Abstract
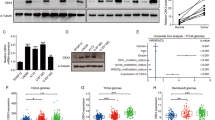
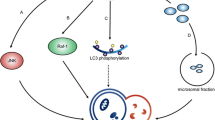
The molecular mechanism of glioblastoma (GBM) radiation resistance remains poorly understood. The aim of this study was to elucidate the potential role of Melanophilin (MLPH) O-GlcNAcylation and the specific mechanism through which it regulates GBM radiotherapy resistance. We found that MLPH was significantly upregulated in recurrent GBM tumor tissues after ionizing radiation (IR). MLPH induced radiotherapy resistance in GBM cells and xenotransplanted human tumors through regulating the NF-κB pathway. MLPH was O-GlcNAcylated at the conserved serine 510, and radiation-resistant GBM cells showed higher levels of O-GlcNAcylation of MLPH. O-GlcNAcylation of MLPH protected its protein stability and tripartite motif containing 21(TRIM21) was identified as an E3 ubiquitin ligase promoting MLPH degradation whose interaction with MLPH was affected by O-GlcNAcylation. Our data demonstrate that MLPH exerts regulatory functions in GBM radiation resistance by promoting the NF-κB signaling pathway and that O-GlcNAcylation of MLPH both stabilizes and protects it from TRIM21-mediated ubiquitination. These results identify a potential mechanism of GBM radiation resistance and suggest a potential therapeutic strategy for GBM treatment.








Similar content being viewed by others
Data availability
All data needed to evaluate the conclusions in the paper are present in the paper and/or the Supplementary Materials. Additional data related to this paper may be requested from the authors.
Change history
21 May 2024
A Correction to this paper has been published: https://doi.org/10.1038/s41388-024-03045-w
References
Miao Z, Tian W, Ye Y, Gu W, Bao Z, Xu L, et al. Hsp90 induces Acsl4-dependent glioma ferroptosis via dephosphorylating Ser637 at Drp1. Cell Death Dis. 2022;13:548.
Wei Y, Lu C, Zhou P, Zhao L, Lyu X, Yin J, et al. EIF4A3-induced circular RNA ASAP1 promotes tumorigenesis and temozolomide resistance of glioblastoma via NRAS/MEK1/ERK1-2 signaling. Neuro Oncol. 2021;23:611–24.
Tu Y, Xu L, Xu J, Bao Z, Tian W, Ye Y, et al. Loss of deubiquitylase USP2 triggers development of glioblastoma via TGF-beta signaling. Oncogene. 2022;41:2597–608.
He Y, Dong XH, Zhu Q, Xu YL, Chen ML, Liu Z. Ultrasound-triggered microbubble destruction enhances the radiosensitivity of glioblastoma by inhibiting PGRMC1-mediated autophagy in vitro and in vivo. Mil Med Res. 2022;9:9.
Gu J, Mu N, Jia B, Guo Q, Pan L, Zhu M, et al. Targeting radiation-tolerant persister cells as a strategy for inhibiting radioresistance and recurrence in glioblastoma. Neuro Oncol. 2022;24:1056–70.
Tu Y, Chen Z, Zhao P, Sun G, Bao Z, Chao H, et al. Smoothened Promotes Glioblastoma Radiation Resistance Via Activating USP3-Mediated Claspin Deubiquitination. Clin Cancer Res. 2020;26:1749–62.
Lee JA, Hwang SJ, Hong SC, Myung CH, Lee JE, Park JI, et al. Identification of MicroRNA Targeting Mlph and Affecting Melanosome Transport. Biomolecules. 2019;9.
Abba MC, Sun H, Hawkins KA, Drake JA, Hu Y, Nunez MI, et al. Breast cancer molecular signatures as determined by SAGE: correlation with lymph node status. Mol Cancer Res. 2007;5:881–90.
Orgaz JL, Benguria A, Sanchez-Martinez C, Ladhani O, Volpert OV, Jimenez B. Changes in the gene expression profile of A375 human melanoma cells induced by overexpression of multifunctional pigment epithelium-derived factor. Melanoma Res. 2011;21:285–97.
