Abstract
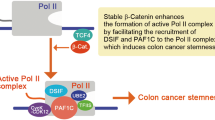
P21-activated kinase 1 (PAK1) is associated with colon cancer progression and metastasis, whereas the molecular mechanism remains elusive. Here, we show that downregulation of PAK1 in colon cancer cells reduces total β-catenin level, as well as cell proliferation. Mechanistically, PAK1 directly phosphorylates β-catenin proteins at Ser675 site and this leads to more stable and transcriptional active β-catenin. Corroborating these results, PAK1 is required for full Wnt signaling, and superactivation of β-catenin is achieved by simultaneous knockdown of adenomatous polyposis coli protein and activation of PAK1. Moreover, we show that Rac1 functions upstream of PAK1 in colon cancer cells and contributes to β-catenin phosphorylation and accumulation. We conclude that a Rac1/PAK1 cascade controls β-catenin S675 phosphorylation and full activation in colon cancer cells. Supporting this conclusion, overexpression of PAK1 is observed in 70% of colon cancer samples and is correlated with massive β-catenin accumulation.







Similar content being viewed by others
References
Anderson CB, Neufeld KL, White RL . (2002). Subcellular distribution of Wnt pathway proteins in normal and neoplastic colon. Proc Natl Acad Sci USA 99: 8683–8688.
Arias-Romero LE, Chernoff J . (2008). A tale of two Paks. Biol Cell 100: 97–108.
Balasenthil S, Sahin AA, Barnes CJ, Wang RA, Pestell RG, Vadlamudi RK et al. (2004). p21-activated kinase-1 signaling mediates cyclin D1 expression in mammary epithelial and cancer cells. J Biol Chem 279: 1422–1428.
Bienz M, Clevers H . (2000). Linking colorectal cancer to Wnt signaling. Cell 103: 311–320.
Bikkavilli RK, Feigin ME, Malbon CC . (2008). G alpha o mediates WNT-JNK signaling through dishevelled 1 and 3, RhoA family members, and MEKK 1 and 4 in mammalian cells. J Cell Sci 121: 234–245.
Billin AN, Thirlwell H, Ayer DE . (2000). Beta-catenin-histone deacetylase interactions regulate the transition of LEF1 from a transcriptional repressor to an activator. Mol Cell Biol 20: 6882–6890.
Bokoch GM . (2003). Biology of the p21-activated kinases. Annu Rev Biochem 72: 743–781.
Carter JH, Douglass LE, Deddens JA, Colligan BM, Bhatt TR, Pemberton JO et al. (2004). Pak-1 expression increases with progression of colorectal carcinomas to metastasis. Clin Cancer Res 10: 3448–3456.
Clevers H . (2006). Wnt/beta-catenin signaling in development and disease. Cell 127: 469–480.
Deacon SW, Beeser A, Fukui JA, Rennefahrt UE, Myers C, Chernoff J et al. (2008). An isoform-selective, small-molecule inhibitor targets the autoregulatory mechanism of p21-activated kinase. Chem Biol 15: 322–331.
Dummler B, Ohshiro K, Kumar R, Field J . (2009). Pak protein kinases and their role in cancer. Cancer Metastasis Rev 28: 51–63.
Esufali S, Bapat B . (2004). Cross-talk between Rac1 GTPase and dysregulated Wnt signaling pathway leads to cellular redistribution of beta-catenin and TCF/LEF-mediated transcriptional activation. Oncogene 23: 8260–8271.
Fang D, Hawke D, Zheng Y, Xia Y, Meisenhelder J, Nika H et al. (2007). Phosphorylation of beta-catenin by AKT promotes beta-catenin transcriptional activity. J Biol Chem 282: 11221–11229.
Galisteo ML, Chernoff J, Su YC, Skolnik EY, Schlessinger J . (1996). The adaptor protein Nck links receptor tyrosine kinases with the serine-threonine kinase Pak1. J Biol Chem 271: 20997–21000.
Giles RH, van Es JH, Clevers H . (2003). Caught up in a Wnt storm: Wnt signaling in cancer. Biochim Biophys Acta 1653: 1–24.
Habas R, Dawid IB, He X . (2003). Coactivation of Rac and Rho by Wnt/frizzled signaling is required for vertebrate gastrulation. Genes Dev 17: 295–309.
He H, Shulkes A, Baldwin GS . (2008). PAK1 interacts with beta-catenin and is required for the regulation of the beta-catenin signalling pathway by gastrins. Biochim Biophys Acta 1783: 1943–1954.
Hendriksen J, Jansen M, Brown CM, van der Velde H, van Ham M, Galjart N et al. (2008). Plasma membrane recruitment of dephosphorylated beta-catenin upon activation of the Wnt pathway. J Cell Sci 121: 1793–1802.
