., Gillingham, D. G. & Ward, T. R. An artificial metalloenzyme for carbene transfer based on a biotinylated dirhodium anchored within streptavidin. Catal. Sci. Technol. 8, 2294–2298 (2018).
Carminati, D. M. & Fasan, R. Stereoselective cyclopropanation of electron-deficient olefins with a cofactor redesigned carbene transferase featuring radical reactivity. ACS Catal. 9, 9683–9697 (2019).
Stenner, R., Steventon, J. W., Seddon, A. & Anderson, J. L. R. A de novo peroxidase is also a promiscuous yet stereoselective carbene transferase. Proc. Natl Acad. Sci. USA 117, 1419–1428 (2020).
Chandgude, A. L., Ren, X. & Fasan, R. Stereodivergent intramolecular cyclopropanation enabled by engineered carbene transferases. J. Am. Chem. Soc. 141, 9145–9150 (2019).
Ren, X. K., Liu, N. Y., Chandgude, A. L. & Fasan, R. An enzymatic platform for the highly enantioselective and stereodivergent construction of cyclopropyl-δ-lactones. Angew. Chem. Int. Ed. 59, 21634–21639 (2020).
Yong, K., Salim, M. & Capretta, A. Intramolecular carbenoid insertions: reactions of α-diazo ketones derived from furanyl-, thienyl-, (benzofuranyl)-, and (benzothienyl)acetic acids with rhodium(II) acetate. J. Org. Chem. 63, 9828–9833 (1998).
Padwa, A., Wisnieff, T. J. & Walsh, E. J. Synthesis of cycloalkenones via the intramolecular cyclopropanation of furanyl diazo ketones. J. Org. Chem. 51, 5036–5038 (1986).
Zhang, R. K. et al. Enzymatic assembly of carbon–carbon bonds via iron-catalysed sp3 C–H functionalization. Nature 565, 67–72 (2019).
Wei, Y., Tinoco, A., Steck, V., Fasan, R. & Zhang, Y. Cyclopropanations via heme carbenes: basic mechanism and effects of carbene substituent, protein axial ligand, and porphyrin substitution. J. Am. Chem. Soc. 140, 1649–1662 (2018).
de Visser, S. P., Ogliaro, F., Harris, N. & Shaik, S. Multi-state epoxidation of ethene by cytochrome P450: a quantum chemical study. J. Am. Chem. Soc. 123, 3037–3047 (2001).
Schroder, D., Shaik, S. & Schwarz, H. Two-state reactivity as a new concept in organometallic chemistry. Acc. Chem. Res. 33, 139–145 (2000).
Sreenilayam, G. & Fasan, R. Myoglobin-catalyzed intermolecular carbene N–H insertion with arylamine substrates. Chem. Commun. 51, 1532–1534 (2015).
Kondrashov, D. A., Zhang, W., Aranda, R. T., Stec, B. & Phillips, G. N. Jr. Sampling of the native conformational ensemble of myoglobin via structures in different crystalline environments. Proteins 70, 353–362 (2008).
Blomberg, M. R. A., Borowski, T., Himo, F., Liao, R. Z. & Siegbahn, P. E. M. Quantum chemical studies of mechanisms for metalloenzymes. Chem. Rev. 114, 3601–3658 (2014).
Himo, F. Recent trends in quantum chemical modeling of enzymatic reactions. J. Am. Chem. Soc. 139, 6780–6786 (2017).
Lind, M. E. S. & Himo, F. Quantum chemistry as a tool in asymmetric biocatalysis: limonene epoxide hydrolase test case. Angew. Chem. Int. Ed. 52, 4563–4567 (2013).
Liao, R. Z. & Thiel, W. On the effect of varying constraints in the quantum mechanics only modeling of enzymatic reactions: the case of acetylene hydratase. J. Phys. Chem. B 117, 3954–3961 (2013).
Ryan, J. et al. Transaminase triggered aza-Michael approach for the enantioselective synthesis of piperidine scaffolds. J. Am. Chem. Soc. 138, 15798–15800 (2016).
Zawodny, W. et al. Chemoenzymatic synthesis of substituted azepanes by sequential biocatalytic reduction and organolithium-mediated rearrangement. J. Am. Chem. Soc. 140, 17872–17877 (2018).
Acknowledgements
This work was supported by the US National Institutes of Health (grant no. GM098628, R.F.). R.F. acknowledges support from the Cancer Prevention and Research Institute of Texas (CPRIT RR230018) and Robert A. Welch Foundation (Chair, AT-0051). D.A.V. acknowledges support from the National Science Foundation Graduate Fellowship Program. K.N.H. and A.S. acknowledge support from the National Science Foundation (CHE-1764328). M.G.-B. acknowledges support from the Spanish Ministerio de Ciencia e Innovación (MICINN; project PID2019-111300GA-I00) and the Ramón y Cajal programme via the RYC 2020-028628-I fellowship. The authors are grateful to W. Brennessel and J. Jenkins (University of Rochester) for assistance with crystallographic analyses. MS and X-ray instrumentation at the University of Rochester are supported by the US National Science Foundation (grant nos. CHE-0946653 and CHE-1725028) and the US National Institutes of Health (grant no. S10OD030302).
Author information
Authors and Affiliations
Contributions
D.A.V., X.R., S.R. and R.F. conceived the project, designed the experiments and analysed all the experiments with guidance from R.F. K.N.H. and M.G.-B. mentored L.Z. and A.S. for MD and quantum mechanics calculations, and contributed to the writing of the mechanistic parts of the paper. D.A.V. and R.F. wrote the paper with input from all of the authors. All authors discussed the results and contributed to the final paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemistry thanks Adrian Mulholland and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
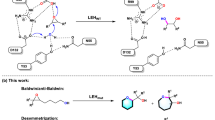
Extended Data Fig. 1 Calculated reaction pathways for C2- and C3-benzothienyl diazo ester substrates.
DFT analysis of the reaction mechanism for intramolecular cyclopropanation of C2- and C3-benzothiophenyl-methyl-diazoacetate catalyzed by a truncated iron porphyrin with an axial 4-methylimidazole ligand as a simplified model called Fe-Porphyrin. The reaction proceeds via a (1) iron−carbene formation followed by (2) cyclopropanation (2 C–C bonds). ΔG values are calculated at the B3LYP-D3BJ/def2TZVP (SMD, ε=4) // B3LYP-D3BJ/6-31G(d)+SDD (Fe) level. For each stationary point, the Gibbs free energy is provided for its lowest energy spin state. Detailed free energy profiles and additional data are provided in Supplementary Fig. 8 and Supplementary Table 5 in Supporting Information.
Supplementary information
Supplementary Information
Supplementary Information
Supplementary Data 1
Compound 2a—crystallographic information file.
Supplementary Data 2
Compound 2a—checkCIF report.
Supplementary Data 3
Compound 2d—crystallographic information file.
Supplementary Data 4
Compound 2d—checkCIF report.
Supplementary Data 5
Compound 4a—crystallographic information file.
Supplementary Data 6
Compound 4a—checkCIF report.
Supplementary Data 7
Compound 4f—crystallographic information file.
Supplementary Data 8
Compound 4f—checkCIF report.
Supplementary Data 9
Compound 4k—crystallographic information file.
Supplementary Data 10
Compound 4k—checkCIF report.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Vargas, D.A., Ren, X., Sengupta, A. et al. Biocatalytic strategy for the construction of sp3-rich polycyclic compounds from directed evolution and computational modelling. Nat. Chem. 16, 817–826 (2024). https://doi.org/10.1038/s41557-023-01435-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-023-01435-3
- Springer Nature Limited