Abstract
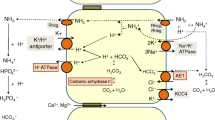
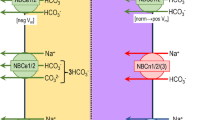
Dent’s disease is characterized by defective endocytosis in renal proximal tubules (PTs) and caused by mutations in the 2Cl−/H+ exchanger, CLC-5. However, the pathological role of endosomal acidification in endocytosis has recently come into question. To clarify the mechanism of pathogenesis for Dent’s disease, we examined the effects of a novel gating glutamate mutation, E211Q, on CLC-5 functions and endosomal acidification. In Xenopus oocytes, wild-type (WT) CLC-5 showed outward-rectifying currents that were inhibited by extracellular acidosis, but E211Q and an artificial pure Cl− channel mutant, E211A, showed linear currents that were insensitive to extracellular acidosis. Moreover, depolarizing pulse trains induced a robust reduction in the surface pH of oocytes expressing WT CLC-5 but not E211Q or E211A, indicating that the E211Q mutant functions as a pure Cl− channel similar to E211A. In HEK293 cells, E211A and E211Q stimulated endosomal acidification and hypotonicity-inducible vacuolar-type H+-ATPase (V-ATPase) activation at the plasma membrane. However, the stimulatory effects of these mutants were reduced compared with WT CLC-5. Furthermore, gene silencing experiments confirmed the functional coupling between V-ATPase and CLC-5 at the plasma membrane of isolated mouse PTs. These results reveal for the first time that the conversion of CLC-5 from a 2Cl−/H+ exchanger into a Cl− channel induces Dent’s disease in humans. In addition, defective endosomal acidification as a result of insufficient V-ATPase activation may still be important in the pathogenesis of Dent’s disease.









Similar content being viewed by others
References
Accardi A, Kolmakova-Partensky L, Williams C, Miller C (2004) Ionic currents mediated by a prokaryotic homologue of CLC Cl− channels. J Gen Physiol 123:109–19. doi:10.1085/jgp.200308935
Accardi A, Miller C (2004) Secondary active transport mediated by a prokaryotic homologue of ClC Cl− channels. Nature 427:803–7. doi:10.1038/nature02314
Amlal H, Goel A, Soleimani M (1998) Activation of H+-ATPase by hypotonicity: a novel regulatory mechanism for H+ secretion in IMCD cells. Am J Physiol 275:F487–501
Cao X, Yang Q, Qin J, Zhao S, Li X, Fan J, Chen W, Zhou Y, Mao H, Yu X (2012) V-ATPase promotes transforming growth factor-beta-induced epithelial-mesenchymal transition of rat proximal tubular epithelial cells. Am J Physiol Renal Physiol 302:F1121–32. doi:10.1152/ajprenal.00278.2011
Carraro-Lacroix LR, Lessa LM, Bezerra CN, Pessoa TD, Souza-Menezes J, Morales MM, Girardi AC, Malnic G (2010) Role of CFTR and ClC-5 in modulating vacuolar H+-ATPase activity in kidney proximal tubule. Cell Physiol Biochem 26:563–76. doi:10.1159/000322324
Claverie-Martin F, Ramos-Trujillo E, Garcia-Nieto V (2011) Dent’s disease: clinical features and molecular basis. Pediatr Nephrol 26:693–704. doi:10.1007/s00467-010-1657-0
D’Antonio C, Molinski S, Ahmadi S, Huan LJ, Wellhauser L, Bear CE (2013) Conformational defects underlie proteasomal degradation of Dent’s disease-causing mutants of ClC-5. Biochem J 452:391–400. doi:10.1042/BJ20121848
Dent CE, Friedman M (1964) Hypercalcuric rickets associated with renal tubular damage. Arch Dis Child 39:240–9
