Abstract
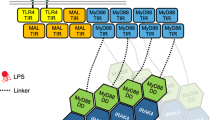
Toll-like receptor (TLR) signaling is a key innate immunity response to pathogens. Recruitment of signaling adapters such as MAL (TIRAP) and MyD88 to the TLRs requires Toll/interleukin-1 receptor (TIR)-domain interactions, which remain structurally elusive. Here we show that MAL TIR domains spontaneously and reversibly form filaments in vitro. They also form cofilaments with TLR4 TIR domains and induce formation of MyD88 assemblies. A 7-Å-resolution cryo-EM structure reveals a stable MAL protofilament consisting of two parallel strands of TIR-domain subunits in a BB-loop-mediated head-to-tail arrangement. Interface residues that are important for the interaction are conserved among different TIR domains. Although large filaments of TLR4, MAL or MyD88 are unlikely to form during cellular signaling, structure-guided mutagenesis, combined with in vivo interaction assays, demonstrated that the MAL interactions defined within the filament represent a template for a conserved mode of TIR-domain interaction involved in both TLR and interleukin-1 receptor signaling.








Similar content being viewed by others
Change history
23 August 2017
In the version of this article initially published online, an incorrect reference to Figure 3g was introduced on page 7 of the article pdf, in section 'Modeled TLR4TIR-TLR4TIR, MALTIR-TLR4TIR and TRAMTIR-TRAMTIR interactions', line 5. The correct reference is to Supplementary Figure 5c. The error has been corrected in the PDF and HTML versions of this article.
References
Akira, S., Uematsu, S. & Takeuchi, O. Pathogen recognition and innate immunity. Cell 124, 783–801 (2006).
Ve, T., Gay, N.J., Mansell, A., Kobe, B. & Kellie, S. Adaptors in toll-like receptor signaling and their potential as therapeutic targets. Curr. Drug Targets 13, 1360–1374 (2012).
Gay, N.J., Symmons, M.F., Gangloff, M. & Bryant, C.E. Assembly and localization of Toll-like receptor signalling complexes. Nat. Rev. Immunol. 14, 546–558 (2014).
Hennessy, E.J., Parker, A.E. & O'Neill, L.A. Targeting Toll-like receptors: emerging therapeutics? Nat. Rev. Drug Discov. 9, 293–307 (2010).
Lin, S.C., Lo, Y.C. & Wu, H. Helical assembly in the MyD88–IRAK4–IRAK2 complex in TLR/IL-1R signalling. Nature 465, 885–890 (2010).
Ferrao, R. et al. IRAK4 dimerization and trans-autophosphorylation are induced by Myddosome assembly. Mol. Cell 55, 891–903 (2014).
Bonham, K.S. et al. A promiscuous lipid-binding protein diversifies the subcellular sites of toll-like receptor signal transduction. Cell 156, 705–716 (2014).
Xu, Y. et al. Structural basis for signal transduction by the Toll/interleukin-1 receptor domains. Nature 408, 111–115 (2000).
Ve, T., Williams, S.J. & Kobe, B. Structure and function of Toll/interleukin-1 receptor/resistance protein (TIR) domains. Apoptosis 20, 250–261 (2015).
Poltorak, A. et al. Defective LPS signaling in C3H/HeJ and C57BL/10ScCr mice: mutations in Tlr4 gene. Science 282, 2085–2088 (1998).
Ozinsky, A. et al. The repertoire for pattern recognition of pathogens by the innate immune system is defined by cooperation between toll-like receptors. Proc. Natl. Acad. Sci. USA 97, 13766–13771 (2000).
Fitzgerald, K.A. et al. Mal (MyD88-adapter-like) is required for Toll-like receptor-4 signal transduction. Nature 413, 78–83 (2001).
Oshiumi, H. et al. TIR-containing adapter molecule (TICAM)-2, a bridging adapter recruiting to toll-like receptor 4 TICAM-1 that induces interferon-beta. J. Biol. Chem. 278, 49751–49762 (2003).
