Abstract
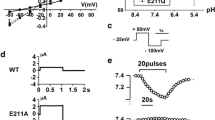
Eukaryotic members of the CLC gene family function as plasma membrane chloride channels, or may provide neutralizing anion currents for V-type H+-ATPases that acidify compartments of the endosomal/lysosomal pathway1. Loss-of-function mutations in the endosomal protein ClC-5 impair renal endocytosis2 and lead to kidney stones3, whereas loss of function of the endosomal/lysosomal protein ClC-7 entails osteopetrosis4 and lysosomal storage disease5. Vesicular CLCs have been thought to be Cl- channels, in particular because ClC-4 and ClC-5 mediate plasma membrane Cl- currents upon heterologous expression6,7. Here we show that these two mainly endosomal CLC proteins instead function as electrogenic Cl-/H+ exchangers (also called antiporters), resembling the transport activity of the bacterial protein ClC-e1 (ref. 8), the crystal structure of which has already been determined9. Neutralization of a critical glutamate residue not only abolished the steep voltage-dependence of transport7, but also eliminated the coupling of anion flux to proton counter-transport. ClC-4 and ClC-5 may still compensate the charge accumulation by endosomal proton pumps, but are expected to couple directly vesicular pH gradients to Cl- gradients.



Similar content being viewed by others
References
Jentsch, T. J., Poët, M., Fuhrmann, J. C. & Zdebik, A. A. Physiological functions of CLC Cl- channels gleaned from human genetic disease and mouse models. Annu. Rev. Physiol. 67, 779–807 (2005)
Piwon, N., Günther, W., Schwake, M., Bösl, M. R. & Jentsch, T. J. ClC-5 Cl--channel disruption impairs endocytosis in a mouse model for Dent's disease. Nature 408, 369–373 (2000)
Lloyd, S. E. et al. A common molecular basis for three inherited kidney stone diseases. Nature 379, 445–449 (1996)
Kornak, U. et al. Loss of the ClC-7 chloride channel leads to osteopetrosis in mice and man. Cell 104, 205–215 (2001)
Kasper, D. et al. Loss of the chloride channel ClC-7 leads to lysosomal storage disease and neurodegeneration. EMBO J. 24, 1079–1091 (2005)
Steinmeyer, K., Schwappach, B., Bens, M., Vandewalle, A. & Jentsch, T. J. Cloning and functional expression of rat CLC-5, a chloride channel related to kidney disease. J. Biol. Chem. 270, 31172–31177 (1995)
Friedrich, T., Breiderhoff, T. & Jentsch, T. J. Mutational analysis demonstrates that ClC-4 and ClC-5 directly mediate plasma membrane currents. J. Biol. Chem. 274, 896–902 (1999)
Accardi, A. & Miller, C. Secondary active transport mediated by a prokaryotic homologue of ClC Cl- channels. Nature 427, 803–807 (2004)
Dutzler, R., Campbell, E. B., Cadene, M., Chait, B. T. & MacKinnon, R. X-ray structure of a ClC chloride channel at 3.0 Å reveals the molecular basis of anion selectivity. Nature 415, 287–294 (2002)
Lingueglia, E., Champigny, G., Lazdunski, M. & Barbry, P. Cloning of the amiloride-sensitive FMRFamide peptide-gated sodium channel. Nature 378, 730–733 (1995)
Poët, M. et al. Exploration of the pore structure of a peptide-gated Na+ channel. EMBO J. 20, 5595–5602 (2001)
Soleimani, M. et al. Pendrin: an apical Cl-/OH-/HCO3- exchanger in the kidney cortex. Am. J. Physiol. Renal Physiol. 280, F356–F364 (2001)
Roos, A. & Boron, W. F. Intracellular pH. Physiol. Rev. 61, 296–434 (1981)
Myers, V. B. & Haydon, D. A. Ion transfer across lipid membranes in the presence of gramicidin A. II. The ion selectivity. Biochim. Biophys. Acta 274, 313–322 (1972)
