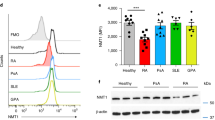
Abstract
Pathogenic T cells in individuals with rheumatoid arthritis (RA) infiltrate non-lymphoid tissue sites, maneuver through extracellular matrix and form lasting inflammatory microstructures. Here we found that RA T cells abundantly express the podosome scaffolding protein TKS5, which enables them to form tissue-invasive membrane structures. TKS5 overexpression was regulated by the intracellular metabolic environment of RA T cells—specifically, by reduced glycolytic flux that led to deficiencies in ATP and pyruvate. ATPlopyruvatelo conditions triggered fatty acid biosynthesis and the formation of cytoplasmic lipid droplets. Restoration of pyruvate production or inhibition of fatty acid synthesis corrected the tissue-invasiveness of RA T cells in vivo and reversed their proarthritogenic behavior. Thus, metabolic control of T cell locomotion provides new opportunities to interfere with T cell invasion into specific tissue sites.







Similar content being viewed by others
References
McInnes, I.B., Buckley, C.D. & Isaacs, J.D. Cytokines in rheumatoid arthritis—shaping the immunological landscape. Nat. Rev. Rheumatol. 12, 63–68 (2016).
Weyand, C.M. & Goronzy, J.J. T-cell-targeted therapies in rheumatoid arthritis. Nat. Clin. Pract. Rheumatol. 2, 201–210 (2006).
McInnes, I.B. & Schett, G. The pathogenesis of rheumatoid arthritis. N. Engl. J. Med. 365, 2205–2219 (2011).
Weyand, C.M., Fujii, H., Shao, L. & Goronzy, J.J. Rejuvenating the immune system in rheumatoid arthritis. Nat. Rev. Rheumatol. 5, 583–588 (2009).
Koetz, K. et al. T cell homeostasis in patients with rheumatoid arthritis. Proc. Natl. Acad. Sci. USA 97, 9203–9208 (2000).
Schmidt, D., Goronzy, J.J. & Weyand, C.M. CD4+CD7−CD28− T cells are expanded in rheumatoid arthritis and are characterized by autoreactivity. J. Clin. Invest. 97, 2027–2037 (1996).
Broux, B., Markovic-Plese, S., Stinissen, P. & Hellings, N. Pathogenic features of CD4+CD28− T cells in immune disorders. Trends Mol. Med. 18, 446–453 (2012).
Yang, Z., Matteson, E.L., Goronzy, J.J. & Weyand, C.M. T-cell metabolism in autoimmune disease. Arthritis Res. Ther. 17, 29 (2015).
Yang, Z. et al. Restoring oxidant signaling suppresses proarthritogenic T cell effector functions in rheumatoid arthritis. Sci. Transl. Med. 8, 331ra38 (2016).
Weyand, C.M., Yang, Z. & Goronzy, J.J. T-cell aging in rheumatoid arthritis. Curr. Opin. Rheumatol. 26, 93–100 (2014).
Yang, Z., Fujii, H., Mohan, S.V., Goronzy, J.J. & Weyand, C.M. Phosphofructokinase deficiency impairs ATP generation, autophagy, and redox balance in rheumatoid arthritis T cells. J. Exp. Med. 210, 2119–2134 (2013).
Shao, L. et al. Deficiency of the DNA repair enzyme ATM in rheumatoid arthritis. J. Exp. Med. 206, 1435–1449 (2009).
Patra, K.C. & Hay, N. The pentose phosphate pathway and cancer. Trends Biochem. Sci. 39, 347–354 (2014).
Wang, R. & Green, D.R. Metabolic checkpoints in activated T cells. Nat. Immunol. 13, 907–915 (2012).
Vander Heiden, M.G., Cantley, L.C. & Thompson, C.B. Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science 324, 1029–1033 (2009).
Menendez, J.A. & Lupu, R. Fatty acid synthase and the lipogenic phenotype in cancer pathogenesis. Nat. Rev. Cancer 7, 763–777 (2007).
