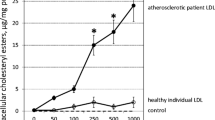
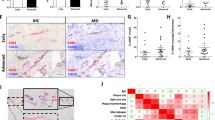
Abstract
Atherosclerosis, a hyperlipidemia-induced chronic inflammatory process of the arterial wall, develops preferentially at sites where disturbed laminar flow compromises endothelial cell (EC) function. Here we show that endothelial miR-126-5p maintains a proliferative reserve in ECs through suppression of the Notch1 inhibitor delta-like 1 homolog (Dlk1) and thereby prevents atherosclerotic lesion formation. Endothelial recovery after denudation was impaired in Mir126−/− mice because lack of miR-126-5p, but not miR-126-3p, reduced EC proliferation by derepressing Dlk1. At nonpredilection sites, high miR-126-5p levels in endothelial cells confer a proliferative reserve that compensates for the antiproliferative effects of hyperlipidemia, such that atherosclerosis was exacerbated in Mir126−/− mice. In contrast, downregulation of miR-126-5p by disturbed flow abrogated EC proliferation at predilection sites in response to hyperlipidemic stress through upregulation of Dlk1 expression. Administration of miR-126-5p rescued EC proliferation at predilection sites and limited atherosclerosis, introducing a potential therapeutic approach.






Similar content being viewed by others
Accession codes
References
Deanfield, J.E., Halcox, J.P. & Rabelink, T.J. Endothelial function and dysfunction: testing and clinical relevance. Circulation 115, 1285–1295 (2007).
Aird, W.C. Phenotypic heterogeneity of the endothelium: I. Structure, function, and mechanisms. Circ. Res. 100, 158–173 (2007).
Ando, J. & Yamamoto, K. Effects of shear stress and stretch on endothelial function. Antioxid. Redox Signal. 15, 1389–1403 (2011).
Chiu, J.J. & Chien, S. Effects of disturbed flow on vascular endothelium: pathophysiological basis and clinical perspectives. Physiol. Rev. 91, 327–387 (2011).
Sakao, S. et al. Initial apoptosis is followed by increased proliferation of apoptosis-resistant endothelial cells. FASEB J. 19, 1178–1180 (2005).
Kaiser, D., Freyberg, M.A. & Friedl, P. Lack of hemodynamic forces triggers apoptosis in vascular endothelial cells. Biochem. Biophys. Res. Commun. 231, 586–590 (1997).
Schwartz, S.M. & Benditt, E.P. Aortic endothelial cell replication. I. Effects of age and hypertension in the rat. Circ. Res. 41, 248–255 (1977).
Wright, H.P. Endothelial mitosis around aortic branches in normal guinea pigs. Nature 220, 78–79 (1968).
Foteinos, G., Hu, Y., Xiao, Q., Metzler, B. & Xu, Q. Rapid endothelial turnover in atherosclerosis-prone areas coincides with stem cell repair in apolipoprotein E–deficient mice. Circulation 117, 1856–1863 (2008).
Hansson, G.K., Chao, S., Schwartz, S.M. & Reidy, M.A. Aortic endothelial cell death and replication in normal and lipopolysaccharide-treated rats. Am. J. Pathol. 121, 123–127 (1985).
Itoh, Y., Toriumi, H., Yamada, S., Hoshino, H. & Suzuki, N. Resident endothelial cells surrounding damaged arterial endothelium reendothelialize the lesion. Arterioscler. Thromb. Vasc. Biol. 30, 1725–1732 (2010).
Weber, C. & Noels, H. Atherosclerosis: current pathogenesis and therapeutic options. Nat. Med. 17, 1410–1422 (2011).
Zhou, Z. et al. Lipoprotein-derived lysophosphatidic acid promotes atherosclerosis by releasing CXCL1 from the endothelium. Cell Metab. 13, 592–600 (2011).
Pober, J.S., Min, W. & Bradley, J.R. Mechanisms of endothelial dysfunction, injury, and death. Annu. Rev. Pathol. 4, 71–95 (2009).
Chen, C.H. et al. Oxidized low-density lipoproteins inhibit endothelial cell proliferation by suppressing basic fibroblast growth factor expression. Circulation 101, 171–177 (2000).
Neth, P., Nazari-Jahantigh, M., Schober, A. & Weber, C. MicroRNAs in flow-dependent vascular remodelling. Cardiovasc. Res. 99, 294–303 (2013).
Fish, J.E. et al. miR-126 regulates angiogenic signaling and vascular integrity. Dev. Cell 15, 272–284 (2008).
