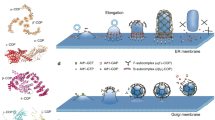
Abstract
The cyclization of peptides is a valuable strategy for the development of binding motifs to target proteins with improved affinity. Microtubules (MTs) are important targets for therapeutics, and a variety of MT-targeted drugs and peptides have recently been developed. We have previously designed a Tau-derived peptide (TP) that binds to the interior of MTs. In the present study, the development of a cyclic TP (TCP) for enhanced binding to tubulin and the stabilization of MTs are described. The fluorescently labeled cyclic peptide containing three glycine linkers (TCP3-TMR) exhibited a remarkably enhanced binding affinity to tubulin. The cyclic peptide was also demonstrated to stabilize MTs by enhancing polymerization and reducing depolymerization. Moreover, MTs were effectively formed by the treatment of cyclic peptides in the presence of guanosine triphosphate (GTP), while the linear peptide showed no such effect. These findings indicate that TCP is a useful binding motif that can stabilize MTs and is valuable for various therapeutic and material applications.







Similar content being viewed by others
References
Milroy L-G, Grossmann TN, Hennig S, Brunsveld L, Ottmann C. Modulators of protein–protein interactions. Chem Rev. 2014;114:4695.
Pelay-Gimeno M, Glas A, Koch O, Grossmann TN. Structure-based design of inhibitors of protein-protein interactions: mimicking peptide binding epitopes. Angew Chem Int Ed. 2015;54:8896.
Wójcik P, Berlicki Ł. Peptide-based inhibitors of protein–protein interactions. Bioorg Med Chem Lett. 2016;26:707.
Hamley IW. Small bioactive peptides for biomaterials design and therapeutics. Chem Rev. 2017;117:14015.
Inaba H, Matsuura K. Peptide nanomaterials designed from natural supramolecular systems. Chem Rec. 2019;19:843.
Driggers EM, Hale SP, Lee J, Terrett NK. The exploration of macrocycles for drug discovery—an underexploited structural class. Nat Rev Drug Discov. 2008;7:608.
Hill TA, Shepherd NE, Diness F, Fairlie DP. Constraining cyclic peptides to mimic protein structure motifs. Angew Chem Int Ed. 2014;53:13020.
Zorzi A, Deyle K, Heinis C. Cyclic peptide therapeutics: past, present and future. Curr Opin Chem Biol. 2017;38:24.
Vinogradov AA, Yin Y, Suga H. Macrocyclic peptides as drug candidates: recent progress and remaining challenges. J Am Chem Soc. 2019;141:4167.
Dougherty PG, Sahni A, Pei D. Understanding cell penetration of cyclic peptides. Chem Rev. 2019;119:10241.
Udugamasooriya G, Saro D, Spaller MR. Bridged peptide macrocycles as ligands for PDZ domain proteins. Org Lett. 2005;7:1203.
Jordan MA, Wilson L. Microtubules as a target for anticancer drugs. Nat Rev Cancer. 2004;4:253.
Matamoros AJ, Baas PW. Microtubules in health and degenerative disease of the nervous system. Brain Res Bull. 2016;126:217.
Fletcher DA, Mullins RD. Cell mechanics and the cytoskeleton. Nature. 2010;463:485.
Hess H, Ross JL. Non-equilibrium assembly of microtubules: from molecules to autonomous chemical robots. Chem Soc Rev. 2017;46:5570.
Xiao H, Verdier-Pinard P, Fernandez-Fuentes N, Burd B, Angeletti R, Fiser A, et al. Insights into the mechanism of microtubule stabilization by taxol. Proc Natl Acad Sci USA. 2006;103:10166.
Biswas A, Kurkute P, Saleem S, Jana B, Mohapatra S, Mondal P, et al. Novel hexapeptide interacts with tubulin and microtubules, inhibits Aβ fibrillation, and shows significant neuroprotection. ACS Chem Neurosci. 2015;6:1309.
Mondal P, Das G, Khan J, Pradhan K, Ghosh S. Crafting of neuroprotective octapeptide from taxol-binding pocket of β-tubulin. ACS Chem Neurosci. 2018;9:615.
Mondal P, Das G, Khan J, Pradhan K, Mallesh R, Saha A, et al. Potential neuroprotective peptide emerged from dual neurotherapeutic targets: a fusion approach for the development of anti-alzheimer’s lead. ACS Chem Neurosci. 2019;10:2609.
Brindisi M, Maramai S, Brogi S, Fanigliulo E, Butini S, Guarino E, et al. Development of novel cyclic peptides as pro-apoptotic agents. Eur J Med Chem. 2016;117:301.
Inaba H, Yamamoto T, Kabir AMR, Kakugo A, Sada K, Matsuura K. Molecular encapsulation inside microtubules based on Tau-derived peptides. Chem Eur J. 2018;24:14958.