Fekonja, O., Bencˇina, M. & Jerala, R. Toll/interleukin-1 receptor domain dimers as the platform for activation and enhanced inhibition of Toll-like receptor signaling. J. Biol. Chem. 287, 30993–31002 (2012).
Nagpal, K. et al. Natural loss-of-function mutation of myeloid differentiation protein 88 disrupts its ability to form Myddosomes. J. Biol. Chem. 286, 11875–11882 (2011).
Into, T., Inomata, M., Niida, S., Murakami, Y. & Shibata, K. Regulation of MyD88 aggregation and the MyD88-dependent signaling pathway by sequestosome 1 and histone deacetylase 6. J. Biol. Chem. 285, 35759–35769 (2010).
Sester, D.P. et al. A novel flow cytometric method to assess inflammasome formation. J. Immunol. 194, 455–462 (2015).
Hughes, M.M. et al. Solution structure of the TLR adaptor MAL/TIRAP reveals an intact BB loop and supports MAL Cys91 glutathionylation for signaling. Proc. Natl. Acad. Sci. USA (in the press).
Valkov, E. et al. Crystal structure of Toll-like receptor adaptor MAL/TIRAP reveals the molecular basis for signal transduction and disease protection. Proc. Natl. Acad. Sci. USA 108, 14879–14884 (2011).
Kagan, J.C. & Medzhitov, R. Phosphoinositide-mediated adaptor recruitment controls Toll-like receptor signaling. Cell 125, 943–955 (2006).
Lin, Z., Lu, J., Zhou, W. & Shen, Y. Structural insights into TIR domain specificity of the bridging adaptor Mal in TLR4 signaling. PLoS One 7, e34202 (2012).
Bovijn, C. et al. Identification of binding sites for myeloid differentiation primary response gene 88 (MyD88) and Toll-like receptor 4 in MyD88 adapter-like (Mal). J. Biol. Chem. 288, 12054–12066 (2013).
Loiarro, M. et al. Mutational analysis identifies residues crucial for homodimerization of myeloid differentiation factor 88 (MyD88) and for its function in immune cells. J. Biol. Chem. 288, 30210–30222 (2013).
Vyncke, L. et al. Reconstructing the TIR side of the Myddosome: a paradigm for TIR-TIR interactions. Structure 24, 437–447 (2016).
Bovijn, C. et al. Identification of interaction sites for dimerization and adapter recruitment in Toll/interleukin-1 receptor (TIR) domain of Toll-like receptor 4. J. Biol. Chem. 287, 4088–4098 (2012).
Matsunaga, N., Tsuchimori, N., Matsumoto, T. & Ii, M. TAK-242 (resatorvid), a small-molecule inhibitor of Toll-like receptor (TLR) 4 signaling, binds selectively to TLR4 and interferes with interactions between TLR4 and its adaptor molecules. Mol. Pharmacol. 79, 34–41 (2011).
Enokizono, Y. et al. Structures and interface mapping of the TIR domain-containing adaptor molecules involved in interferon signaling. Proc. Natl. Acad. Sci. USA 110, 19908–19913 (2013).
Stylianou, E. et al. Interleukin 1 induces NF-kappa B through its type I but not its type II receptor in lymphocytes. J. Biol. Chem. 267, 15836–15841 (1992).
Gioannini, T.L. & Weiss, J.P. Regulation of interactions of Gram-negative bacterial endotoxins with mammalian cells. Immunol. Res. 39, 249–260 (2007).
Cheng, Z., Taylor, B., Ourthiague, D.R. & Hoffmann, A. Distinct single-cell signaling characteristics are conferred by the MyD88 and TRIF pathways during TLR4 activation. Sci. Signal. 8, ra69 (2015).
Wu, H. & Fuxreiter, M. The structure and dynamics of higher-order assemblies: amyloids, signalosomes, and granules. Cell 165, 1055–1066 (2016).
von Bernuth, H. et al. Pyogenic bacterial infections in humans with MyD88 deficiency. Science 321, 691–696 (2008).
Ngo, V.N. et al. Oncogenically active MYD88 mutations in human lymphoma. Nature 470, 115–119 (2011).
