Abstract
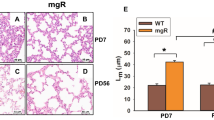
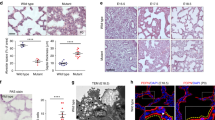
Marfan syndrome is an autosomal dominant disorder of connective tissue caused by mutations in fibrillin-1 (encoded by FBN1 in humans and Fbn1 in mice), a matrix component of extracellular microfibrils. A distinct subgroup of individuals with Marfan syndrome have distal airspace enlargement, historically described as emphysema, which frequently results in spontaneous lung rupture (pneumothorax; refs. 1–3). To investigate the pathogenesis of genetically imposed emphysema, we analyzed the lung phenotype of mice deficient in fibrillin-1, an accepted model of Marfan syndrome4. Lung abnormalities are evident in the immediate postnatal period and manifest as a developmental impairment of distal alveolar septation. Aged mice deficient in fibrillin-1 develop destructive emphysema consistent with the view that early developmental perturbations can predispose to late-onset, seemingly acquired phenotypes. We show that mice deficient in fibrillin-1 have marked dysregulation of transforming growth factor-β (TGF-β) activation and signaling, resulting in apoptosis in the developing lung. Perinatal antagonism of TGF-β attenuates apoptosis and rescues alveolar septation in vivo. These data indicate that matrix sequestration of cytokines is crucial to their regulated activation and signaling and that perturbation of this function can contribute to the pathogenesis of disease.






Similar content being viewed by others
References
Dwyer, E.M. & Troncale, F. Spontaneous pneumothorax and pulmonary disease in the Marfan syndrome. Ann. Intern. Med. 62, 1285–1292 (1965).
Hall, J.R., Pyeritz, R.E., Dudgeon, D.L. & Haller, J.A., Jr. Pneumothorax in the Marfan syndrome: prevalence and therapy. Ann. Thorac. Surg. 37, 500–504 (1984).
Wood, J.R., Bellamy, D., Child, A.H. & Citron, K.M. Pulmonary disease in patients with Marfan syndrome. Thorax 39, 780–784 (1984).
Pereira, L. et al. Targetting of the gene encoding fibrillin-1 recapitulates the vascular aspect of Marfan syndrome. Nat. Genet. 17, 218–222 (1997).
Thurlbeck, W.M. Measurement of pulmonary emphysema. Am. Rev. Respir. Dis. 95, 752–764 (1967).
Zhou, L., Lim, L., Costa, R.H. & Whitsett, J.A. Thyroid transcription factor-1, hepatocyte nuclear factor-3beta, surfactant protein B, C, and Clara cell secretory protein in developing mouse lung. J Histochem. Cytochem. 44, 1183–1193 (1996).
Bostrom, H. et al. PDGF-A signaling is a critical event in lung alveolar myofibroblast development and alveogenesis. Cell 85, 863–873 (1996).
Nakamura, T. et al. Fibulin-5/DANCE is essential for elastogenesis in vivo. Nature 415, 171–175 (2002).
Weinstein, M., Xu, X., Ohyama, K. & Deng, C.X. FGFR-3 and FGFR-4 function cooperatively to direct alveogenesis in the murine lung. Development 125, 3615–3623 (1998).
Wendel, D.P., Taylor, D.G., Albertine, K.H., Keating, M.T. & Li, D.Y. Impaired distal airway development in mice lacking elastin. Am. J. Respir. Cell Mol. Biol. 23, 320–326 (2000).
Taipale, J., Saharinen, J., Hedman, K. & Keski-Oja, J. Latent transforming growth factor-β 1 and its binding protein are components of extracellular matrix microfibrils. J. Histochem. Cytochem. 44, 875–889 (1996).
Unsold, C., Hyytiainen, M., Bruckner-Tuderman, L. & Keski-Oja, J. Latent TGF-β binding protein LTBP-1 contains three potential extracellular matrix interacting domains. J. Cell. Sci. 114, 187–197 (2001).
Dallas, S.L., Miyazono, K., Skerry, T.M., Mundy, G.R. & Bonewald, L.F. Dual role for the latent transforming growth factor-β binding protein in storage of latent TGF-β in the extracellular matrix and as a structural matrix protein. J. Cell Biol. 131, 539–549 (1995).
