Abstract
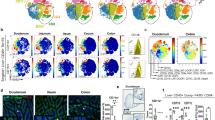
Dendritic cells (DCs) that orchestrate mucosal immunity have been studied in mice. Here we characterized human gut DC populations and defined their relationship to previously studied human and mouse DCs. CD103+Sirpα− DCs were related to human blood CD141+ DCs and to mouse intestinal CD103+CD11b− DCs and expressed markers of cross-presenting DCs. CD103+Sirpα+ DCs aligned with human blood CD1c+ DCs and mouse intestinal CD103+CD11b+ DCs and supported the induction of regulatory T cells. Both CD103+ DC subsets induced the TH17 subset of helper T cells, while CD103−Sirpα+ DCs induced the TH1 subset of helper T cells. Comparative analysis of transcriptomes revealed conserved transcriptional programs among CD103+ DC subsets and identified a selective role for the transcriptional repressors Bcl-6 and Blimp-1 in the specification of CD103+CD11b− DCs and intestinal CD103+CD11b+ DCs, respectively. Our results highlight evolutionarily conserved and divergent programming of intestinal DCs.








Similar content being viewed by others
References
Varol, C. et al. Intestinal lamina propria dendritic cell subsets have different origin and functions. Immunity 31, 502–512 (2009).
Bogunovic, M. et al. Origin of the lamina propria dendritic cell network. Immunity 31, 513–525 (2009).
Miller, J.C. et al. Deciphering the transcriptional network of the dendritic cell lineage. Nat. Immunol. 13, 888–899 (2012).
Helft, J., Ginhoux, F., Bogunovic, M. & Merad, M. Origin and functional heterogeneity of non-lymphoid tissue dendritic cells in mice. Immunol. Rev. 234, 55–75 (2010).
Uematsu, S. et al. Regulation of humoral and cellular gut immunity by lamina propria dendritic cells expressing Toll-like receptor 5. Nat. Immunol. 9, 769–776 (2008).
Kinnebrew, M.A. et al. Interleukin 23 production by intestinal CD103+CD11b+ dendritic cells in response to bacterial flagellin enhances mucosal innate immune defense. Immunity 36, 276–287 (2012).
Coombes, J.L. et al. A functionally specialized population of mucosal CD103+ DCs induces Foxp3+ regulatory T cells via a TGF-β and retinoic acid-dependent mechanism. J. Exp. Med. 204, 1757–1764 (2007).
Manicassamy, S. & Pulendran, B. Dendritic cell control of tolerogenic responses. Immunol. Rev. 241, 206–227 (2011).
Persson, E.K. et al. IRF4 Transcription-factor-dependent CD103CD11b dendritic cells drive mucosal T helper 17 cell differentiation. Immunity 38, 958–969 (2013).
Lewis, K.L. et al. Notch2 receptor signaling controls functional differentiation of dendritic cells in the spleen and intestine. Immunity 35, 780–791 (2011).
Poulin, L.F. et al. DNGR-1 is a specific and universal marker of mouse and human Batf3-dependent dendritic cells in lymphoid and nonlymphoid tissues. Blood 119, 6052–6062 (2012).
Haniffa, M. et al. Human tissues contain CD141hi cross-presenting dendritic cells with functional homology to mouse CD103+ nonlymphoid dendritic cells. Immunity 37, 60–73 (2012).
Villadangos, J.A. & Shortman, K. Found in translation: the human equivalent of mouse CD8+ dendritic cells. J. Exp. Med. 207, 1131–1134 (2010).
Schlitzer, A. et al. IRF4 rranscription factor-dependent CD11b dendritic cells in human and mouse control mucosal IL-17 cytokine responses. Immunity 38, 970–983 (2013).
Jaensson, E. et al. Small intestinal CD103+ dendritic cells display unique functional properties that are conserved between mice and humans. J. Exp. Med. 205, 2139–2149 (2008).
Mittag, D. et al. Human dendritic cell subsets from spleen and blood are similar in phenotype and function but modified by donor health status. J. Immunol. 186, 6207–6217 (2011).