Avbelj, M. et al. Activation of lymphoma-associated MyD88 mutations via allostery-induced TIR-domain oligomerization. Blood 124, 3896–3904 (2014).
Ogus, A.C. et al. The Arg753GLn polymorphism of the human toll-like receptor 2 gene in tuberculosis disease. Eur. Respir. J. 23, 219–223 (2004).
Guven-Maiorov, E. et al. The architecture of the TIR domain signalosome in the Toll-like receptor-4 signaling pathway. Sci. Rep. 5, 13128 (2015).
Khan, J.A., Brint, E.K., O'Neill, L.A. & Tong, L. Crystal structure of the Toll/interleukin-1 receptor domain of human IL-1RAPL. J. Biol. Chem. 279, 31664–31670 (2004).
Núñez Miguel, R. et al. A dimer of the Toll-like receptor 4 cytoplasmic domain provides a specific scaffold for the recruitment of signalling adaptor proteins. PLoS One 2, e788 (2007).
Ohnishi, H. et al. Structural basis for the multiple interactions of the MyD88 TIR domain in TLR4 signaling. Proc. Natl. Acad. Sci. USA 106, 10260–10265 (2009).
Tao, X., Xu, Y., Zheng, Y., Beg, A.A. & Tong, L. An extensively associated dimer in the structure of the C713S mutant of the TIR domain of human TLR2. Biochem. Biophys. Res. Commun. 299, 216–221 (2002).
Toshchakov, V.Y., Szmacinski, H., Couture, L.A., Lakowicz, J.R. & Vogel, S.N. Targeting TLR4 signaling by TLR4 Toll/IL-1 receptor domain-derived decoy peptides: identification of the TLR4 Toll/IL-1 receptor domain dimerization interface. J. Immunol. 186, 4819–4827 (2011).
Nimma, S., Ve, T., Williams, S.J. & Kobe, B. Towards the structure of the TIR-domain signalosome. Curr. Opin. Struct. Biol. 43, 122–130 (2017).
Alaidarous, M. et al. Mechanism of bacterial interference with TLR4 signaling by Brucella Toll/interleukin-1 receptor domain-containing protein TcpB. J. Biol. Chem. 289, 654–668 (2014).
Williams, S.J. et al. Structural basis for assembly and function of a heterodimeric plant immune receptor. Science 344, 299–303 (2014).
Kaplan-Türköz, B. et al. Structure of the Toll/interleukin 1 receptor (TIR) domain of the immunosuppressive Brucella effector BtpA/Btp1/TcpB. FEBS Lett. 587, 3412–3416 (2013).
Snyder, G.A. et al. Crystal structures of the Toll/Interleukin-1 receptor (TIR) domains from the Brucella protein TcpB and host adaptor TIRAP reveal mechanisms of molecular mimicry. J. Biol. Chem. 289, 669–679 (2014).
Stols, L. et al. A new vector for high-throughput, ligation-independent cloning encoding a tobacco etch virus protease cleavage site. Protein Expr. Purif. 25, 8–15 (2002).
Darmanin, C. et al. Protein crystal screening and characterization for serial femtosecond nanocrystallography. Sci. Rep. 6, 25345 (2016).
Hoover, D.M. & Lubkowski, J. DNAWorks: an automated method for designing oligonucleotides for PCR-based gene synthesis. Nucleic Acids Res. 30, e43 (2002).
Stemmer, W.P., Crameri, A., Ha, K.D., Brennan, T.M. & Heyneker, H.L. Single-step assembly of a gene and entire plasmid from large numbers of oligodeoxyribonucleotides. Gene 164, 49–53 (1995).
Mindell, J.A. & Grigorieff, N. Accurate determination of local defocus and specimen tilt in electron microscopy. J. Struct. Biol. 142, 334–347 (2003).
Tang, G. et al. EMAN2: an extensible image processing suite for electron microscopy. J. Struct. Biol. 157, 38–46 (2007).
Frank, J. et al. SPIDER and WEB: processing and visualization of images in 3D electron microscopy and related fields. J. Struct. Biol. 116, 190–199 (1996).