Li, X., Wang, T., Zhao, Z. & Weinman, S. A. The ClC-3 chloride channel promotes acidification of lysosomes in CHO-K1 and Huh-7 cells. Am. J. Physiol. Cell Physiol. 282, C1483–C1491 (2002)
Dutzler, R., Campbell, E. B. & MacKinnon, R. Gating the selectivity filter in ClC chloride channels. Science 300, 108–112 (2003)
Traverso, S., Elia, L. & Pusch, M. Gating competence of constitutively open CLC-0 mutants revealed by the interaction with a small organic inhibitor. J. Gen. Physiol. 122, 295–306 (2003)
Waldegger, S. & Jentsch, T. J. Functional and structural analysis of ClC-K chloride channels involved in renal disease. J. Biol. Chem. 275, 24527–24533 (2000)
Fahlke, C., Yu, H. T., Beck, C. L., Rhodes, T. H. & George, A. L. Jr Pore-forming segments in voltage-gated chloride channels. Nature 390, 529–532 (1997)
Günther, W., Piwon, N. & Jentsch, T. J. The ClC-5 chloride channel knock-out mouse — an animal model for Dent's disease. Pflügers Arch. 445, 456–462 (2003)
Stobrawa, S. M. et al. Disruption of ClC-3, a chloride channel expressed on synaptic vesicles, leads to a loss of the hippocampus. Neuron 29, 185–196 (2001)
Hara-Chikuma, M. et al. ClC-3 chloride channels facilitate endosomal acidification and chloride accumulation. J. Biol. Chem. 280, 1241–1247 (2005)
Yoshikawa, M. et al. CLC-3 deficiency leads to phenotypes similar to human neuronal ceroid lipofuscinosis. Genes Cells 7, 597–605 (2002)
Hara-Chikuma, M., Wang, Y., Guggino, S. E., Guggino, W. B. & Verkman, A. S. Impaired acidification in early endosomes of ClC-5 deficient proximal tubule. Biochem. Biophys. Res. Commun. 329, 941–946 (2005)
Davis-Kaplan, S. R., Askwith, C. C., Bengtzen, A. C., Radisky, D. & Kaplan, J. Chloride is an allosteric effector of copper assembly for the yeast multicopper oxidase Fet3p: an unexpected role for intracellular chloride channels. Proc. Natl Acad. Sci. USA 95, 13641–13645 (1998)
Jentsch, T. J., Stein, V., Weinreich, F. & Zdebik, A. A. Molecular structure and physiological function of chloride channels. Physiol. Rev. 82, 503–568 (2002)
Acknowledgements
We thank M. Lazdunski for the gift of the FaNaC-CD8 expression vector, and M. Petersen and P. Breiden for technical assistance. This work was supported in part by the Prix Louis-Jeantet de Médecine.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Scheel, O., Zdebik, A., Lourdel, S. et al. Voltage-dependent electrogenic chloride/proton exchange by endosomal CLC proteins. Nature 436, 424–427 (2005). https://doi.org/10.1038/nature03860
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature03860
- Springer Nature Limited
This article is cited by
-
Prenatal diagnosis of dent disease type I with a nonsense pathogenic variant in CLCN5: a case study
BMC Medical Genomics (2024)
-
Physiological roles of chloride ions in bodily and cellular functions
The Journal of Physiological Sciences (2023)
-
Proton-gated anion transport governs macropinosome shrinkage
Nature Cell Biology (2022)
-
Revealing novel synergistic defense and acid tolerant performance of Escherichia coli in response to organic acid stimulation
Applied Microbiology and Biotechnology (2022)
-
Clinical manifestation and genetic findings in three boys with low molecular Weight Proteinuria - three case reports for exploring Dent Disease and Fanconi syndrome
BMC Nephrology (2021)