Wei, Y., Nazari-Jahantigh, M., Neth, P., Weber, C. & Schober, A. MicroRNA-126, -145, and -155: a therapeutic triad in atherosclerosis? Arterioscler. Thromb. Vasc. Biol. 33, 449–454 (2013).
Wang, S. et al. The endothelial-specific microRNA miR-126 governs vascular integrity and angiogenesis. Dev. Cell 15, 261–271 (2008).
Kuhnert, F. et al. Attribution of vascular phenotypes of the murine Egfl7 locus to the microRNA miR-126. Development 135, 3989–3993 (2008).
Zernecke, A. et al. Delivery of microRNA-126 by apoptotic bodies induces CXCL12-dependent vascular protection. Sci. Signal. 2, ra81 (2009).
Lechman, E.R. et al. Attenuation of miR-126 activity expands HSC in vivo without exhaustion. Cell Stem Cell 11, 799–811 (2012).
Baladrón, V. et al. dlk acts as a negative regulator of Notch1 activation through interactions with specific EGF-like repeats. Exp. Cell Res. 303, 343–359 (2005).
Cybulsky, M.I. et al. A major role for VCAM-1, but not ICAM-1, in early atherosclerosis. J. Clin. Invest. 107, 1255–1262 (2001).
Harris, T.A., Yamakuchi, M., Ferlito, M., Mendell, J.T. & Lowenstein, C.J. MicroRNA-126 regulates endothelial expression of vascular cell adhesion molecule 1. Proc. Natl. Acad. Sci. USA 105, 1516–1521 (2008).
Ni, C.W. et al. Discovery of novel mechanosensitive genes in vivo using mouse carotid artery endothelium exposed to disturbed flow. Blood 116, e66–e73 (2010).
Sul, H.S. Minireview: Pref-1: role in adipogenesis and mesenchymal cell fate. Mol. Endocrinol. 23, 1717–1725 (2009).
Mortensen, S.B. et al. Membrane-tethered delta-like 1 homolog (DLK1) restricts adipose tissue size by inhibiting preadipocyte proliferation. Diabetes 61, 2814–2822 (2012).
Rodríguez, P. et al. The non-canonical NOTCH ligand DLK1 exhibits a novel vascular role as a strong inhibitor of angiogenesis. Cardiovasc. Res. 93, 232–241 (2012).
Qin, L. et al. Notch1-mediated signaling regulates proliferation of porcine satellite cells (PSCs). Cell. Signal. 25, 561–569 (2013).
Nielsen, L.B. Transfer of low density lipoprotein into the arterial wall and risk of atherosclerosis. Atherosclerosis 123, 1–15 (1996).
Zeng, L. et al. Sustained activation of XBP1 splicing leads to endothelial apoptosis and atherosclerosis development in response to disturbed flow. Proc. Natl. Acad. Sci. USA 106, 8326–8331 (2009).
Civelek, M., Manduchi, E., Riley, R.J., Stoeckert, C.J. Jr. & Davies, P.F. Chronic endoplasmic reticulum stress activates unfolded protein response in arterial endothelium in regions of susceptibility to atherosclerosis. Circ. Res. 105, 453–461 (2009).
Colles, S.M., Maxson, J.M., Carlson, S.G. & Chisolm, G.M. Oxidized LDL-induced injury and apoptosis in atherosclerosis. Potential roles for oxysterols. Trends Cardiovasc. Med. 11, 131–138 (2001).
Wei, Y. et al. The microRNA-342–5p fosters inflammatory macrophage activation through an Akt1- and microRNA-155-dependent pathway during atherosclerosis. Circulation 127, 1609–1619 (2013).
Jansen, F. et al. Endothelial microparticle-mediated transfer of microRNA-126 promotes vascular endothelial cell repair via SPRED1 and is abrogated in glucose-damaged endothelial microparticles. Circulation 128, 2026–2038 (2013).
Zhou, J. et al. Regulation of vascular smooth muscle cell turnover by endothelial cell-secreted microRNA-126: role of shear stress. Circ. Res. 113, 40–51 (2013).
Fichtlscherer, S. et al. Circulating microRNAs in patients with coronary artery disease. Circ. Res. 107, 677–684 (2010).
Zampetaki, A. et al. Plasma microRNA profiling reveals loss of endothelial miR-126 and other microRNAs in type 2 diabetes. Circ. Res. 107, 810–817 (2010).
Mocharla, P. et al. AngiomiR-126 expression and secretion from circulating CD34+ and CD14+ PBMCs: role for proangiogenic effects and alterations in type 2 diabetics. Blood 121, 226–236 (2013).
