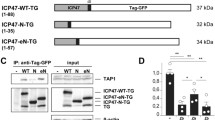
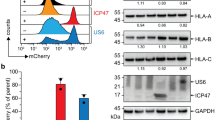
Abstract
Cellular immunity against viral infection and tumour cells depends on antigen presentation by major histocompatibility complex class I (MHC I) molecules. Intracellular antigenic peptides are transported into the endoplasmic reticulum by the transporter associated with antigen processing (TAP) and then loaded onto the nascent MHC I molecules, which are exported to the cell surface and present peptides to the immune system1. Cytotoxic T lymphocytes recognize non-self peptides and program the infected or malignant cells for apoptosis. Defects in TAP account for immunodeficiency and tumour development. To escape immune surveillance, some viruses have evolved strategies either to downregulate TAP expression or directly inhibit TAP activity. So far, neither the architecture of TAP nor the mechanism of viral inhibition has been elucidated at the structural level. Here we describe the cryo-electron microscopy structure of human TAP in complex with its inhibitor ICP47, a small protein produced by the herpes simplex virus I. Here we show that the 12 transmembrane helices and 2 cytosolic nucleotide-binding domains of the transporter adopt an inward-facing conformation with the two nucleotide-binding domains separated. The viral inhibitor ICP47 forms a long helical hairpin, which plugs the translocation pathway of TAP from the cytoplasmic side. Association of ICP47 precludes substrate binding and prevents nucleotide-binding domain closure necessary for ATP hydrolysis. This work illustrates a striking example of immune evasion by persistent viruses. By blocking viral antigens from entering the endoplasmic reticulum, herpes simplex virus is hidden from cytotoxic T lymphocytes, which may contribute to establishing a lifelong infection in the host.




Similar content being viewed by others
References
Blum, J. S., Wearsch, P. A. & Cresswell, P. Pathways of antigen processing. Annu. Rev. Immunol. 31, 443–473 (2013)
van de Weijer, M. L., Luteijn, R. D. & Wiertz, E. J. Viral immune evasion: lessons in MHC class I antigen presentation. Semin. Immunol. 27, 125–137 (2015)
Verweij, M. C. et al. Viral inhibition of the transporter associated with antigen processing (TAP): a striking example of functional convergent evolution. PLoS Pathog. 11, e1004743 (2015)
Koch, J., Guntrum, R., Heintke, S., Kyritsis, C. & Tampé, R. Functional dissection of the transmembrane domains of the transporter associated with antigen processing (TAP). J. Biol. Chem. 279, 10142–10147 (2004)
Hill, A. B., Barnett, B. C., McMichael, A. J. & McGeoch, D. J. HLA class I molecules are not transported to the cell surface in cells infected with herpes simplex virus types 1 and 2. J. Immunol. 152, 2736–2741 (1994)
York, I. A. et al. A cytosolic herpes simplex virus protein inhibits antigen presentation to CD8+ T lymphocytes. Cell 77, 525–535 (1994)
Hill, A. et al. Herpes simplex virus turns off the TAP to evade host immunity. Nature 375, 411–415 (1995)
Früh, K. et al. A viral inhibitor of peptide transporters for antigen presentation. Nature 375, 415–418 (1995)
Tomazin, R. et al. Stable binding of the herpes simplex virus ICP47 protein to the peptide binding site of TAP. EMBO J. 15, 3256–3266 (1996)
Ahn, K. et al. Molecular mechanism and species specificity of TAP inhibition by herpes simplex virus ICP47. EMBO J. 15, 3247–3255 (1996)
Galocha, B. et al. The active site of ICP47, a herpes simplex virus-encoded inhibitor of the major histocompatibility complex (MHC)-encoded peptide transporter associated with antigen processing (TAP), maps to the NH2-terminal 35 residues. J. Exp. Med. 185, 1565–1572 (1997)