Tamoutounour, S. et al. CD64 distinguishes macrophages from dendritic cells in the gut and reveals the Th1-inducing role of mesenteric lymph node macrophages during colitis. Eur. J. Immunol. 42, 3150–3166 (2012).
Denning, T.L. et al. Functional specializations of intestinal dendritic cell and macrophage subsets that control Th17 and regulatory T cell responses are dependent on the T cell/APC ratio, source of mouse strain, and regional localization. J. Immunol. 187, 733–747 (2011).
Baumgart, D.C. et al. Patients with active inflammatory bowel disease lack immature peripheral blood plasmacytoid and myeloid dendritic cells. Gut 54, 228–236 (2005).
Hadeiba, H. et al. CCR9 expression defines tolerogenic plasmacytoid dendritic cells able to suppress acute graft-versus-host disease. Nat. Immunol. 9, 1253–1260 (2008).
Cerovic, V. et al. Intestinal CD103− dendritic cells migrate in lymph and prime effector T cells. Mucosal Immunol. 6, 104–113 (2013).
Ito, T., Carson, W.F.t., Cavassani, K.A., Connett, J.M. & Kunkel, S.L. CCR6 as a mediator of immunity in the lung and gut. Exp. Cell Res. 317, 613–619 (2011).
Jang, M.H. et al. CCR7 is critically important for migration of dendritic cells in intestinal lamina propria to mesenteric lymph nodes. J. Immunol. 176, 803–810 (2006).
Takeuchi, A. et al. CRTAM confers late-stage activation of CD8+ T cells to regulate retention within lymph node. J. Immunol. 183, 4220–4228 (2009).
Kobayashi, K.S. & van den Elsen, P.J. NLRC5: a key regulator of MHC class I-dependent immune responses. Nat. Rev. Immunol. 12, 813–820 (2012).
Bouloc, A., Bagot, M., Delaire, S., Bensussan, A. & Boumsell, L. Triggering CD101 molecule on human cutaneous dendritic cells inhibits T cell proliferation via IL-10 production. Eur. J. Immunol. 30, 3132–3139 (2000).
Huber, S. et al. IL-22BP is regulated by the inflammasome and modulates tumorigenesis in the intestine. Nature 491, 259–263 (2012).
Sonnenberg, G.F., Fouser, L.A. & Artis, D. Border patrol: regulation of immunity, inflammation and tissue homeostasis at barrier surfaces by IL-22. Nat. Immunol. 12, 383–390 (2011).
Bakri, Y. et al. Balance of MafB and PU.1 specifies alternative macrophage or dendritic cell fate. Blood 105, 2707–2716 (2005).
Liu, W. et al. CD127 expression inversely correlates with FoxP3 and suppressive function of human CD4+ T reg cells. J. Exp. Med. 203, 1701–1711 (2006).
Mora, J.R. Homing imprinting and immunomodulation in the gut: role of dendritic cells and retinoids. Inflamm. Bowel Dis. 14, 275–289 (2008).
Ivanov, I.I. et al. Induction of intestinal Th17 cells by segmented filamentous bacteria. Cell 139, 485–498 (2009).
Atarashi, K. et al. ATP drives lamina propria TH17 cell differentiation. Nature 455, 808–812 (2008).
Rivollier, A., He, J., Kole, A., Valatas, V. & Kelsall, B.L. Inflammation switches the differentiation program of Ly6Chi monocytes from antiinflammatory macrophages to inflammatory dendritic cells in the colon. J. Exp. Med. 209, 139–155 (2012).
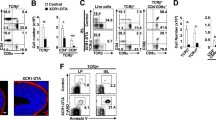
Dent, A.L., Shaffer, A.L., Yu, X., Allman, D. & Staudt, L.M. Control of inflammation, cytokine expression, and germinal center formation by BCL-6. Science 276, 589–592 (1997).
Johnston, R.J. et al. Bcl6 and Blimp-1 are reciprocal and antagonistic regulators of T follicular helper cell differentiation. Science 325, 1006–1010 (2009).
Crotty, S., Johnston, R.J. & Schoenberger, S.P. Effectors and memories: Bcl-6 and Blimp-1 in T and B lymphocyte differentiation. Nat. Immunol. 11, 114–120 (2010).