Egelman, E.H. A robust algorithm for the reconstruction of helical filaments using single-particle methods. Ultramicroscopy 85, 225–234 (2000).
Pettersen, E.F. et al. UCSF Chimera: a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Croll, T.I. et al. Higher-resolution structure of the human insulin receptor ectodomain: multi-modal inclusion of the insert domain. Structure 24, 469–476 (2016).
Humphrey, W., Dalke, A. & Schulten, K. VMD: visual molecular dynamics. J. Mol. Graph 14, 33–38 (1996).
DiMaio, F. et al. Atomic-accuracy models from 4.5-Å cryo-electron microscopy data with density-guided iterative local refinement. Nat. Methods 12, 361–365 (2015).
Song, Y. et al. High-resolution comparative modeling with RosettaCM. Structure 21, 1735–1742 (2013).
Gipson, B., Zeng, X., Zhang, Z.Y. & Stahlberg, H. 2dx: user-friendly image processing for 2D crystals. J. Struct. Biol. 157, 64–72 (2007).
Holm, L. & Rosenström, P. Dali server: conservation mapping in 3D. Nucleic Acids Res. 38, W545–W549 (2010).
Krissinel, E. & Henrick, K. Inference of macromolecular assemblies from crystalline state. J. Mol. Biol. 372, 774–797 (2007).
Krissinel, E. & Henrick, K. Secondary-structure matching (SSM), a new tool for fast protein structure alignment in three dimensions. Acta Crystallogr. D Biol. Crystallogr. 60, 2256–2268 (2004).
Webb, B. & Sali, A. Comparative protein structure modeling using MODELLER. Curr. Protoc. Bioinformatics 47, 5.6.1–5.6.32 (2014).
Edgar, R.C. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 32, 1792–1797 (2004).
Robert, X. & Gouet, P. Deciphering key features in protein structures with the new ENDscript server. Nucleic Acids Res. 42, W320–W324 (2014).
George, J. et al. MyD88 adaptor-like D96N is a naturally occurring loss-of-function variant of TIRAP. J. Immunol. 184, 3025–3032 (2010).
Vajjhala, P.R. et al. The inflammasome adaptor ASC induces procaspase-8 death effector domain filaments. J. Biol. Chem. 290, 29217–29230 (2015).
Mureev, S., Kovtun, O., Nguyen, U.T. & Alexandrov, K. Species-independent translational leaders facilitate cell-free expression. Nat. Biotechnol. 27, 747–752 (2009).
Gambin, Y. et al. Confocal spectroscopy to study dimerization, oligomerization and aggregation of proteins: a practical guide. Int. J. Mol. Sci. 17, E655 (2016).
Sierecki, E. et al. Rapid mapping of interactions between human SNX-BAR proteins measured in vitro by AlphaScreen and single-molecule spectroscopy. Mol. Cell. Proteomics 13, 2233–2245 (2014).
Kovtun, O. et al. Leishmania cell-free protein expression system. Methods 55, 58–64 (2011).
Gagoski, D. et al. Gateway-compatible vectors for high-throughput protein expression in pro- and eukaryotic cell-free systems. J. Biotechnol. 195, 1–7 (2015).
Škalamera, D. et al. A high-throughput platform for lentiviral overexpression screening of the human ORFeome. PLoS One 6, e20057 (2011).
Walhout, A.J. et al. GATEWAY recombinational cloning: application to the cloning of large numbers of open reading frames or ORFeomes. Methods Enzymol. 328, 575–592 (2000).
Gambin, Y. et al. Single-molecule analysis reveals self assembly and nanoscale segregation of two distinct cavin subcomplexes on caveolae. eLife 3, e01434 (2013).
Acknowledgements
We thank T. Monie (Department of Biochemistry, University of Cambridge) for providing the TLR4TIR expression construct, D. Brown and J. Stow for early cell work, and the members of B.K.'s laboratory for discussion. This work was supported by the National Health and Medical Research Council (NHMRC grants 1003326 and 1107804 to B.K., A.M., M.M. and T.V.; 1071659 to B.K.; and 1108859 to Y.G., E.S. and T.V.) and NIH grant EB001567 (to E.H.E.). B.K. is supported as an NHMRC Principal Research Fellow (grants 1003325 and 1110971), and K.J.S. is supported as an NHMRC Senior Research Fellow (grant 1059729).