Kar S, Fan J, Smith MJ, Goedert M, Amos LA. Repeat motifs of tau bind to the insides of microtubules in the absence of taxol. EMBO J. 2003;22:70.
Kadavath H, Jaremko M, Jaremko Ł, Biernat J, Mandelkow E, Zweckstetter M. Folding of the Tau protein on microtubules. Angew Chem Int Ed. 2015;54:10347.
Inaba H, Yamamoto T, Iwasaki T, Kabir AMR, Kakugo A, Sada K, et al. Fluorescent Tau-derived peptide for monitoring microtubules in living cells. ACS Omega. 2019;4:11245.
Inaba H, Yamamoto T, Iwasaki T, Kabir AMR, Kakugo A, Sada K, et al. Stabilization of microtubules by encapsulation of the GFP using a Tau-derived peptide. Chem Commun. 2019;55:9072.
Löwe J, Li H, Downing KH, Nogales E. Refined structure of αβ-tubulin at 3.5 Å resolution. J Mol Biol. 2001;313:1045.
Hipolito CJ, Suga H. Ribosomal production and in vitro selection of natural product-like peptidomimetics: the FIT and RaPID systems. Curr Opin Chem Biol. 2012;16:196.
Kamber B, Hartmann A, Eisler K, Riniker B, Rink H, Sieber P, et al. The synthesis of cystine peptides by iodine oxidation of S‐trityl‐cysteine and S‐acetamidomethyl‐cysteine peptides. Helv Chim Acta. 1980;63:899.
Jiang S, Liao C, Bindu L, Yin B, Worthy KW, Fisher RJ, et al. Discovery of thioether-bridged cyclic pentapeptides binding to Grb2-SH2 domain with high affinity. Bioorg Med Chem Lett. 2009;19:2693.
Roxin Á, Chen J, Scully CCG, Rotestein BH, Yudin AK, Zheng G. Conformational modulation of in vitro activity of cyclic RGD peptides via aziridine aldehyde-driven macrocyclization chemistry. Bioconjugate Chem. 2012;23:1387.
Hyman AA, Salser S, Drechsel DN, Unwin N, Mitchison TJ. Role of GTP hydrolysis in microtubule dynamics: information from a slowly hydrolyzable analogue, GMPCPP. Mol Biol Cell. 1992;3:1155.
Caplow M, Shanks J, Ruhlen R. How taxol modulates microtubule disassembly. J Biol Chem. 1994;269:23399.
Alushin GM, Lander GC, Kellogg EH, Zhang R, Baker D, Nogales E. High-resolution microtubule structures reveal the structural transitions in αβ-tubulin upon GTP hydrolysis. Cell. 2014;157:1117.
Hess H, Vogel V. Molecular shuttles based on motor proteins: active transport in synthetic environments. Rev Mol Biotechnol. 2001;82:67.
Kabir AMR, Kakugo A. Study of active self-assembly using biomolecular motors. Polym J. 2018;50:1139.
Bachand GD, Spoerke ED, Stevens MJ. Microtubule-based nanomaterials: exploiting nature’s dynamic biopolymers. Biotechnol Bioeng. 2015;112:1065.
Sato Y, Hiratsuka Y, Kawamata I, Murata S, Nomura S-IM. Micrometer-sized molecular robot changes its shape in response to signal molecules. Sci Rob. 2017;2:eaal3735.
Keya JJ, Suzuki R, Kabir AMR, Inoue D, Asanuma H, Sada K, et al. DNA-assisted swarm control in a biomolecular motor system. Nat Commun. 2018;9:453.
Matsuda K, Kabir AMR, Akamatsu N, Saito A, Ishikawa S, Matsuyama T, et al. Artificial smooth muscle model composed of hierarchically ordered microtubule asters mediated by DNA origami nanostructures. Nano Lett. 2019;19:3933.
Inoue D, Gutmann G, Nitta T, Kabir AMR, Konagaya A, Tokuraku K, et al. Adaptation of patterns of motile filaments under dynamic boundary conditions. ACS Nano. 2019;13:12452.
Acknowledgements
We thank Professor Y. Manabe and Professor K. Fukase (Osaka University) for the analysis of the peptides by LC-MS. This work was supported by KAKENHI (No. 17K14517 and 19K15699 for HI) from the Japan Society for the Promotion of Science (JSPS), the Inamori Foundation, and Konica Minolta Science and Technology Foundation for Konica Minolta Imaging Science Encouragement Award.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Inaba, H., Nagata, M., Miyake, K.J. et al. Cyclic Tau-derived peptides for stabilization of microtubules. Polym J 52, 1143–1151 (2020). https://doi.org/10.1038/s41428-020-0356-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41428-020-0356-3
- Springer Nature Limited
This article is cited by
-
Construction of functional microtubules and artificial motile systems based on peptide design
Polymer Journal (2023)
-
PJ ZEON Award for outstanding papers in Polymer Journal 2020
Polymer Journal (2021)