Miller, J.C. et al. Deciphering the transcriptional network of the dendritic cell lineage. Nat. Immunol. 13, 888–899 (2012).
Kim, S.J., Zou, Y.R., Goldstein, J., Reizis, B. & Diamond, B. Tolerogenic function of Blimp-1 in dendritic cells. J. Exp. Med. 208, 2193–2199 (2011).
Sancho, D. et al. Identification of a dendritic cell receptor that couples sensing of necrosis to immunity. Nature 458, 899–903 (2009).
Dorner, B.G. et al. Selective expression of the chemokine receptor XCR1 on cross-presenting dendritic cells determines cooperation with CD8+ T cells. Immunity 31, 823–833 (2009).
Galibert, L. et al. Nectin-like protein 2 defines a subset of T-cell zone dendritic cells and is a ligand for class-I-restricted T-cell-associated molecule. J. Biol. Chem. 280, 21955–21964 (2005).
Yeh, J.H., Sidhu, S.S. & Chan, A.C. Regulation of a late phase of T cell polarity and effector functions by Crtam. Cell 132, 846–859 (2008).
Sigmundsdottir, H. & Butcher, E.C. Environmental cues, dendritic cells and the programming of tissue-selective lymphocyte trafficking. Nat. Immunol. 9, 981–987 (2008).
Fujikado, N. et al. Dcir deficiency causes development of autoimmune diseases in mice due to excess expansion of dendritic cells. Nat. Med. 14, 176–180 (2008).
Smythies, L.E. et al. Human intestinal macrophages display profound inflammatory anergy despite avid phagocytic and bacteriocidal activity. J. Clin. Invest. 115, 66–75 (2005).
Ohtsuka, H. et al. Bcl6 is required for the development of mouse CD4+ and CD8α+ dendritic cells. J. Immunol. 186, 255–263 (2011).
Milling, S., Yrlid, U., Cerovic, V. & MacPherson, G. Subsets of migrating intestinal dendritic cells. Immunol. Rev. 234, 259–267 (2010).
Bull, D.M. & Bookman, M.A. Isolation and functional characterization of human intestinal mucosal lymphoid cells. J. Clin. Invest. 59, 966–974 (1977).
Zeng, R. et al. Retinoic acid regulates the development of a gut-homing precursor for intestinal dendritic cells. Mucosal Immunol. 6, 847–856 (2013).
Kitano, M. et al. Bcl6 protein expression shapes pre-germinal center B cell dynamics and follicular helper T cell heterogeneity. Immunity 34, 961–972 (2011).
Rutishauser, R.L. et al. Transcriptional repressor Blimp-1 promotes CD8+ T cell terminal differentiation and represses the acquisition of central memory T cell properties. Immunity 31, 296–308 (2009).
Suzuki, R. & Shimodaira, H. Pvclust: an R package for assessing the uncertainty in hierarchical clustering. Bioinformatics 22, 1540–1542 (2006).
Acknowledgements
We thank T. Okada (RIKEN, Japan) for YFP–Bcl-6 mice52; E. Meffre (Yale University) for Blimp-1–YFP mice53; J. Sweere and H. Kiefel for critical reading of the manuscript; L. Rott for assistance with flow cytometry and cell sorting; E. Resurreccion for assistance with immunohistochemistry; K. Bhat for writing Python scripts; all members of the Butcher laboratory for discussions; and the Immunological Genome Project and the Merad laboratory (Mount Sinai School of Medicine) for generating the publicly available microarray data of sorted mouse intestinal DC. Supported by the US National Institutes of Health (R01 AI093981, R01 DK084647 and R37 AI047822 to E.C.B.; 5T32AI007290-25 to M.L.; K01 AR59378 to S.J.K.; and AI07290 to H.H.), the US Department of Veterans Affairs (E.C.B.), the Digestive Disease Center of Stanford University (under P30 DK056339), the German Research Foundation (K.L.), the Crohn's and Colitis Foundation of America (K.L. and M.L.), The Stanford Institute for Immunity, Transplantation and Infection (K.L.), the Agency for Science, Technology And Research of Singapore (R.Z.), the Swiss National Science Foundation (PBBEP3-133516 to D.B.), the Swiss Foundation for Grants in Biology and Medicine (PASMP3-142725 to D.B.), the Leukemia & Lymphoma Society (K.M.A.), the California Institute for Regenerative Medicine (H.H.) and the Arthritis Foundation (H.H.).
