Abstract
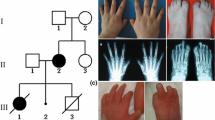
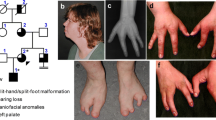
There are several human syndromes which involve defects of the limbs and the Müllerian ducts or its derivatives1–3. The hand-foot-genital (HFG) syndrome is an autosomal dominant, fully penetrant disorder that was originally described by Stern et al.4 Additional reports describing other affected families have also been published1,5–11. Limb anomalies include short first metacarpals of normal thickness, small distal phalanges of the thumbs, short middle phalanges of the fifth fingers, and fusion or delayed ossification of wrist bones. In the feet, the great toe is shorter due to a short first metatarsal and a small, pointed distal phalanx. Uterine anomalies are common in females with HFG, and typically involve a partially divided (bicornuate) or completely divided (didelphic) uterus, representing defects of MÜllerian duct fusion. Urinary tract malformations in affected HFG females include a displaced urethral opening and malposition of ureteral orifices in the bladder wall; affected males may have hypospadias (ventrally misplaced urethral opening) of variable severity1,5,7–9. We report the identification of a HOXA13 nonsense mutation in a family with hand-foot-genital syndrome. The mutation converts a highly conserved tryptophan residue in the homeodomain to a stop codon, which truncates 20 amino acids from the protein and likely eliminates or greatly reduces the ability of the protein to bind to DNA.
Similar content being viewed by others
References
Verp, M.S. et al. Heritable aspects of uterine anomalies. I. Three familial aggregates with Müllerian fusion anomalies. Fert Steril. 40, 80–65 (1983).
Pinsky, L. A community of human malformation syndromes involving the Müllerian ducts, distal extremities, urinary tract, and ears. Teratol. 9, 65–80 (1973).
Halal, F. A new syndrome of severe upper limb hypoplasia and Müllerian duct anomalies. Am.J.Med.Genet. 24, 119–126 (1986).
Stern, A.M. et al. The hand-foot-uterus syndrome. J.Pediat 77, 109–116 (1970).
Giedion, A. & Prader, A. Hand-foot-uterus (HFU) syndrome with hypospadias: the hand-foot-genital (HFG) syndrome. Pediat.Radiol. 4, 96–102 (1976).
Halal, F., Hand-Foot-Genital (Hand-Foot-Uterus) Syndrome: family report and update. Am.J.Med.Genet. 30, 793–803 (1988).
Donnenfeld, A.E., Schrager, D.S. & Corson, S.L. Update on a family with Hand-foot-genital syndrome: hypospadias and urinary tract abnormalities in two boys from the fourth generation. Am.J.Med.Genet. 44, 482–484 (1992).
Fryns, J.P., Vogels, A., Decock, P. & van den Berghe, H. . The hand-foot-genital syndrome: on the variable expression in affected males. Clinical Genet. 43, 232–234 (1993).
Verp, M.S. Urinary tract abnormalities in Hand-foot-genital syndrome. Am. J. Med. Genet. 32, 555 (1989).
Poznanski, A.K., Stern, A.S. & Gall, J.C. Radiographic findings in the Hand-Foot-Uterus Syndrome (HFUS). Radiol. 95, 129–134 (1970).
Cleveland, R.H. & Holmes, L.B. Hand-foot-genital syndrome: the importance of hallux varus. Pediat.Radiol. 20, 339–343 (1990).
Mortlock, D.P., Post, L.C. & Innis, J.W. The molecular basis of hypodactyly (Hd): a deletion in Hoxa13 leads to arrest of digital arch formation. Nature Genet 13, 284–289 (1996).
Hummel, K., Hypodactyly, a semidominant lethal mutation in mice. J.Hered. 61, 219–220 (1970).
Borrow, J. et al. The t(7;11)(p15;p15) translocation in acute myeloid leukaemia fuses the genes for nucleoporin NUP98 and class I homeoprotein HOXA9. Nature Genet 12, 159–167 (1996).
Gyapay, G. et al. The 1993-94 Genethon human genetic linkage map. Nature Genet 7, 246–339 (1994).
Duboule, D. Guidebook to the Homeobox Genes (Oxford University Press, New York, 1994).
Muragaki, Y., Mundlos, S., Upton, J. & Olsen, B.R. Altered growth and branching patterns in synpolydactyly caused by mutations in HOXD13. Science 272, 548–551 (1996).
Copeland, J.W.R., Nasiadka, A., Dietrich, B. & Krause, H. Patterning of the Drosophila embryo by a homeodomain-deleted Ftz polypeptide. Nature 379, 162–165 (1996).
Schnabel, C. & Abate-Shen, C. Repression by HoxA7 is mediated by the homeodomain and the modulatory action of its N-terminal-arm residues. Mol.Cell.Biol. 16, 2678–2688 (1996).
Vershon, A., Jin, Y. & Johnson, A. A homeo domain protein lacking specific side chains of helix 3 can still bind DNA and direct transcriptional repression. Genes.Dev. 9, 182–192 (1995).
Zappavigna, V., Sartori, D. & Mavilio, F. Specificity of HOX protein function depends on DNA-protein and protein-protein interactions, both mediated by the homeo domain. Genes.Dev. 8, 732–744 (1994).
Acién, P. Embryological observations on the female genital tract. Human Reprod. 7, 437–445 (1992).
Blüm, V. Vertebrate Reproduction (Springer-Verlag, Berlin/Heidelberg, 1986).
Gruenwald, P. The relation of the growing Müllerian duct to the Wolffian duct and its importance for the genesis of malformations. Anat.Rec. 81, 1–19 (1941).
Sharman, G.B. Reproductive physiology of marsupials. Science 167, 1221–1228 (1970).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mortlock, D., Innis, J. Mutation of HOXA13 in hand-foot-genital syndrome . Nat Genet 15, 179–180 (1997). https://doi.org/10.1038/ng0297-179
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0297-179
- Springer Nature America, Inc.
This article is cited by
-
The urothelial gene regulatory network: understanding biology to improve bladder cancer management
Oncogene (2024)
-
Regional effect on the molecular clock rate of protein evolution in Eutherian and Metatherian genomes
BMC Ecology and Evolution (2021)
-
Transcriptional landscape of the embryonic chicken Müllerian duct
BMC Genomics (2020)
-
The transcriptional regulator CBX2 and ovarian function: A whole genome and whole transcriptome approach
Scientific Reports (2019)