Abstract
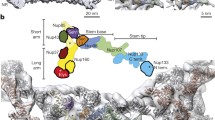
The nuclear pore complex mediates nucleocytoplasmic transport in all eukaryotes and is among the largest cellular assemblies of proteins, collectively known as nucleoporins. Nucleoporins are organized into distinct subcomplexes. We optimized the isolation of a putative membrane-coating subcomplex of the nuclear pore complex, the heptameric Nup84 complex, and analyzed its structure by EM. Our data confirmed the previously reported 'Y' shape. We discerned additional structural details, including specific hinge regions at which the particle shows great flexibility. We determined the three-dimensional structures of two conformers, mapped the localization of two nucleoporins within the subcomplex and docked known crystal structures into the EM maps. The free ends of the Y-shaped particle are formed by β-propellers; the connecting segments consist of α-solenoids. Notably, the same organizational principle is found in the clathrin triskelion, which may share a common evolutionary origin with the heptameric complex.






Similar content being viewed by others
Change history
14 June 2009
In the version of the supplementary information originally posted online, the captions for the Supplementary Movies were in the incorrect order. This has been corrected as of 14 June 2009.
References
Lim, R.Y., Ullman, K.S. & Fahrenkrog, B. Biology and biophysics of the nuclear pore complex and its components. Int. Rev. Cell Mol. Biol. 267, 299–342 (2008).
Beck, M. et al. Nuclear pore complex structure and dynamics revealed by cryoelectron tomography. Science 306, 1387–1390 (2004).
Beck, M., Lucić, V., Förster, F., Baumeister, W. & Medalia, O. Snapshots of nuclear pore complexes in action captured by cryo-electron tomography. Nature 449, 611–615 (2007).
Melcák, I., Hoelz, A. & Blobel, G. Structure of Nup58/45 suggests flexible nuclear pore diameter by intermolecular sliding. Science 315, 1729–1732 (2007).
Boehmer, T., Jeudy, S., Berke, I.C. & Schwartz, T.U. Structural and functional studies of Nup107/Nup133 interaction and its implications for the architecture of the nuclear pore complex. Mol. Cell 30, 721–731 (2008).
Debler, E.W. et al. A fence-like coat for the nuclear pore membrane. Mol. Cell 32, 815–826 (2008).
Glavy, J.S. et al. Cell-cycle-dependent phosphorylation of the nuclear pore Nup107–160 subcomplex. Proc. Natl. Acad. Sci. USA 104, 3811–3816 (2007).
Belgareh, N. et al. An evolutionarily conserved NPC subcomplex, which redistributes in part to kinetochores in mammalian cells. J. Cell Biol. 154, 1147–1160 (2001).
Loïodice, I. et al. The entire Nup107–160 complex, including three new members, is targeted as one entity to kinetochores in mitosis. Mol. Biol. Cell 15, 3333–3344 (2004).
Walther, T.C. et al. The conserved Nup107–160 complex is critical for nuclear pore complex assembly. Cell 113, 195–206 (2003).
Harel, A. et al. Removal of a single pore subcomplex results in vertebrate nuclei devoid of nuclear pores. Mol. Cell 11, 853–864 (2003).
D'Angelo, M.A., Anderson, D.J., Richard, E. & Hetzer, M.W. Nuclear pores form de novo from both sides of the nuclear envelope. Science 312, 440–443 (2006).
Orjalo, A.V. et al. The Nup107–160 nucleoporin complex is required for correct bipolar spindle assembly. Mol. Biol. Cell 17, 3806–3818 (2006).
Zuccolo, M. et al. The human Nup107–160 nuclear pore subcomplex contributes to proper kinetochore functions. EMBO J. 26, 1853–1864 (2007).
Siniossoglou, S. et al. Structure and assembly of the Nup84p complex. J. Cell Biol. 149, 41–54 (2000).
Lutzmann, M., Kunze, R., Buerer, A., Aebi, U. & Hurt, E. Modular self-assembly of a Y-shaped multiprotein complex from seven nucleoporins. EMBO J. 21, 387–397 (2002).
Brohawn, S.G., Leksa, N.C., Spear, E.D., Rajashankar, K.R. & Schwartz, T.U. Structural evidence for common ancestry of the nuclear pore complex and vesicle coats. Science 322, 1369–1373 (2008).
Hsia, K.C., Stavropoulos, P., Blobel, G. & Hoelz, A. Architecture of a coat for the nuclear pore membrane. Cell 131, 1313–1326 (2007).
Devos, D. et al. Components of coated vesicles and nuclear pore complexes share a common molecular architecture. PLoS Biol. 2, e380 (2004).
Blobel, G. Intracellular protein topogenesis. Proc. Natl. Acad. Sci. USA 77, 1496–1500 (1980).
Rout, M.P. et al. The yeast nuclear pore complex: composition, architecture, and transport mechanism. J. Cell Biol. 148, 635–652 (2000).
Drin, G. et al. A general amphipathic alpha-helical motif for sensing membrane curvature. Nat. Struct. Mol. Biol. 14, 138–146 (2007).
Radermacher, M., Wagenknecht, T., Verschoor, A. & Frank, J. A new 3-D reconstruction scheme applied to the 50S ribosomal subunit of E. coli. J. Microsc. 141, RP1–RP2 (1986).
Siniossoglou, S. et al. A novel complex of nucleoporins, which includes Sec13p and a Sec13p homolog, is essential for normal nuclear pores. Cell 84, 265–275 (1996).
Cristea, I.M., Williams, R., Chait, B.T. & Rout, M.P. Fluorescent proteins as proteomic probes. Mol. Cell. Proteomics 4, 1933–1941 (2005).