Author information
Authors and Affiliations
Contributions
P.B.W. designed and did human DC experiments, including microarrays, analyzed gene expression and wrote the manuscript; K.L. designed and did all mouse experiments, analyzed gene expression and wrote the manuscript; M.L. prepared RNA and did quality control for microarray studies; D.B. provided YFP–Bcl-6 and Blimp-1–YFP mice and helped to design and to do the experiments; J.M. provided human tissue; S.J.K. designed and did experiments with mice with DC-specific Blimp-1 deficiency; R.Z. helped with YFP–Bcl-6 experiments; A.D. provided Bcl6−/− mice and feedback; K.M.A. provided feedback and YFP–Bcl-6 and Blimp-1–YFP mice; B.D. provided mice with DC-specific Blimp-1 deficiency; H.H. drove the initial interest and provided advice; and E.C.B. analyzed gene expression, guided the study and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Gating strategy for human intestinal DC subsets.
Lamina propria mononuclear cells obtained from jejunal tissue sections were stained for live/dead viability marker, CD45, lineage markers (CD3, CD14, CD16, CD56, CD19), HLADR, CD11c, CD123. Phenotypic Analysis of CD11c+ DC was done by successive gating on live CD45+ population and then the HLADR+ lineage− population.
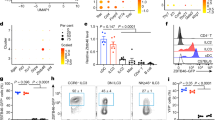
Supplementary Figure 2 Absence of canonical macrophage marker expression on CD103−Sirpα+ DCs.
a) Expression of CD64 and on CD14+ lamina propria macrophages (open black histograms) and CD103−Sirpα+ DCs (black dashed lines). b) Expression of CX3CR1 (same as in a)).
Supplementary Figure 4 Pairwise comparison of gene expression profiles of gut DC subsets with blood and skin CD1c+ and CD141+ DCs, skin CD14+ DCs and blood CD14+ monocytes.
Pearson correlation of expression profiles using the same genes as in Fig. 3a. In the comparison of CD103+Sirpα+ (hDP) DCs with blood and skin CD1c+, the individual samples with solid and open symbols correlate significantly (p≤0.001, ANOVA) with blood CD1c+ or with skin CD1c+ DCs, respectively
Supplementary Figure 5 Classification of DCs vs macrophages.
a) CD4 T cell proliferation in allogeneic cultures of responder T cells stimulated by the indicated subsets. b) Gene Expression values of MafB and Spi1 and their ratio in gut and blood DC subsets and tissue macrophages. Dataset for tissue macrophages was obtained from GSE40484.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 and Supplementary Table 3 (PDF 492 kb)
Supplementary Table 1
Gene list to accompany Figure 3. (XLSX 235 kb)
Supplementary Table 2
Gene list to accompany Figure 5. (XLSX 127 kb)
Rights and permissions
About this article
Cite this article
Watchmaker, P., Lahl, K., Lee, M. et al. Comparative transcriptional and functional profiling defines conserved programs of intestinal DC differentiation in humans and mice. Nat Immunol 15, 98–108 (2014). https://doi.org/10.1038/ni.2768
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.2768
- Springer Nature America, Inc.
This article is cited by
-
Dual RNA sequencing reveals dendritic cell reprogramming in response to typhoidal Salmonella invasion
Communications Biology (2022)
-
The role of retinoic acid in the production of immunoglobulin A
Mucosal Immunology (2022)
-
Dendritic cell functions in the inductive and effector sites of intestinal immunity
Mucosal Immunology (2022)
-
Bacteroides thetaiotaomicron-derived outer membrane vesicles promote regulatory dendritic cell responses in health but not in inflammatory bowel disease
Microbiome (2020)
-
Adjustment of dendritic cells to the breast-cancer microenvironment is subset specific
Nature Immunology (2018)