Neumann, L., Kraas, W., Uebel, S., Jung, G. & Tampé, R. The active domain of the herpes simplex virus protein ICP47: a potent inhibitor of the transporter associated with antigen processing. J. Mol. Biol. 272, 484–492 (1997)
Aisenbrey, C. et al. Structure and dynamics of membrane-associated ICP47, a viral inhibitor of the MHC I antigen-processing machinery. J. Biol. Chem. 281, 30365–30372 (2006)
Powis, S. H. et al. Polymorphism in a second ABC transporter gene located within the class II region of the human major histocompatibility complex. Proc. Natl Acad. Sci. USA 89, 1463–1467 (1992)
Henderson, R. et al. Outcome of the first electron microscopy validation task force meeting. Structure 20, 205–214 (2012)
Ward, A., Reyes, C. L., Yu, J., Roth, C. B. & Chang, G. Flexibility in the ABC transporter MsbA: alternating access with a twist. Proc. Natl Acad. Sci. USA 104, 19005–19010 (2007)
Lin, D. Y., Huang, S. & Chen, J. Crystal structures of a polypeptide processing and secretion transporter. Nature 523, 425–430 (2015)
Aller, S. G. et al. Structure of P-glycoprotein reveals a molecular basis for poly-specific drug binding. Science 323, 1718–1722 (2009)
Jin, M. S., Oldham, M. L., Zhang, Q. & Chen, J. Crystal structure of the multidrug transporter P-glycoprotein from Caenorhabditis elegans. Nature 490, 566–569 (2012)
Leonhardt, R. M., Keusekotten, K., Bekpen, C. & Knittler, M. R. Critical role for the tapasin-docking site of TAP2 in the functional integrity of the MHC class I-peptide-loading complex. J. Immunol. 175, 5104–5114 (2005)
Nijenhuis, M. & Hämmerling, G. J. Multiple regions of the transporter associated with antigen processing (TAP) contribute to its peptide binding site. J. Immunol. 157, 5467–5477 (1996)
Corradi, V., Singh, G. & Tieleman, D. P. The human transporter associated with antigen processing: molecular models to describe peptide binding competent states. J. Biol. Chem. 287, 28099–28111 (2012)
Gorbulev, S., Abele, R. & Tampé, R. Allosteric crosstalk between peptide-binding, transport, and ATP hydrolysis of the ABC transporter TAP. Proc. Natl Acad. Sci. USA 98, 3732–3737 (2001)
Geng, J., Sivaramakrishnan, S. & Raghavan, M. Analyses of conformational states of the transporter associated with antigen processing (TAP) protein in a native cellular membrane environment. J. Biol. Chem. 288, 37039–37047 (2013)
Herget, M. et al. Conformation of peptides bound to the transporter associated with antigen processing (TAP). Proc. Natl Acad. Sci. USA 108, 1349–1354 (2011)
Chen, S., Oldham, M. L., Davidson, A. L. & Chen, J. Carbon catabolite repression of the maltose transporter revealed by X-ray crystallography. Nature 499, 364–368 (2013)
Kadaba, N. S., Kaiser, J. T., Johnson, E., Lee, A. & Rees, D. C. The high-affinity E. coli methionine ABC transporter: structure and allosteric regulation. Science 321, 250–253 (2008)
Gerber, S., Comellas-Bigler, M., Goetz, B. A. & Locher, K. P. Structural basis of trans-inhibition in a molybdate/tungstate ABC transporter. Science 321, 246–250 (2008)
Li, X. et al. Electron counting and beam-induced motion correction enable near-atomic-resolution single-particle cryo-EM. Nature Methods 10, 584–590 (2013)
Penczek, P. A. et al. CTER-rapid estimation of CTF parameters with error assessment. Ultramicroscopy 140, 9–19 (2014)
Ludtke, S. J., Baldwin, P. R. & Chiu, W. EMAN: semiautomated software for high-resolution single-particle reconstructions. J. Struct. Biol. 128, 82–97 (1999)
Yang, Z., Fang, J., Chittuluru, J., Asturias, F. J. & Penczek, P. A. Iterative stable alignment and clustering of 2D transmission electron microscope images. Structure 20, 237–247 (2012)
Hohn, M. et al. SPARX, a new environment for cryo-EM image processing. J. Struct. Biol. 157, 47–55 (2007)
Mastronarde, D. N. Automated electron microscope tomography using robust prediction of specimen movements. J. Struct. Biol. 152, 36–51 (2005)