Pearce, E.L., Poffenberger, M.C., Chang, C.H. & Jones, R.G. Fueling immunity: insights into metabolism and lymphocyte function. Science 342, 1242454 (2013).
Thiam, A.R., Farese, R.V. Jr. & Walther, T.C. The biophysics and cell biology of lipid droplets. Nat. Rev. Mol. Cell Biol. 14, 775–786 (2013).
van Meer, G., Voelker, D.R. & Feigenson, G.W. Membrane lipids: where they are and how they behave. Nat. Rev. Mol. Cell Biol. 9, 112–124 (2008).
Marat, A.L. & Haucke, V. Phosphatidylinositol 3-phosphates—at the interface between cell signalling and membrane traffic. EMBO J. 35, 561–579 (2016).
Wellen, K.E. & Thompson, C.B. A two-way street: reciprocal regulation of metabolism and signalling. Nat. Rev. Mol. Cell Biol. 13, 270–276 (2012).
Akram, M. Citric acid cycle and role of its intermediates in metabolism. Cell Biochem. Biophys. 68, 475–478 (2014).
Robichaud, P.P., Boulay, K., Munganyiki, J.E. & Surette, M.E. Fatty acid remodeling in cellular glycerophospholipids following the activation of human T cells. J. Lipid Res. 54, 2665–2677 (2013).
O'Sullivan, D. et al. Memory CD8+ T cells use cell-intrinsic lipolysis to support the metabolic programming necessary for development. Immunity 41, 75–88 (2014).
Zhang, X., Nakajima, T., Goronzy, J.J. & Weyand, C.M. Tissue trafficking patterns of effector memory CD4+ T cells in rheumatoid arthritis. Arthritis Rheum. 52, 3839–3849 (2005).
Carman, C.V. et al. Transcellular diapedesis is initiated by invasive podosomes. Immunity 26, 784–797 (2007).
Wolf, K., Müller, R., Borgmann, S., Bröcker, E.B. & Friedl, P. Amoeboid shape change and contact guidance: T-lymphocyte crawling through fibrillar collagen is independent of matrix remodeling by MMPs and other proteases. Blood 102, 3262–3269 (2003).
Nobes, C.D. & Hall, A. Rho GTPases control polarity, protrusion, and adhesion during cell movement. J. Cell Biol. 144, 1235–1244 (1999).
Uruno, T. et al. Activation of Arp2/3 complex-mediated actin polymerization by cortactin. Nat. Cell Biol. 3, 259–266 (2001).
Stylli, S.S. et al. Nck adaptor proteins link Tks5 to invadopodia actin regulation and ECM degradation. J. Cell Sci. 122, 2727–2740 (2009).
Thompson, O. et al. Dystroglycan, Tks5 and Src mediated assembly of podosomes in myoblasts. PLoS One 3, e3638 (2008).
Krummel, M.F., Bartumeus, F. & Gérard, A. T cell migration, search strategies and mechanisms. Nat. Rev. Immunol. 16, 193–201 (2016).
Takemura, S., Klimiuk, P.A., Braun, A., Goronzy, J.J. & Weyand, C.M. T cell activation in rheumatoid synovium is B cell dependent. J. Immunol. 167, 4710–4718 (2001).
Murphy, D.A. & Courtneidge, S.A. The 'ins' and 'outs' of podosomes and invadopodia: characteristics, formation and function. Nat. Rev. Mol. Cell Biol. 12, 413–426 (2011).
Oikawa, T. et al. Tks5-dependent formation of circumferential podosomes/invadopodia mediates cell-cell fusion. J. Cell Biol. 197, 553–568 (2012).
Seyler, T.M. et al. BLyS and APRIL in rheumatoid arthritis. J. Clin. Invest. 115, 3083–3092 (2005).
Kidani, Y. et al. Sterol regulatory element–binding proteins are essential for the metabolic programming of effector T cells and adaptive immunity. Nat. Immunol. 14, 489–499 (2013).
Walsh, M.J. et al. ML265: a potent PKM2 activator induces tetramerization and reduces tumor formation and size in a mouse xenograft model. In Probe Reports from the NIH Molecular Libraries Program (National Center for Biotechnology Information, 2010).
