Abstract
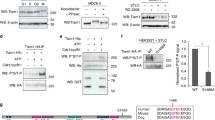
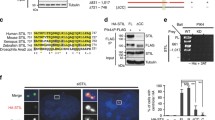
Formation of a bipolar spindle is essential for faithful chromosome segregation at mitosis. Because centrosomes define spindle poles, defects in centrosome number and structural organization can lead to a loss of bipolarity1. In addition, microtubule-mediated pulling and pushing forces acting on centrosomes and chromosomes are also important for bipolar spindle formation2. Polo-like kinase 1 (Plk1) is a highly conserved Ser/Thr kinase that has essential roles in the formation of a bipolar spindle with focused poles3,4,5. However, the mechanism by which Plk1 regulates spindle-pole formation is poorly understood. Here, we identify a novel centrosomal substrate of Plk1, Kizuna (Kiz), depletion of which causes fragmentation and dissociation of the pericentriolar material from centrioles at prometaphase, resulting in multipolar spindles. We demonstrate that Kiz is critical for establishing a robust mitotic centrosome architecture that can endure the forces that converge on the centrosomes during spindle formation, and suggest that Plk1 maintains the integrity of the spindle poles by phosphorylating Kiz.





Similar content being viewed by others
Accession codes
References
Nigg, E. A. Centrosome aberrations: cause or consequence of cancer progression? Nature Rev. Cancer 2, 815–825 (2002).
Scholey, J. M., Brust Mascher, I. & Mogilner, A. Cell division. Nature 422, 746–752 (2003).
Lane, H. A. & Nigg, E. A. Antibody microinjection reveals an essential role for human polo-like kinase 1 (Plk1) in the functional maturation of mitotic centrosomes. J. Cell Biol. 135, 1701–1713 (1996).
Sumara, I. et al. Roles of polo-like kinase 1 in the assembly of functional mitotic spindles. Curr. Biol. 14, 1712–1722 (2004).
van Vugt, M. A. et al. Polo-like kinase-1 is required for bipolar spindle formation but is dispensable for anaphase promoting complex/Cdc20 activation and initiation of cytokinesis. J. Biol. Chem. 279, 36841–36854 (2004).
Bornens, M. Centrosome composition and microtubule anchoring mechanisms. Curr. Opin. Cell Biol. 14, 25–34 (2002).
Palazzo, R. E., Vogel, J. M., Schnackenberg, B. J., Hull, D. R. & Wu, X. Centrosome maturation. Curr. Top. Dev. Biol. 49, 449–470 (2000).
Blagden, S. P. & Glover, D. M. Polar expeditions — provisioning the centrosome for mitosis. Nature Cell Biol. 5, 505–511 (2003).
Fry, A. M., Mayor, T. & Nigg, E. A. Regulating centrosomes by protein phosphorylation. Curr. Top. Dev. Biol. 49, 291–312 (2000).
Barr, F. A., Sillje, H. H. & Nigg, E. A. Polo-like kinases and the orchestration of cell division. Nature Rev. Mol. Cell Biol. 5, 429–440 (2004).
Fukunaga, R. & Hunter, T. MNK1, a new MAP kinase-activated protein kinase, isolated by a novel expression screening method for identifying protein kinase substrates. EMBO J. 16, 1921–1933 (1997).
Tachibana, K. E., Gonzalez, M. A., Guarguaglini, G., Nigg, E. A. & Laskey, R. A. Depletion of licensing inhibitor geminin causes centrosome overduplication and mitotic defects. EMBO Rep. 6, 1052–1057 (2005).
Saunders, W. Centrosomal amplification and spindle multipolarity in cancer cells. Semin. Cancer Biol. 15, 25–32 (2005).
Hut, H. M. et al. Centrosomes split in the presence of impaired DNA integrity during mitosis. Mol. Biol. Cell 14, 1993–2004 (2003).
Abal, M., Keryer, G. & Bornens, M. Centrioles resist forces applied on centrosomes during G2/M transition. Biol. Cell 97, 425–434 (2005).
Paoletti, A., Moudjou, M., Paintrand, M., Salisbury, J. L. & Bornens, M. Most of centrin in animal cells is not centrosome-associated and centrosomal centrin is confined to the distal lumen of centrioles. J. Cell Sci. 109, 3089–3102 (1996).
Ishikawa, H., Kubo, A., Tsukita, S. & Tsukita, S. Odf2-deficient mother centrioles lack distal/subdistal appendages and the ability to generate primary cilia. Nature Cell Biol. 7, 517–524 (2005).
Mogensen, M. M., Malik, A., Piel, M., Bouckson Castaing, V. & Bornens, M. Microtubule minus-end anchorage at centrosomal and non-centrosomal sites: the role of ninein. J. Cell Sci. 113, 3013–3023 (2000).
Dictenberg, J. B. et al. Pericentrin and γ-tubulin form a protein complex and are organized into a novel lattice at the centrosome. J. Cell Biol. 141, 163–174 (1998).