Brilot, A. F. et al. Beam-induced motion of vitrified specimen on holey carbon film. J. Struct. Biol. 177, 630–637 (2012)
Campbell, M. G. et al. Movies of ice-embedded particles enhance resolution in electron cryo-microscopy. Structure 20, 1823–1828 (2012)
Rohou, A. & Grigorieff, N. CTFFIND4: fast and accurate defocus estimation from electron micrographs. J. Struct. Biol. 192, 216–221 (2015)
Grant, T. & Grigorieff, N. Measuring the optimal exposure for single particle cryo-EM using a 2.6 Å reconstruction of rotavirus VP6. eLife 4, e06980 (2015)
Scheres, S. H. RELION: implementation of a Bayesian approach to cryo-EM structure determination. J. Struct. Biol. 180, 519–530 (2012)
Grigorieff, N. FREALIGN: high-resolution refinement of single particle structures. J. Struct. Biol. 157, 117–125 (2007)
Gaudet, R. & Wiley, D. C. Structure of the ABC ATPase domain of human TAP1, the transporter associated with antigen processing. EMBO J. 20, 4964–4972 (2001)
Eswar, N. et al. Comparative protein structure modeling using Modeller. Curr. Protoc. Bioinformat. Chapter 5, Unit 5.6 (2006)
Shintre, C. A. et al. Structures of ABCB10, a human ATP-binding cassette transporter in apo- and nucleotide-bound states. Proc. Natl Acad. Sci. USA 110, 9710–9715 (2013)
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D 66, 486–501 (2010)
DeLano, W. L. The PyMOL molecular graphics system (DeLano Scientific, 2002)
Pettersen, E. F. et al. UCSF Chimera—a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004)
Acknowledgements
We thank Z. Yu, C. Hong and R. Huang for assistance in data collection and processing. We thank X. Zhang for training in preparation of cryo-EM grids. We also thank S. McCarry for editing the manuscript. R.K.H. is a Howard Hughes Medical Institute fellow of the Helen Hay Whitney Foundation and J.C. is an Investigator of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Contributions
M.L.O. performed protein purification and cryo-EM experiments. R.K.H. provided guidance in data processing. A.M.S. provided assistance with protein expression and purification. E.D. performed the fluorescence-activated cell sorting (FACS) experiments. Z.L. and T.W. collected preliminary cryo-EM data and generated the initial model. M.L.O. and J.C. prepared the manuscript with input from all co-authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 3 FSC indicating the resolution of the density map.
FSC plots were generated between reconstructions from random halves of the data. The frequency at which the dashed line passes through FSC = 0.143 indicates the reported resolution. Corresponding values are given in Extended Data Table 1.
Rights and permissions
About this article
Cite this article
Oldham, M., Hite, R., Steffen, A. et al. A mechanism of viral immune evasion revealed by cryo-EM analysis of the TAP transporter. Nature 529, 537–540 (2016). https://doi.org/10.1038/nature16506
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature16506
- Springer Nature Limited
This article is cited by
-
In vivo topical gene therapy for recessive dystrophic epidermolysis bullosa: a phase 1 and 2 trial
Nature Medicine (2022)
-
Chemotherapy agents stimulate dendritic cells against human colon cancer cells through upregulation of the transporter associated with antigen processing
Scientific Reports (2021)
-
Novel transcription regulatory sequences and factors of the immune evasion protein ICP47 (US12) of herpes simplex viruses
Virology Journal (2020)
-
Mechanics and pharmacology of substrate selection and transport by eukaryotic ABC exporters
Nature Structural & Molecular Biology (2019)
-
On the use of Pichia pastoris for isotopic labeling of human GPCRs for NMR studies
Journal of Biomolecular NMR (2018)