Zhang T, Sun Y, Zheng T, Wang R, Jia D, Zhang W. MLPH Accelerates the Epithelial-Mesenchymal Transition in Prostate Cancer. Onco Targets Ther. 2020;13:701–8.
Li WS, Chen CI, Chen HP, Liu KW, Tsai CJ, Yang CC. Overexpression of MLPH in Rectal Cancer Patients Correlates with a Poorer Response to Preoperative Chemoradiotherapy and Reduced Patient Survival. Diagnostics (Basel). 2021;11.
Yu F, Zhang Q, Liu H, Liu J, Yang S, Luo X, et al. Dynamic O-GlcNAcylation coordinates ferritinophagy and mitophagy to activate ferroptosis. Cell Discovery. 2022;8.
Song T, Zou Q, Yan Y, Lv S, Li N, Zhao X, et al. DOT1L O-GlcNAcylation promotes its protein stability and MLL-fusion leukemia cell proliferation. Cell Rep. 2021;36:109739.
Chu Y, Jiang M, Wu N, Xu B, Li W, Liu H, et al. O-GlcNAcylation of SIX1 enhances its stability and promotes Hepatocellular Carcinoma Proliferation. Theranostics. 2020;10:9830–42.
Liu YY, Liu HY, Yu TJ, Lu Q, Zhang FL, Liu GY, et al. O-GlcNAcylation of MORC2 at threonine 556 by OGT couples TGF-beta signaling to breast cancer progression. Cell Death Differ. 2022;29:861–73.
Shi Q, Shen Q, Liu Y, Shi Y, Huang W, Wang X, et al. Increased glucose metabolism in TAMs fuels O-GlcNAcylation of lysosomal Cathepsin B to promote cancer metastasis and chemoresistance. Cancer Cell. 2022;40:1207–22.e10.
Oliveira-Nunes MC, Juliao G, Menezes A, Mariath F, Hanover JA, Evaristo JAM, et al. O-GlcNAcylation protein disruption by Thiamet G promotes changes on the GBM U87-MG cells secretome molecular signature. Clin Proteom. 2021;18:14.
Ciraku L, Bacigalupa ZA, Ju J, Moeller RA, Le Minh G, Lee RH, et al. O-GlcNAc transferase regulates glioblastoma acetate metabolism via regulation of CDK5-dependent ACSS2 phosphorylation. Oncogene. 2022;41:2122–36.
Jin Y, Zhang Y, Li B, Zhang J, Dong Z, Hu X, et al. TRIM21 mediates ubiquitination of Snail and modulates epithelial to mesenchymal transition in breast cancer cells. Int J Biol Macromol. 2019;124:846–53.
Sabile A, Meyer AM, Wirbelauer C, Hess D, Kogel U, Scheffner M, et al. Regulation of p27 degradation and S-phase progression by Ro52 RING finger protein. Mol Cell Biol. 2006;26:5994–6004.
Ding Q, He D, He K, Zhang Q, Tang M, Dai J, et al. Downregulation of TRIM21 contributes to hepatocellular carcinoma carcinogenesis and indicates poor prognosis of cancers. Tumour Biol. 2015;36:8761–72.
Kobayashi-Nakamura K, Kudo M, Naito K. Rhamnazin suppresses melanosome transport by promoting the ubiquitin-mediated proteasomal degradation of melanophilin. J Dermatol Sci. 2022;105:45–54.
Magne N, Toillon RA, Bottero V, Didelot C, Houtte PV, Gerard JP, et al. NF-kappaB modulation and ionizing radiation: mechanisms and future directions for cancer treatment. Cancer Lett. 2006;231:158–68.
Fan Y, Mao R, Yang J. NF-kappaB and STAT3 signaling pathways collaboratively link inflammation to cancer. Protein Cell. 2013;4:176–85.