Dallas, S.L. et al. Role of the latent transforming growth factor-β binding protein 1 in fibrillin-containing microfibrils in bone cells in vitro and in vivo. J. Bone Miner. Res. 15, 68–81 (2000).
Isogai, Z. et al. Latent transforming growth factor-β binding protein 1 interacts with fibrillin and is a microfibril-associated protein. J. Biol. Chem. 278, 2750–2757 (2003).
Barcellos-Hoff, M.H. et al. Immunohistochemical detection of active transforming growth factor-β in situ using engineered tissue. Am. J. Pathol. 147, 1228–1237 (1995).
Flanders, K.C. et al. Transforming growth factor-β 1: histochemical localization with antibodies to different epitopes. J. Cell Biol. 108, 653–660 (1989).
Dennler, S. et al. Direct binding of Smad3 and Smad4 to critical TGFβ-inducible elements in the promoter of human plasminogen activator inhibitor-type 1 gene. EMBO J. 17, 3091–3100 (1998).
Tomita, H. et al. Early induction of transforming growth factor-β via angiotensin II type 1 receptors contributes to cardiac fibrosis induced by long-term blockade of nitric oxide synthesis in rats. Hypertension 32, 273–279 (1998).
Yamamoto, T., Takagawa, S., Katayama, I. & Nishioka, K. Anti-sclerotic effect of transforming growth factor-β antibody in a mouse model of bleomycin-induced scleroderma. Clin. Immunol. 92, 6–13 (1999).
Moustakas, A., Pardali, K., Gaal, A. & Heldin, C.H. Mechanisms of TGF-β signaling in regulation of cell growth and differentiation. Immunol. Lett. 82, 85–91 (2002).
Pereira, L. et al. Pathogenetic sequence for aneurysm revealed in mice underexpressing fibrillin-1. Proc. Natl. Acad. Sci. USA 96, 3819–3823 (1999).
Bunton, T.E. et al. Phenotypic alteration of vascular smooth muscle cells precedes elastolysis in a mouse model of Marfan syndrome. Circ. Res. 88, 37–43 (2001).
Nijbroek, G. et al. Fifteen novel FBN1 mutations causing Marfan syndrome detected by heteroduplex analysis of genomic amplicons. Am. J. Hum. Genet. 57, 8–21 (1995).
Acknowledgements
We thank J. Gauthier for providing reagents, D. Valle for helpful comments about the manuscript, M. Barcellos-Hoffs for advice about the use of TGF-β antibodies, M. Dellanoy for help with confocal imaging and members of H.C.D.'s laboratory for their support and advice. This work was supported by Robert Wood Johnson Foundation (to E.R.N.), Howard Hughes Medical Institute (to H.C.D.), US National Institutes of Health grants (to E.R.N., H.C.D. and F.R.), the Michael Murray fund from the National Marfan Foundation (to H.C.D.), Smilow Foundation (to H.C.D.) and Dr. Amy and James Elster Research Fund (to F.R.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Neptune, E., Frischmeyer, P., Arking, D. et al. Dysregulation of TGF-β activation contributes to pathogenesis in Marfan syndrome. Nat Genet 33, 407–411 (2003). https://doi.org/10.1038/ng1116
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng1116
- Springer Nature America, Inc.
This article is cited by
-
Association of MLL3 and TGF-β signaling gene polymorphisms with the susceptibility and prognostic outcomes of Stanford type B aortic dissection
BMC Cardiovascular Disorders (2023)
-
Interdisciplinary German clinical practice guidelines on the management of type B aortic dissection
Gefässchirurgie (2023)
-
Association of gene polymorphisms in FBN1 and TGF-β signaling with the susceptibility and prognostic outcomes of Stanford type B aortic dissection
BMC Medical Genomics (2022)
-
SMAD3 contributes to ascending aortic dilatation independent of transforming growth factor-beta in bicuspid and unicuspid aortic valve disease
Scientific Reports (2022)
-
The jam session between muscle stem cells and the extracellular matrix in the tissue microenvironment
npj Regenerative Medicine (2022)