Author information
Authors and Affiliations
Contributions
T.V., P.R.V., E.S., Y.G., M.J.L., K.J.S., E.H.E. and B.K. designed the research. T.V., P.R.V., A.H., S.H., X.Y., P.L., Z.H., G.P.M., M.M. and M.J.L. performed the experiments. T.C. and F.D.M. contributed tools. T.V., P.R.V., T.C., F.D.M., E.S., Y.G., M.J.L., K.J.S., E.H.E. and B.K. analyzed the data. T.V. and B.K. wrote the paper, and all authors edited and contributed to writing the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 MAL forms filaments in solution
a, Negative-stain EM images of MALTIR at 60 μM, 52.5 μM, 45 μM, and 30 μM. b, SDS-PAGE precipitation analysis of MALTIR and MALFL. SF, soluble fraction; IF, insoluble fraction. c-d, Turbidity assays of MALTIR (c) and MALFL (d) at 30°C. MAL polymerization could be quantified by following the increase in absorbance at 350 nm as a function of time. Typically, with 60 μM MALTIR at 30°C, an initial lag time of 10 min was observed, followed by a rapid increase in turbidity that approached saturation after approximately 20 min. This suggests that MAL-filament growth proceeds through small early assembly intermediates, for which disassembly is energetically favored over assembly. After a sufficiently large oligomer assembles, MAL-filament growth becomes energetically favorable and proceeds rapidly. The increase in turbidity correlated well with the observation of filaments by negative-stain EM. The critical concentration for MALTIR-filament formation is estimated to be 50-60 μM. e-f, Effect of NaCl (e) and pH (f) on MALTIR filament formation. The original image of the gel in (b) can be found in Supplementary Data Set 1.
Supplementary Figure 2 Single molecule fluorescence analyses of MALFL and MALTIR, and analyses of MALTIR interaction with TLR3TIR
a-b, MALFL and MALTIR tagged with GFP were expressed in LTE and fluorescence time traces were acquired. Representative traces are shown. The trace for MALFL shows the presence of larger peaks (>4000 photons per ms or cpms) compared to MALTIR (peaks at 2000 cpms). c, Schematic diagram of the seeding assay. d, mCherry MALFL was used to create the seeds, which were added to a solution of monomeric GFP MALFL. Formation of MALFL polymers was detected immediately after addition of seeds and increase in the intensities of the peaks upon time indicate polymer elongation. e, The ragged profile of the peak in the insert suggests that the objects are fibrillar. f, As a control for the TLR4TIR-MALTIR interaction, we incubated equal amounts of TLR3TIR with MALTIR (60 μM, 30 °C, 1 hour). TLR3 only engages the TRIF-dependent pathway, which does not require MAL. SDS-PAGE analysis revealed that both proteins were present in the insoluble fraction but similar amounts of TLR3 was observed in the TLR3TIR control sample without MAL, suggesting that co-precipitation of TLR3TIR and MALTIR is not due to formation of co-filaments. g, Supporting this conclusion, SDS-PAGE analyses of TLR3TIR and MALTIR incubated at a concentration below the critical concentration for MALTIR oligomerization (30 μM of each, 30°C for 1 h) only revealed the presence of a faint MALTIR band in the insoluble fraction. Results in (f-g) are representative of three experiments. h, Negative-stain EM images of TLR3TIR + MALTIR, 60 μM of each, and TLR3TIR (60 μM). Ordered assemblies were not observed in the TLR3TIR samples, while only the MALTIR filaments were observed in the TLR3TIR:MALTIR samples. Original gel images in (f-g) can be found in Supplementary Data Set 1.