Hino S, Tanji C, Nakayama KI, Kikuchi A . (2005). Phosphorylation of beta-catenin by cyclic AMP-dependent protein kinase stabilizes beta-catenin through inhibition of its ubiquitination. Mol Cell Biol 25: 9063–9072.
Huynh N, Liu KH, Baldwin GS, He H . (2010). P21-activated kinase 1 stimulates colon cancer cell growth and migration/invasion via ERK- and AKT-dependent pathways. Biochim Biophys Acta 1803: 1106–1113.
Ilyas M, Tomlinson IP, Rowan A, Pignatelli M, Bodmer WF . (1997). Beta-catenin mutations in cell lines established from human colorectal cancers. Proc Natl Acad Sci USA 94: 10330–10334.
Jacobs T, Causeret F, Nishimura YV, Terao M, Norman A, Hoshino M et al. (2007). Localized activation of p21-activated kinase controls neuronal polarity and morphology. J Neurosci 27: 8604–8615.
Korinek V, Barker N, Morin PJ, van Wichen D, de Weger R, Kinzler KW et al. (1997). Constitutive transcriptional activation by a beta-catenin-Tcf complex in APC−/− colon carcinoma. Science 275: 1784–1787.
Kumar R, Gururaj AE, Barnes CJ . (2006). p21-activated kinases in cancer. Nat Rev Cancer 6: 459–471.
Lee SH, Kunz J, Lin SH, Yu-Lee LY . (2007). 16-kDa prolactin inhibits endothelial cell migration by down-regulating the Ras-Tiam1-Rac1-Pak1 signaling pathway. Cancer Res 67: 11045–11053.
Logan CY, Nusse R . (2004). The Wnt signaling pathway in development and disease. Annu Rev Cell Dev Biol 20: 781–810.
Lu W, Katz S, Gupta R, Mayer BJ . (1997). Activation of Pak by membrane localization mediated by an SH3 domain from the adaptor protein Nck. Curr Biol 7: 85–94.
Lu Z, Hunter T . (2004). Wnt-independent beta-catenin transactivation in tumor development. Cell Cycle 3: 571–573.
MacDonald BT, Tamai K, He X . (2009). Wnt/beta-catenin signaling: components, mechanisms, and diseases. Dev Cell 17: 9–26.
Manser E, Leung T, Salihuddin H, Zhao ZS, Lim L . (1994). A brain serine/threonine protein kinase activated by Cdc42 and Rac1. Nature 367: 40–46.
Molli PR, Li DQ, Murray BW, Rayala SK, Kumar R . (2009). PAK signaling in oncogenesis. Oncogene 28: 2545–2555.
Moon RT, Kohn AD, De Ferrari GV, Kaykas A . (2004). WNT and beta-catenin signalling: diseases and therapies. Nat Rev Genet 5: 691–701.
Morin PJ, Sparks AB, Korinek V, Barker N, Clevers H, Vogelstein B et al. (1997). Activation of beta-catenin-Tcf signaling in colon cancer by mutations in beta-catenin or APC. Science 275: 1787–1790.
Mosimann C, Hausmann G, Basler K . (2009). Beta-catenin hits chromatin: regulation of Wnt target gene activation. Nat Rev Mol Cell Biol 10: 276–286.
Nheu T, He H, Hirokawa Y, Walker F, Wood J, Maruta H . (2004). PAK is essential for RAS-induced upregulation of cyclin D1 during the G1 to S transition. Cell Cycle 3: 71–74.
Phelps RA, Chidester S, Dehghanizadeh S, Phelps J, Sandoval IT, Rai K et al. (2009). A two-step model for colon adenoma initiation and progression caused by APC loss. Cell 137: 623–634.
Polakis P . (2000). Wnt signaling and cancer. Genes Dev 14: 1837–1851.
Polakis P . (2002). Casein kinase 1: a Wnt'er of disconnect. Curr Biol 12: R499–R501.
Rennefahrt UE, Deacon SW, Parker SA, Devarajan K, Beeser A, Chernoff J et al. (2007). Specificity profiling of Pak kinases allows identification of novel phosphorylation sites. J Biol Chem 282: 15667–15678.
Rong R, Surace EI, Haipek CA, Gutmann DH, Ye K . (2004). Serine 518 phosphorylation modulates merlin intramolecular association and binding to critical effectors important for NF2 growth suppression. Oncogene 23: 8447–8454.
Segditsas S, Sieber OM, Rowan A, Setien F, Neale K, Phillips RK et al. (2008). Promoter hypermethylation leads to decreased APC mRNA expression in familial polyposis and sporadic colorectal tumours, but does not substitute for truncating mutations. Exp Mol Pathol 85: 201–206.
