Abstract
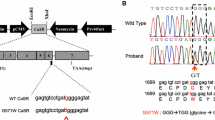
Adaptor protein-2 (AP2), a central component of clathrin-coated vesicles (CCVs), is pivotal in clathrin-mediated endocytosis, which internalizes plasma membrane constituents such as G protein–coupled receptors (GPCRs)1,2,3. AP2, a heterotetramer of α, β, μ and σ subunits, links clathrin to vesicle membranes and binds to tyrosine- and dileucine-based motifs of membrane-associated cargo proteins1,4. Here we show that missense mutations of AP2 σ subunit (AP2S1) affecting Arg15, which forms key contacts with dileucine-based motifs of CCV cargo proteins4, result in familial hypocalciuric hypercalcemia type 3 (FHH3), an extracellular calcium homeostasis disorder affecting the parathyroids, kidneys and bone5,6,7. We found AP2S1 mutations in >20% of cases of FHH without mutations in calcium-sensing GPCR (CASR)8,9,10,11,12, which cause FHH1. AP2S1 mutations decreased the sensitivity of CaSR-expressing cells to extracellular calcium and reduced CaSR endocytosis, probably through loss of interaction with a C-terminal CaSR dileucine-based motif, whose disruption also decreased intracellular signaling. Thus, our results identify a new role for AP2 in extracellular calcium homeostasis.



Similar content being viewed by others
Accession codes
References
Collins, B.M., McCoy, A.J., Kent, H.M., Evans, P.R. & Owen, D.J. Molecular architecture and functional model of the endocytic AP2 complex. Cell 109, 523–535 (2002).
Edeling, M.A. et al. Molecular switches involving the AP-2β2 appendage regulate endocytic cargo selection and clathrin coat assembly. Dev. Cell 10, 329–342 (2006).
Ohno, H. Physiological roles of clathrin adaptor AP complexes: lessons from mutant animals. J. Biochem. 139, 943–948 (2006).
Kelly, B.T. et al. A structural explanation for the binding of endocytic dileucine motifs by the AP2 complex. Nature 456, 976–979 (2008).
McMurtry, C.T. et al. Significant developmental elevation in serum parathyroid hormone levels in a large kindred with familial benign (hypocalciuric) hypercalcemia. Am. J. Med. 93, 247–258 (1992).
Lloyd, S.E., Pannett, A.A., Dixon, P.H., Whyte, M.P. & Thakker, R.V. Localization of familial benign hypercalcemia, Oklahoma variant (FBHOk), to chromosome 19q13. Am. J. Hum. Genet. 64, 189–195 (1999).
Nesbit, M.A. et al. Identification of a second kindred with familial hypocalciuric hypercalcemia type 3 (FHH3) narrows localization to a <3.5 megabase pair region on chromosome 19q13.3. J. Clin. Endocrinol. Metab. 95, 1947–1954 (2010).
Pollak, M.R. et al. Mutations in the human Ca2+-sensing receptor gene cause familial hypocalciuric hypercalcemia and neonatal severe hyperparathyroidism. Cell 75, 1297–1303 (1993).
Hannan, F.M. et al. Identification of 70 calcium-sensing receptor mutations in hyper- and hypo-calcaemic patients: evidence for clustering of extracellular domain mutations at calcium-binding sites. Hum. Mol. Genet. 21, 2768–2778 (2012).
Brown, E.M. & MacLeod, R.J. Extracellular calcium sensing and extracellular calcium signaling. Physiol. Rev. 81, 239–297 (2001).
Pearce, S.H. et al. Calcium-sensing receptor mutations in familial benign hypercalcemia and neonatal hyperparathyroidism. J. Clin. Invest. 96, 2683–2692 (1995).
Pearce, S.H. et al. Functional characterization of calcium-sensing receptor mutations expressed in human embryonic kidney cells. J. Clin. Invest. 98, 1860–1866 (1996).
Kirchhausen, T. et al. AP17 and AP19, the mammalian small chains of the clathrin-associated protein complexes show homology to Yap17p, their putative homolog in yeast. J. Biol. Chem. 266, 11153–11157 (1991).
Heath, H. III, Jackson, C.E., Otterud, B. & Leppert, M.F. Genetic linkage analysis in familial benign (hypocalciuric) hypercalcemia: evidence for locus heterogeneity. Am. J. Hum. Genet. 53, 193–200 (1993).
Wagener, B.M., Marjon, N.A., Revankar, C.M. & Prossnitz, E.R. Adaptor protein-2 interaction with arrestin regulates GPCR recycling and apoptosis. Traffic 10, 1286–1300 (2009).
Brown, E.M. et al. Cloning and characterization of an extracellular Ca2+-sensing receptor from bovine parathyroid. Nature 366, 575–580 (1993).
Hofer, A.M. & Brown, E.M. Extracellular calcium sensing and signalling. Nat. Rev. Mol. Cell Biol. 4, 530–538 (2003).
Grant, M.P., Stepanchick, A., Cavanaugh, A. & Breitwieser, G.E. Agonist-driven maturation and plasma membrane insertion of calcium-sensing receptors dynamically control signal amplitude. Sci. Signal. 4, ra78 (2011).
Hough, T.A. et al. Activating calcium-sensing receptor mutation in the mouse is associated with cataracts and ectopic calcification. Proc. Natl. Acad. Sci. USA 101, 13566–13571 (2004).
Reyes-Ibarra, A.P. et al. Calcium-sensing receptor endocytosis links extracellular calcium signaling to parathyroid hormone-related peptide secretion via a Rab11a-dependent and AMSH-sensitive mechanism. Mol. Endocrinol. 21, 1394–1407 (2007).
Leach, K. et al. Identification of molecular phenotypes and biased signaling induced by naturally occurring mutations of the human calcium-sensing receptor. Endocrinology 153, 4304–4316 (2012).