Takahashi, M., Yamagiwa, A., Nishimura, T., Mukai, H. & Ono, Y. Centrosomal proteins CG-NAP and kendrin provide microtubule nucleation sites by anchoring γ-tubulin ring complex. Mol. Biol. Cell 13, 3235–3245 (2002).
Murphy, S. M., Urbani, L. & Stearns, T. The mammalian γ-tubulin complex contains homologues of the yeast spindle pole body components spc97p and spc98p. J. Cell Biol. 141, 663–674 (1998).
Levesque, A. A. & Compton, D. A. The chromokinesin Kid is necessary for chromosome arm orientation and oscillation, but not congression, on mitotic spindles. J. Cell Biol. 154, 1135–1146 (2001).
Tokai-Nishizumi, N., Ohsugi, M., Suzuki, E. & Yamamoto, T. The chromokinesin Kid is required for maintenance of proper metaphase spindle size. Mol. Biol. Cell 16, 5455–5463 (2005).
Nakajima, H., Toyoshima Morimoto, F., Taniguchi, E. & Nishida, E. Identification of a consensus motif for Plk (Polo-like kinase) phosphorylation reveals Myt1 as a Plk1 substrate. J. Biol. Chem. 278, 25277–25280 (2003).
Davis, F. M., Tsao, T. Y., Fowler, S. K. & Rao, P. N. Monoclonal antibodies to mitotic cells. Proc. Natl Acad. Sci. USA 80, 2926–2930 (1983).
Kumagai, A. & Dunphy, W. G. Purification and molecular cloning of Plx1, a Cdc25-regulatory kinase from Xenopus egg extracts. Science 273, 1377–1380 (1996).
Elia, A. E. et al. The molecular basis for phosphodependent substrate targeting and regulation of Plks by the Polo-box domain. Cell 115, 83–95 (2003).
Merdes, A., Heald, R., Samejima, K., Earnshaw, W. C. & Cleveland, D. W. Formation of spindle poles by dynein/dynactin-dependent transport of NuMA. J. Cell Biol. 149, 851–862 (2000).
Garrett, S., Auer, K., Compton, D. A. & Kapoor, T. M. hTPX2 is required for normal spindle morphology and centrosome integrity during vertebrate cell division. Curr. Biol. 12, 2055–2059 (2002).
Zimmerman, W. C., Sillibourne, J., Rosa, J. & Doxsey, S. J. Mitosis-specific anchoring of γ-tubulin complexes by pericentrin controls spindle organization and mitotic entry. Mol. Biol. Cell 15, 3642–3657 (2004).
Sumara, I. et al. The dissociation of cohesin from chromosomes in prophase is regulated by Polo-like kinase. Mol. Cell 9, 515–525 (2002).
Boyle, W. J., van der Geer, P. & Hunter, T. Phosphopeptide mapping and phosphoamino acid analysis by two-dimensional separation on thin-layer cellulose plates. Methods. Enzymol. 201, 110–149 (1991).
Ohsugi, M. et al. Cdc2-mediated phosphorylation of Kid controls its distribution to spindle and chromosomes. EMBO J. 22, 2091–2103 (2003).
Acknowledgements
We thank R. Fukunaga for λGEX–5 HeLa cDNA library and kind advice on solid-phase phosphorylation screening, Y. Ono for antibodies against pericentrin–kendrin, CG-NAP and GCP2, and pTB701–HA–kendrin expression plasmids. S. Tsukita for antibodies against ODF2 and ninein, Y. Watanabe for Plk1 siRNA, N. Nomura for help in RNAi experiments. R. F. Whittier, Y. Watanabe, K. Tanaka, N. Tokai-Nishizumi and K. Yokoyama for helpful discussions. This work was supported by grants-in-aid from the Japan Society for the Promotion of Science and from the Ministry of Education, Cultures, Sports, Science and Technology, Japan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures S1, S2, S3 and Supplementary Data (PDF 4489 kb)
Rights and permissions
About this article
Cite this article
Oshimori, N., Ohsugi, M. & Yamamoto, T. The Plk1 target Kizuna stabilizes mitotic centrosomes to ensure spindle bipolarity. Nat Cell Biol 8, 1095–1101 (2006). https://doi.org/10.1038/ncb1474
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1474
- Springer Nature Limited
This article is cited by
-
Immune adaptor SKAP1 acts a scaffold for Polo-like kinase 1 (PLK1) for the optimal cell cycling of T-cells
Scientific Reports (2019)
-
Selective autophagy maintains centrosome integrity and accurate mitosis by turnover of centriolar satellites
Nature Communications (2019)
-
γ-tubulin as a signal-transducing molecule and meshwork with therapeutic potential
Signal Transduction and Targeted Therapy (2018)
-
Platform-independent gene expression signature differentiates sessile serrated adenomas/polyps and hyperplastic polyps of the colon
BMC Medical Genomics (2017)
-
Plk1 bound to Bub1 contributes to spindle assembly checkpoint activity during mitosis
Scientific Reports (2017)