Chen Z, Wang HW, Wang S, Fan L, Feng S, Cai X, et al. USP9X deubiquitinates ALDH1A3 and maintains mesenchymal identity in glioblastoma stem cells. J Clin Invest. 2019;129:2043–55.
Stackhouse CT, Anderson JC, Yue Z, Nguyen T, Eustace NJ, Langford CP, et al. An in vivo model of glioblastoma radiation resistance identifies long noncoding RNAs and targetable kinases. JCI Insight. 2022;7.
Zhu GD, Yu J, Sun ZY, Chen Y, Zheng HM, Lin ML, et al. Genome-wide CRISPR/Cas9 screening identifies CARHSP1 responsible for radiation resistance in glioblastoma. Cell Death Dis. 2021;12:724.
Liu J, Sareddy GR, Zhou M, Viswanadhapalli S, Li X, Lai Z, et al. Differential Effects of Estrogen Receptor beta Isoforms on Glioblastoma Progression. Cancer Res. 2018;78:3176–89.
Zhou M, Sareddy GR, Li M, Liu J, Luo Y, Venkata PP, et al. Estrogen receptor beta enhances chemotherapy response of GBM cells by down regulating DNA damage response pathways. Sci Rep. 2019;9:6124.
Chen J, Dong X, Cheng X, Zhu Q, Zhang J, Li Q, et al. Ogt controls neural stem/progenitor cell pool and adult neurogenesis through modulating Notch signaling. Cell Rep. 2021;34:108905.
Efimova EV, Appelbe OK, Ricco N, Lee SS, Liu Y, Wolfgeher DJ, et al. O-GlcNAcylation Enhances Double-Strand Break Repair, Promotes Cancer Cell Proliferation, and Prevents Therapy-Induced Senescence in Irradiated Tumors. Mol Cancer Res. 2019;17:1338–50.
Very N, El Yazidi-Belkoura I. Targeting O-GlcNAcylation to overcome resistance to anti-cancer therapies. Front Oncol. 2022;12:960312.
Ilchovska DD, Barrow DM. An Overview of the NF-kB mechanism of pathophysiology in rheumatoid arthritis, investigation of the NF-kB ligand RANKL and related nutritional interventions. Autoimmun Rev. 2021;20:102741.
Ren C, Han X, Lu C, Yang T, Qiao P, Sun Y, et al. Ubiquitination of NF-kappaB p65 by FBXW2 suppresses breast cancer stemness, tumorigenesis, and paclitaxel resistance. Cell Death Differ. 2022;29:381–92.
Ahmed KM, Li JJ. NF-kappa B-mediated adaptive resistance to ionizing radiation. Free Radic Biol Med. 2008;44:1–13.
Funding
This study was supported by grants from the National Natural Science Foundation of China (82120108018 and 81972153); China Postdoctoral Science Foundation (2021M701495); Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD, Grant No. JX10231803); Gusu School, Nanjing Medical University (GSKY202201010); Natural Science Foundation of Jiangsu Province for Distinguished Young Scholars (BK20220727); National Natural Science Youth Fund of China (82303835).
Author information
Authors and Affiliations
Contributions
Conception and design: JJ, YT and LX; development of methodology: JJ, YT and LX; acquisition of data: LX, ZT and YY; analysis and interpretation of data: LX, TW, PZ, WG, BG, LZ, WC, XW and YT; writing the manuscript: LX; administrative, technical, or material support: JJ, YT and LX. All authors read and approved the final paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics
The human brain tissues were obtained from the Department of Neurosurgery at the First Affiliated Hospital of Nanjing Medical University, and was approved by the Institutional Review Board (2021-SR-202). The animal study was reviewed and approved by Nanjing Medical University’ s Institutional Animal Care and Use Committee (IACUC).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xu, L., Ye, Y., Tao, Z. et al. O-GlcNAcylation of melanophilin enhances radiation resistance in glioblastoma via suppressing TRIM21 mediated ubiquitination. Oncogene 43, 61–75 (2024). https://doi.org/10.1038/s41388-023-02881-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-023-02881-6
- Springer Nature Limited