Lutzmann, M. et al. Reconstitution of Nup157 and Nup145N into the Nup84 complex. J. Biol. Chem. 280, 18442–18451 (2005).
Penczek, P., Radermacher, M. & Frank, J. Three-dimensional reconstruction of single particles embedded in ice. Ultramicroscopy 40, 33–53 (1992).
van Heel, M. & Frank, J. Use of multivariate statistics in analysing the images of biological macromolecules. Ultramicroscopy 6, 187–194 (1981).
Gilbert, P. Iterative methods for the three-dimensional reconstruction of an object from projections. J. Theor. Biol. 36, 105–117 (1972).
Berke, I.C., Boehmer, T., Blobel, G. & Schwartz, T.U. Structural and functional analysis of Nup133 domains reveals modular building blocks of the nuclear pore complex. J. Cell Biol. 167, 591–597 (2004).
Fotin, A. et al. Molecular model for a complete clathrin lattice from electron cryomicroscopy. Nature 432, 573–579 (2004).
Paoli, M. Protein folds propelled by diversity. Prog. Biophys. Mol. Biol. 76, 103–130 (2001).
Fath, S., Mancias, J.D., Bi, X. & Goldberg, J. Structure and organization of coat proteins in the COPII cage. Cell 129, 1325–1336 (2007).
Ferguson, M.L. et al. Clathrin triskelia show evidence of molecular flexibility. Biophys. J. 95, 1945–1955 (2008).
Akey, C.W. Structural plasticity of the nuclear pore complex. J. Mol. Biol. 248, 273–293 (1995).
Chernomordik, L.V. & Kozlov, M.M. Mechanics of membrane fusion. Nat. Struct. Mol. Biol. 15, 675–683 (2008).
Ghaemmaghami, S. et al. Global analysis of protein expression in yeast. Nature 425, 737–741 (2003).
Sheff, M.A. & Thorn, K.S. Optimized cassettes for fluorescent protein tagging in Saccharomyces cerevisiae. Yeast 21, 661–670 (2004).
Mastronarde, D.N. Automated electron microscope tomography using robust prediction of specimen movements. J. Struct. Biol. 152, 36–51 (2005).
Frank, J. et al. SPIDER and WEB: processing and visualization of images in 3D electron microscopy and related fields. J. Struct. Biol. 116, 190–199 (1996).
Ludtke, S.J., Baldwin, P.R. & Chiu, W. EMAN: semiautomated software for high-resolution single-particle reconstructions. J. Struct. Biol. 128, 82–97 (1999).
Wagenknecht, T., Frank, J., Boublik, M., Nurse, K. & Ofengand, J. Direct localization of the tRNA–anticodon interaction site on the Escherichia coli 30 S ribosomal subunit by electron microscopy and computerized image averaging. J. Mol. Biol. 203, 753–760 (1988).
Pettersen, E.F. et al. UCSF Chimera–a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Chacón, P. & Wriggers, W. Multi-resolution contour-based fitting of macromolecular structures. J. Mol. Biol. 317, 375–384 (2002).
Wriggers, W., Milligan, R.A. & McCammon, J.A. Situs: a package for docking crystal structures into low-resolution maps from electron microscopy. J. Struct. Biol. 125, 185–195 (1999).
Acknowledgements
We thank R. Diaz-Avalos, W. Rice and other staff at the New York Structural Biology Center for their support and stimulating discussions; D. King (Howard Hughes Medical Institute Mass Spectrometry Facility) for MS; E. Debler, V. Nagy, C. Atkinson and A. Mattheyses for critical reading of the manuscript; and D. Andor, K.-C. Hsia and other members of the Blobel laboratory for helpful discussions. M.K. was supported by the Howard Hughes Medical Institute Predoctoral Fellowship.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 and Supplementary Discussion (PDF 10037 kb)
Supplementary Movie 1
Overlay of final map 1 (orange) and map 1 reconstructed from the 25% best matching particles (purple), see Supplementary Discussion. (MOV 3846 kb)
Supplementary Movie 2
Overlay of final map 2 (orange) and map 2 reconstructed from the 25% best matching particles (purple), see Supplementary Discussion. (MOV 3858 kb)
Supplementary Movie 3
Crystal structures docked into map 1. The color code is the same as in Figure 6. (MOV 2028 kb)
Supplementary Movie 4
Crystal structures docked into map 2. The color code is the same as in Figure 6. (MOV 1992 kb)
Supplementary Movie 5
Crystal structures docked into map 2, detail. Blue and red spheres mark the N and C termini, respectively, which belong to crystal structures of partial proteins, in order to indicate were the protein domains that are absent from the crystal structure would originate. (MOV 2947 kb)
Rights and permissions
About this article
Cite this article
Kampmann, M., Blobel, G. Three-dimensional structure and flexibility of a membrane-coating module of the nuclear pore complex. Nat Struct Mol Biol 16, 782–788 (2009). https://doi.org/10.1038/nsmb.1618
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.1618
- Springer Nature America, Inc.
This article is cited by
-
Sigma-1 receptor chaperones rescue nucleocytoplasmic transport deficit seen in cellular and Drosophila ALS/FTD models
Nature Communications (2020)
-
The Quest for the Blueprint of the Nuclear Pore Complex
The Protein Journal (2019)
-
Resistance to Phytophthora infestans: exploring genes required for disease resistance in Solanaceae plants
Journal of General Plant Pathology (2018)
-
The nuclear pore complex: understanding its function through structural insight
Nature Reviews Molecular Cell Biology (2017)
-
Toward the atomic structure of the nuclear pore complex: when top down meets bottom up
Nature Structural & Molecular Biology (2016)