Guarnieri, V. et al. Calcium-sensing receptor (CASR) mutations in hypercalcemic states: studies from a single endocrine clinic over three years. J. Clin. Endocrinol. Metab. 95, 1819–1829 (2010).
Fang, Y., Huang, C.C., Kain, S.R. & Li, X. Use of coexpressed enhanced green fluorescent protein as a marker for identifying transfected cells. Methods Enzymol. 302, 207–212 (1999).
Nesbit, M.A. et al. Characterization of GATA3 mutations in the hypoparathyroidism, deafness, and renal dysplasia (HDR) syndrome. J. Biol. Chem. 279, 22624–22634 (2004).
Gaynor, K.U. et al. A missense GATA3 mutation, Thr272Ile, causes the hypoparathyroidism, deafness, and renal dysplasia syndrome. J. Clin. Endocrinol. Metab. 94, 3897–3904 (2009).
Goldsmith, P.K., Fan, G., Miller, J.L., Rogers, K.V. & Spiegel, A.M. Monoclonal antibodies against synthetic peptides corresponding to the extracellular domain of the human Ca2+ receptor: characterization and use in studying concanavalin A inhibition. J. Bone Miner. Res. 12, 1780–1788 (1997).
Mounkes, L., Kozlov, S., Burke, B. & Stewart, C.L. The laminopathies: nuclear structure meets disease. Curr. Opin. Genet. Dev. 13, 223–230 (2003).
Kim, Y.M. & Benovic, J.L. Differential roles of arrestin-2 interaction with clathrin and adaptor protein 2 in G protein–coupled receptor trafficking. J. Biol. Chem. 277, 30760–30768 (2002).
Nesterov, A., Carter, R.E., Sorkina, T., Gill, G.N. & Sorkin, A. Inhibition of the receptor-binding function of clathrin adaptor protein AP-2 by dominant-negative mutant mu2 subunit and its effects on endocytosis. EMBO J. 18, 2489–2499 (1999).
Hannan, S. et al. γ-aminobutyric acid type B (GABA(B)) receptor internalization is regulated by the R2 subunit. J. Biol. Chem. 286, 24324–24335 (2011).
Clark, M.J. et al. Performance comparison of exome DNA sequencing technologies. Nat. Biotechnol. 29, 908–914 (2011).
Boyden, L.M. et al. Mutations in kelch-like 3 and cullin 3 cause hypertension and electrolyte abnormalities. Nature 482, 98–102 (2012).
Lunter, G. & Goodson, M. Stampy: a statistical algorithm for sensitive and fast mapping of Illumina sequence reads. Genome Res. 21, 936–939 (2011).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Robinson, J.T. et al. Integrative genomics viewer. Nat. Biotechnol. 29, 24–26 (2011).
Bataille, S., Berland, Y., Fontes, M. & Burtey, S. High Resolution Melt analysis for mutation screening in PKD1 and PKD2. BMC Nephrol. 12, 57 (2011).
Larkin, M.A. et al. Clustal W and Clustal X version 2.0. Bioinformatics 23, 2947–2948 (2007).
Acknowledgements
This work was supported by the UK Medical Research Council (MRC) programme grants G9825289 and G1000467 (to M.A.N., F.M.H., A.A.C.R., C.E.T. and R.V.T.); the National Institute for Health Research (NIHR) Oxford Biomedical Research Centre Programme (to M.A.N. and R.V.T.); the High-Throughput Genomics Group, Wellcome Trust Centre for Human Genetics (Wellcome Trust grant reference 090532/Z/09/Z and MRC Hub grant G0900747 91070); the Research and Development Office, Northern Ireland (to U.G., S.J.H. and P.J.M.); and the Shriners Hospitals for Children (grant 15958) (to M.P.W.). S.A.H. is a Wellcome Trust Clinical Research Training Fellow.
Author information
Authors and Affiliations
Contributions
M.A.N. and R.V.T. designed the experiments. M.A.N., F.M.H., S.A.H., A.A.C.R. and C.E.T. performed experiments and analyzed data. M.A.N., S.A.H., A.A.C.R., T.C., C.E.T., L.G. and A.J.R. carried out sequencing and information technology. G.M. and D.B. directed the information technology and the exome capture DNA sequencing infrastructure. T.C., U.G., P.J.M., S.J.H., M.P.W. and R.V.T. recruited subjects and families with FHH. M.A.N., F.M.H., S.A.H. and N.R. performed the AP2S1 functional calcium assays. M.A.N., F.M.H. and R.V.T. wrote the manuscript. All authors checked the manuscript for scientific content and contributed to the final drafting of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–3, Supplementary Table 1 and Supplementary Note (PDF 321 kb)
Rights and permissions
About this article
Cite this article
Nesbit, M., Hannan, F., Howles, S. et al. Mutations in AP2S1 cause familial hypocalciuric hypercalcemia type 3. Nat Genet 45, 93–97 (2013). https://doi.org/10.1038/ng.2492
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.2492
- Springer Nature America, Inc.
This article is cited by
-
Genetica dell’ipercalcemia ipocalciurica familiare
L'Endocrinologo (2023)
-
Computational insights into missense mutations in HTT gene causing Huntington’s disease and its interactome networks
Irish Journal of Medical Science (1971 -) (2023)
-
Visualization of the process of a nanocarrier-mediated gene delivery: stabilization, endocytosis and endosomal escape of genes for intracellular spreading
Journal of Nanobiotechnology (2022)
-
Construction and application of star polycation nanocarrier-based microRNA delivery system in Arabidopsis and maize
Journal of Nanobiotechnology (2022)
-
Genetic causes of neonatal and infantile hypercalcaemia
Pediatric Nephrology (2022)