Shirai, T. et al. The glycolytic enzyme PKM2 bridges metabolic and inflammatory dysfunction in coronary artery disease. J. Exp. Med. 213, 337–354 (2016).
Marx, N. PPARγ and vascular inflammation: adding another piece to the puzzle. Circ. Res. 91, 373–374 (2002).
Pearce, E.L. et al. Enhancing CD8 T-cell memory by modulating fatty acid metabolism. Nature 460, 103–107 (2009).
Nobile, C. et al. HIV-1 Nef inhibits ruffles, induces filopodia, and modulates migration of infected lymphocytes. J. Virol. 84, 2282–2293 (2010).
Hwang, S.H. et al. Leukocyte-specific protein 1 regulates T-cell migration in rheumatoid arthritis. Proc. Natl. Acad. Sci. USA 112, E6535–E6543 (2015).
Buck, M.D., O'Sullivan, D. & Pearce, E.L. T cell metabolism drives immunity. J. Exp. Med. 212, 1345–1360 (2015).
Zeng, H. et al. mTORC1 couples immune signals and metabolic programming to establish Treg-cell function. Nature 499, 485–490 (2013).
Takemura, S. et al. Lymphoid neogenesis in rheumatoid synovitis. J. Immunol. 167, 1072–1080 (2001).
Nakano, A. et al. AMPK controls the speed of microtubule polymerization and directional cell migration through CLIP-170 phosphorylation. Nat. Cell Biol. 12, 583–590 (2010).
Li, Y. et al. Deficient activity of the nuclease MRE11A induces T cell aging and promotes arthritogenic effector functions in patients with rheumatoid arthritis. Immunity 45, 903–916 (2016).
Acknowledgements
This work was supported by the US National Institutes of Health (grants R01 AR042527, R01 HL 117913, R01 AI108906 and P01 HL129941 to C.M.W.; grants R01 AI108891, R01 AG045779, U19 AI057266 and I01 BX001669 to J.J.G.).
Author information
Authors and Affiliations
Contributions
C.M.W., Y.S. and J.J.G. designed the study and analyzed the data. Y.S., Z.W. and Y.L. performed experiments. Y.S. created all of the figures. E.L.M. and J.H. were responsible for subject selection, evaluation and recruitment. C.M.W., Y.S. and J.J.G. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Genetic manipulation of TKS5 expression in human T cells.
(a, b) CD4+CD45RA+ T cells from RA patients were transfected with SH3PXD2A siRNA or control siRNA on day 2 after stimulation. SH3PXD2A transcripts were quantified 24 h later in control and SH3PXD2A siRNA-transfected cells by RT-PCR. Data are from ten independent experiments. TKS5 protein expression was measured in T cells transfected with control or SH3PXD2A siRNA by flow cytometry. (c, d) CD4+CD45RA+ T cells from healthy donors were transfected with control plasmids or a flag-SH3PXD2A construct. After 24 h, transfection efficiency was monitored by qPCR (n = 14) (c) and western blotting (d). Results are given as mean ± s.e.m. **P < 0.01 (Mann-Whitney U-test).
Supplementary Figure 2 Dose–response analysis for pharmacological inhibitors.
CD4+CD45RA+ T cells were isolated from healthy individuals and stimulated in the absence or presence of the PFKFB3 inhibitor 3PO (a) or the PKM2 inhibitor Shikonin (b). CD4+CD45RA+ T cells from RA patients were incubated with C75 (c), an inhibitor of fatty acid synthase, at concentrations indicated. Expression of the SH3PXD2A gene was assessed by RT-PCR (n = 6 samples). Results are mean ± s.e.m. *P < 0.05. **P < 0.01 (Mann-Whitney U-test).
Supplementary Figure 3 Effect of metabolic interference on intracellular pyruvate, ATP and SH3PXD2A expression.