Sun J, Khalid S, Rozakis-Adcock M, Fantus IG, Jin T . (2009). P-21-activated protein kinase-1 functions as a linker between insulin and Wnt signaling pathways in the intestine. Oncogene 28: 3132–3144.
Tang Y, Chen Z, Ambrose D, Liu J, Gibbs JB, Chernoff J et al. (1997). Kinase-deficient Pak1 mutants inhibit Ras transformation of Rat-1 fibroblasts. Mol Cell Biol 17: 4454–4464.
Taurin S, Sandbo N, Qin Y, Browning D, Dulin NO . (2006). Phosphorylation of beta-catenin by cyclic AMP-dependent protein kinase. J Biol Chem 281: 9971–9976.
Valentini AM, Pirrelli M, Renna L, Armentano R, Caruso ML . (2003). P53 and beta-catenin in colorectal cancer progression. Curr Pharm Des 9: 1932–1936.
van de Wetering M, Oving I, Muncan V, Pon Fong MT, Brantjes H, van Leenen D et al. (2003). Specific inhibition of gene expression using a stably integrated, inducible small-interfering-RNA vector. EMBO Rep 4: 609–615.
van de Wetering M, Sancho E, Verweij C, de Lau W, Oving I, Hurlstone A et al. (2002). The beta-catenin/TCF-4 complex imposes a crypt progenitor phenotype on colorectal cancer cells. Cell 111: 241–250.
Wang RA, Mazumdar A, Vadlamudi RK, Kumar R . (2002). P21-activated kinase-1 phosphorylates and transactivates estrogen receptor-alpha and promotes hyperplasia in mammary epithelium. EMBO J 21: 5437–5447.
Wang Y, Fu Y, Gao L, Zhu G, Liang J, Gao C et al. (2010a). Xenopus skip modulates Wnt/beta-catenin signaling and functions in neural crest induction. J Biol Chem 285: 10890–10901.
Wang Z, Pedersen E, Basse A, Lefever T, Peyrollier K, Kapoor S et al. (2010b). Rac1 is crucial for Ras-dependent skin tumor formation by controlling Pak1-Mek-Erk hyperactivation and hyperproliferation in vivo. Oncogene 29: 3362–3373.
Wells CM, Jones GE . (2010). The emerging importance of group II PAKs. Biochem J 425: 465–473.
Wu X, Tu X, Joeng KS, Hilton MJ, Williams DA, Long F . (2008). Rac1 activation controls nuclear localization of beta-catenin during canonical Wnt signaling. Cell 133: 340–353.
Xiao GH, Beeser A, Chernoff J, Testa JR . (2002). p21-activated kinase links Rac/Cdc42 signaling to merlin. J Biol Chem 277: 883–886.
Xing Y, Clements WK, Le Trong I, Hinds TR, Stenkamp R, Kimelman D et al. (2004). Crystal structure of a beta-catenin/APC complex reveals a critical role for APC phosphorylation in APC function. Mol Cell 15: 523–533.
Acknowledgements
We thank Drs Ye-Guang Chen, Jonathan Chernoff, Raymond Habas, Alan Hall, Tianhui Hu, Edward Leof, Chaojun Li, Randy Moon, Christof Niehrs, Roel Nusse and Thomas Rudel for reagents. This work was supported by grants to WW from the National Natural Science Foundation of China (No. 30730048, 30921004), the Major Science Programs of China (2006CB943402, 2011CB943803), Tsinghua University Initiative Scientific Research Program (2010THZ0) and Bayer Healthcare.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Zhu, G., Wang, Y., Huang, B. et al. A Rac1/PAK1 cascade controls β-catenin activation in colon cancer cells. Oncogene 31, 1001–1012 (2012). https://doi.org/10.1038/onc.2011.294
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.294
- Springer Nature Limited
Keywords
This article is cited by
-
Loss of Pkd1 limits susceptibility to colitis and colorectal cancer
Oncogenesis (2023)
-
LncRNA MALAT1 promotes growth and metastasis of head and neck squamous cell carcinoma by repressing VHL through a non-canonical function of EZH2
Cell Death & Disease (2023)
-
Rac1 activation in oral squamous cell carcinoma as a predictive factor associated with lymph node metastasis
International Journal of Clinical Oncology (2023)
-
Comprehensive analysis of the prognostic implications and functional exploration of PAK gene family in human cancer
Cancer Cell International (2022)
-
Long non-coding RNA LBX2-AS1 predicts poor survival of colon cancer patients and promotes its progression via regulating miR-627-5p/RAC1/PI3K/AKT pathway
Human Cell (2022)