Supplementary Figure 3 MALTIR induced formation of MyD88TIR assemblies, and single molecule fluorescence analyses of MyD88FL and MyD88TIR
a, SDS-PAGE analyses of the soluble fraction (SF) and insoluble fraction (IF) after incubation of MALTIR (60 μM) + MyD88TIR (60 μM) at 30°C for 1 hour. b, Negative-stain EM images of MALTIR + MyD88TIR, 60 μM of each. c, Turbidity assay of MyD88TIR (30-80 μM) assembly formation in the presence MALTIR (2 μM). d-e, Negative-stain EM image of a MAL induced MyD88TIR assembly (d), and the corresponding fast Fourier transform analysis (e). f-g, MyD88FL and MyD88TIR tagged with a N-terminal GFP were expressed in LTE and 180 seconds fluorescence time traces were obtained. Representative traces for MyD88FL (f) and MyD88TIR (g) are presented. MyD88FL (f) forms numerous large objects (>10,000 cpms), whereas MyD88TIR (g) is mainly oligomeric (peaks at 4,000 cpms). h, MyD88FL N-terminally tagged with mCherry was used to create the seeds, which were added to a solution of monomeric GFP MyD88FL. i, The insert shows, in more detail, the coincidence between the mCherry and the GFP channels. Note that the difference of intensities between the two channels indicates that the GFP monomers are elongating the mCherry seeds rather than just coating them. The original gel image in (a) can be found in Supplementary Data Set 1.
Supplementary Figure 4 Development of a flow cytometry-based assay to quantify MAL-induced clustering of MyD88 in HEK293 cells.
a, HEK293 cells were transfected with a plasmid expressing V5-tagged MyD88FL alone or with a plasmid expressing Myc-tagged MALFL. Cells were immuno-stained with antibodies to V5 and Myc tags (green and red, respectively) and the nuclei stained with DAPI (blue). Representative images are from an experiment performed twice with 1-3 images captured. b-d, Analysis of MyD88 clustering by flow cytometry. b, Plots of MyD88 (V5) stain vs. MAL (Myc) stain for cells transfected with empty vector or with plasmids expressing V5-tagged MyD88FL or Myc-tagged MALFL either together or alone. Cells were immuno-stained and analyzed by flow cytometry. Quadrants representing the MyD88-positive, MAL-positive or double-positive populations are labeled in the panel with empty vector alone. Gates to select broad-expression-window or low-expressing MyD88 cells are indicated in the panels with MyD88. c, Plots of fluorescence pulse height vs. area for the MyD88 (V5) signal for cells expressing MyD88 alone or with MAL that were gated to obtain cells with low or broad level expression of MyD88 as indicated in (b). In each plot, cells with an elevated height-to-area ratio are boxed and represent the percentage of cells with a MyD88 cluster. d, Gating of cells for Fig. 1i. Transfected cells were immuno-stained and analysed by flow cytometry. Cells expressing a low level of MyD88 were first selected (green gate) and within this gate, cells expressing MALFL or MALTIR at a similar expression level were selected (red gate). Clustering of MyD88 was quantified, within the red gate for samples expressing MAL and MyD88 or within the green gate for samples expressing MyD88 alone, as described in (c).
Supplementary Figure 5 Power spectra analyses, model-map comparison, and structural analyses of the MALTIR filament
a, Comparison of the observed versus the calculated power spectra, annotated with Bessel order assignments: on the left is the power spectrum from the projection of the reconstruction, while on the right is the averaged power spectrum from the image segments. b, FSC between the fitted model and the cryoEM map. c, Superposition of the inner MALTIR subunit (cyan) and the TIR domain of TLR1 (PDB ID 1FYV), TLR2 (PDB ID 1FYW), TLR6 (PDB ID 4OM7), TLR10 (PDB ID 2J67), IL-1RAPL (PDB ID 1T3G), MyD88 (PDB ID 2Z5V), MyD88 (PDB ID 4EO7), TRAM (PDB ID 2M1W), TRIF (PDB ID 2M1X) and TRR2 (PDB ID 4W8G).