CD4+CD45RA+ T cells were stimulated and treated with pharmacologic inhibitors as in Fig. 5. ML265 or pyruvate was added to the cultures at the indicated concentrations. (a, b) Pyruvate and intracellular ATP were measured in T cells from 8 donors. Vehicle (Veh), shikonin (SK). All data are mean ± s.e.m. *P < 0.05. **P < 0.01 (paired t-test). (c, d) Dose response analysis for RA T cells (n = 5) treated with ML265 or exogenous pyruvate. SH3PXD2A transcripts were measured by RT-PCR. Data are mean ± s.e.m. (paired t-test). Non-significant (ns).
Supplementary Figure 4 Metabolic interference and induction of lipid droplets does not affect PPAR-γ expression.
CD4+CD45RA+ T cells from 3 healthy controls were stimulated and cultured with (a, b) the glycolytic flux inhibitors (3PO, 200 nM or Shikonin, SK, 250 nM). (c, d) Alternatively, glycolytic flux was enhanced in RA T-cells by treating with ML265 or adding exogenous pyruvate (ML265, 10 μM or pyruvate, PY, 1 μM) as described in Fig.5. After 3 days, PPAR-γ expression levels were assessed by flow cytometry. Data are mean ± s.e.m. from three experiments (Mann-Whitney U-test). Non-significant (ns).
Supplementary Figure 5 Knockdown of PFKFB3 expression in human T cells.
(a) CD4+CD45RA+ T cells were transfected with PFKFB3 siRNA or control siRNA on day 2 after stimulation. PFKFB3 transcripts were quantified 24 h later by RT-PCR. Data are from twelve independent experiments. Results are given as mean ± s.e.m. *P < 0.05 (Mann-Whitney U-test). (b)To test for the durability of the PFKFB3 knockdown, transcripts were quantified at 6, 24, 48, 72 and 96 h after transfection (n = 6). Data are shown as fold change compared to control siRNA.
Supplementary Figure 6 Toxicity of the FAS inhibitor C75 in vivo.
CD4+CD45RA+ T cells were isolated from healthy controls and adoptively transferred into human synovium-NSG chimeric mice. Mice were randomly assigned to one of four treatment arms and received IP injections every other day: control arm (vehicle); C75 (1 mg/kg), C75 (5 mg/kg) or C75 (10 mg/kg). Human cells in the peripheral blood and the spleens of the mice were enumerated and stained with anti-CD3, anti-CD4 and anti-CD8 ab. Frequencies of the CD3+CD4+ population were determined by flow cytometry. Data are mean ± s.e.m. from three experiments (Mann-Whitney U-teat). Non-significant (ns).
Supplementary Figure 7 Purity of CD4+CD45RA+ T cell populations.
CD4+CD45RA+ T cells were purified as described in the Methods section from 10 healthy-RA pairs. To identify memory CD4+ T cells which have reverted to a naïve phenotype, CD4+CD45RA+ T cell populations from healthy individuals and RA patients were analyzed for: (a) CD4+CD45RA+CD28+CD95+ T cells. Representative histograms from a healthy control and a RA patient. (b) CD4+CD45RA+IFN-γ+ T cells. Intracellular IFN-γ was detected by flow cytometry 3 h after stimulation with PMA (50 ng/ml) plus ionomycin (1 μg/ml). Representative data from 10 experiments.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 and Supplementary Tables 1 and 2. (PDF 1051 kb)
Rights and permissions
About this article
Cite this article
Shen, Y., Wen, Z., Li, Y. et al. Metabolic control of the scaffold protein TKS5 in tissue-invasive, proinflammatory T cells. Nat Immunol 18, 1025–1034 (2017). https://doi.org/10.1038/ni.3808
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.3808
- Springer Nature America, Inc.
This article is cited by
-
Role of T Cells in the Pathogenesis of Rheumatoid Arthritis: Focus on Immunometabolism Dysfunctions
Inflammation (2023)
-
Energiemetabolismus des Immunsystems
Zeitschrift für Rheumatologie (2023)
-
Cellular metabolic adaptations in rheumatoid arthritis and their therapeutic implications
Nature Reviews Rheumatology (2022)
-
The transcription factor RFX5 coordinates antigen-presenting function and resistance to nutrient stress in synovial macrophages
Nature Metabolism (2022)
-
Immunosenescence of T cells: a key player in rheumatoid arthritis
Inflammation Research (2022)