Akhtar, S., Gremse, F., Kiessling, F., Weber, C. & Schober, A. CXCL12 promotes the stabilization of atherosclerotic lesions mediated by smooth muscle progenitor cells in Apoe-deficient mice. Arterioscler. Thromb. Vasc. Biol. 33, 679–686 (2013).
Schober, A., Knarren, S., Lietz, M., Lin, E.A. & Weber, C. Crucial role of stromal cell-derived factor-1α in neointima formation after vascular injury in apolipoprotein E–deficient mice. Circulation 108, 2491–2497 (2003).
Appelbe, O.K., Yevtodiyenko, A., Muniz-Talavera, H. & Schmidt, J.V. Conditional deletions refine the embryonic requirement for Dlk1. Mech. Dev. 130, 143–159 (2013).
Ehling, M., Adams, S., Benedito, R. & Adams, R.H. Notch controls retinal blood vessel maturation and quiescence. Development 140, 3051–3061 (2013).
Nazari-Jahantigh, M. et al. MicroRNA-155 promotes atherosclerosis by repressing Bcl6 in macrophages. J. Clin. Invest. 122, 4190–4202 (2012).
Nuovo, G., Lee, E.J., Lawler, S., Godlewski, J. & Schmittgen, T. In situ detection of mature microRNAs by labeled extension on ultramer templates. Biotechniques 46, 115–126 (2009).
Gremse, F. et al. Virtual elastic sphere processing enables reproducible quantification of vessel stenosis at CT and MR angiography. Radiology 260, 709–717 (2011).
Schmitt, M.M. et al. Endothelial junctional adhesion molecule-a guides monocytes into flow-dependent predilection sites of atherosclerosis. Circulation 129, 66–76 (2014).
Bidzhekov, K. et al. Rafs constitute a nodal point in the regulation of embryonic endothelial progenitor cell growth and differentiation. J. Cell. Mol. Med. 11, 1395–1407 (2007).
Acknowledgements
A.S., F.K. and C.W. are funded by the Deutsche Forschungsgemeinschaft, Germany (FOR809, WE1913/11-2 and SCHO1056/3-2). A.S. and C.W. are funded by the German Federal Ministry of Education and Research (01KU1213A) and by the German Centre for Cardiovascular Research (MHA VD1.2). M.N.-J. was funded by the Interdisciplinary Centre for Clinical Research within the Faculty of Medicine at RWTH Aachen University, Germany. We thank J. Schmidt (University of Illinois) and R.H. Adams (Max-Planck-Institute for Molecular Biomedicine) for providing Dlk1flox and Bmx-CreERT2 mice, respectively. We thank L. Natarelli, L. Pawig, J. Corbalán Campos, R. Soltan, M. Garbe, C. Geissler, P. Hartmann and S. Elbin for technical assistance.
Author information
Authors and Affiliations
Contributions
A.S. and C.W. designed the study, analyzed data and wrote the paper. M.N.-J. performed mouse experiments and collected and processed the histological and qRT-PCR data. Y.W. collected data and performed luciferase assays and mimic treatments. K.B. performed and analyzed in vitro experiments. F.G. and F.K. collected data and performed the fluorescence molecular tomography and computed tomography analyses. J.G. was involved in the analysis of human atherosclerotic lesions. R.T.M. performed the multi-photon microscopy analysis. K.H. collected data and processed the in situ hybridization and immunostaining data. H.N. performed immunoblots and in vitro experiments. M.H. performed flow cytometry analysis and cell culture experiments under flow conditions. S.W. and E.N.O. were involved in study design and contributed to the mouse experiments. A.S. and M.N.-J. contributed equally to the study. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–9 and Supplementary Table 1. (PDF 4371 kb)
Rights and permissions
About this article
Cite this article
Schober, A., Nazari-Jahantigh, M., Wei, Y. et al. MicroRNA-126-5p promotes endothelial proliferation and limits atherosclerosis by suppressing Dlk1. Nat Med 20, 368–376 (2014). https://doi.org/10.1038/nm.3487
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3487
- Springer Nature America, Inc.
This article is cited by
-
MicroRNAs and Cardiovascular Disease Risk
Current Cardiology Reports (2024)
-
The various role of microRNAs in breast cancer angiogenesis, with a special focus on novel miRNA-based delivery strategies
Cancer Cell International (2023)
-
Flow-induced reprogramming of endothelial cells in atherosclerosis
Nature Reviews Cardiology (2023)
-
The biomarkers’ landscape of post-COVID-19 patients can suggest selective clinical interventions
Scientific Reports (2023)
-
LncRNA HCG11 Accelerates Atherosclerosis via Regulating the miR-224-3p/JAK1 Axis
Biochemical Genetics (2023)