Supplementary Figure 6 Structural comparisons of the MALTIR filament with the crystal structures of TRR-2
a, Comparison of the intrastrand and interstrand interactions in the MALTIR proto-filament (teal) with TIR:TIR interfaces in the TRR-2TIR (PDB ID 4W8G) crystal structure (violet). b, TIR:TIR interactions in crystal form II of the TRR-2 TIR domain (PDB ID 4W8H). c, Structure-based sequence alignment of MALTIR and TRR-2 (PDB ID 4W8G). The alignment was formatted using ESPript. Green and purple circles indicated residues involved in intrastrand, or interstrand TIR:TIR interactions, respectively. Strictly conserved residues are indicated in white letters with a red box and similar residues are indicated in red letters with a blue frame.
Supplementary Figure 7 MALTIR proto-filament interactions and structure-based mutagenesis
a, Detailed interactions in the MALTIR proto-filament:proto-filament interface. b, Precipitation analysis of MALTIR W156A Y159A double mutant, and W156A Y159A L165A triple mutant (100 μM incubated at 30°C for 12 hours). The soluble and insoluble fraction of each mutant was analyzed by 15% SDS PAGE. c, Negative-stain EM analysis of MALTIR alanine mutants. d, Cartoon representation of a MALTIR subunit (outer strand). Residues mutated to alanine (stick representation) are highlighted in green (intrastrand interface), orange (interstrand interface), blue (interface between proto-filaments) and magenta (surface exposed residues outside the interaction interfaces of the filament). The original image of gel in (b) can be found in Supplementary Data Set 1.
Supplementary Figure 8 Modeling, cryoEM analysis, and structure-based mutagenesis of MyD88TIR assemblies, and modeling of TRAMTIR:TRAMTIR interactions.
a, Cryo-EM image of a MyD88TIR assembly. b, Power spectrum calculated from the raw image in (a). Spots extending beyond 10 Å resolution can be observed in the power spectrum. c, Overall size of a intrastrand MALTIR dimer in the proto-filament. The MyD88TIR assemblies were found to have unit cell dimensions of approximately a = 60 Å b = 30 Å, γ = 105, which are similar to the dimensions of a single interstrand MALTIR dimer as arranged in the MALTIR proto-filament. d, Sequence alignment of MALTIR and MyD88TIR. Strictly conserved residues are indicated in white letters with a red box and similar residues are indicated in red letters with a blue frame. e, Negative-stain EM images of MyD88TIR mutants. The scale bar is indicated on the images. f, Turbidity assays of MyD88TIR mutants. The assays were performed in triplicate (blue, green, and red lines). g-h, Superposition of the inner MALTIR subunit (cyan) with TRAMTIR (light-orange, PDB ID 2M1W). Both the monomer (g) and the proto-filament of MALTIR are displayed (h). i, Model of TRAMTIR intrastrand and interstrand interactions using the MALTIR proto-filament as a template.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8, Supplementary Tables 1–3 and Supplementary Note. (PDF 2506 kb)
Supplementary Data Set 1
Original gel images for Fig. 1c,d,f, Fig .5a, Fig. 7a, Supplementary Fig. 1b, Supplementary Fig. 3a,b,d, and Supplementary Fig. 7b. (PDF 2076 kb)
Rights and permissions
About this article
Cite this article
Ve, T., Vajjhala, P., Hedger, A. et al. Structural basis of TIR-domain-assembly formation in MAL- and MyD88-dependent TLR4 signaling. Nat Struct Mol Biol 24, 743–751 (2017). https://doi.org/10.1038/nsmb.3444
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.3444
- Springer Nature America, Inc.
This article is cited by
-
Target ssDNA activates the NADase activity of prokaryotic SPARTA immune system
Nature Chemical Biology (2024)
-
Nucleic-acid-triggered NADase activation of a short prokaryotic Argonaute
Nature (2024)
-
Auto-inhibition and activation of a short Argonaute-associated TIR-APAZ defense system
Nature Chemical Biology (2024)
-
TLR3 forms a laterally aligned multimeric complex along double-stranded RNA for efficient signal transduction
Nature Communications (2023)
-
Emodin Alleviates the Th17/Treg Imbalance by Targeting Myd88/NF-κB Signaling in Subacute Thyroiditis
Revista Brasileira de Farmacognosia (2023)