Devuyst O, Thakker RV (2010) Dent’s disease. Orphanet J Rare Dis 5:28. doi:10.1186/1750-1172-5-28
Dutzler R, Campbell EB, Cadene M, Chait BT, MacKinnon R (2002) X-ray structure of a ClC chloride channel at 3.0 A reveals the molecular basis of anion selectivity. Nature 415:287–94. doi:10.1038/415287a
Dutzler R, Campbell EB, MacKinnon R (2003) Gating the selectivity filter in ClC chloride channels. Science 300:108–12. doi:10.1126/science.1082708
Gleixner EM, Canaud G, Hermle T, Guida MC, Kretz O, Helmstadter M, Huber TB, Eimer S, Terzi F, Simons M (2014) V-ATPase/mTOR signaling regulates megalin-mediated apical endocytosis. Cell Rep 8:10–9. doi:10.1016/j.celrep.2014.05.035
Grand T, Mordasini D, L’Hoste S, Pennaforte T, Genete M, Biyeyeme MJ, Vargas-Poussou R, Blanchard A, Teulon J, Lourdel S (2009) Novel CLCN5 mutations in patients with Dent’s disease result in altered ion currents or impaired exchanger processing. Kidney Int 76:999–1005. doi:10.1038/ki.2009.305
Gunther W, Luchow A, Cluzeaud F, Vandewalle A, Jentsch TJ (1998) ClC-5, the chloride channel mutated in Dent’s disease, colocalizes with the proton pump in endocytotically active kidney cells. Proc Natl Acad Sci U S A 95:8075–80
Gunther W, Piwon N, Jentsch TJ (2003) The ClC-5 chloride channel knock-out mouse—an animal model for Dent’s disease. Pflugers Arch 445:456–62. doi:10.1007/s00424-002-0950-6
Hara-Chikuma M, Wang Y, Guggino SE, Guggino WB, Verkman AS (2005) Impaired acidification in early endosomes of ClC-5 deficient proximal tubule. Biochem Biophys Res Commun 329:941–6. doi:10.1016/j.bbrc.2005.02.060
Horita S, Yamada H, Inatomi J, Moriyama N, Sekine T, Igarashi T, Endo Y, Dasouki M, Ekim M, Al-Gazali L, Shimadzu M, Seki G, Fujita T (2005) Functional analysis of NBC1 mutants associated with proximal renal tubular acidosis and ocular abnormalities. J Am Soc Nephrol 16:2270–8. doi:10.1681/ASN.2004080667
Hurtado-Lorenzo A, Skinner M, El Annan J, Futai M, Sun-Wada GH, Bourgoin S, Casanova J, Wildeman A, Bechoua S, Ausiello DA, Brown D, Marshansky V (2006) V-ATPase interacts with ARNO and Arf6 in early endosomes and regulates the protein degradative pathway. Nat Cell Biol 8:124–36. doi:10.1038/ncb1348
Ishida Y, Nayak S, Mindell JA, Grabe M (2013) A model of lysosomal pH regulation. J Gen Physiol 141:705–20. doi:10.1085/jgp.201210930
Jentsch TJ, Steinmeyer K, Schwarz G (1990) Primary structure of Torpedo marmorata chloride channel isolated by expression cloning in Xenopus oocytes. Nature 348:510–4. doi:10.1038/348510a0
Kondo Y, Fromter E (1987) Axial heterogeneity of sodium-bicarbonate cotransport in proximal straight tubule of rabbit kidney. Pflugers Arch 410:481–6
Lloyd SE, Pearce SH, Fisher SE, Steinmeyer K, Schwappach B, Scheinman SJ, Harding B, Bolino A, Devoto M, Goodyer P, Rigden SP, Wrong O, Jentsch TJ, Craig IW, Thakker RV (1996) A common molecular basis for three inherited kidney stone diseases. Nature 379:445–9. doi:10.1038/379445a0
Lobet S, Dutzler R (2006) Ion-binding properties of the ClC chloride selectivity filter. EMBO J 25:24–33. doi:10.1038/sj.emboj.7600909
Lourdel S, Grand T, Burgos J, Gonzalez W, Sepulveda FV, Teulon J (2012) ClC-5 mutations associated with Dent’s disease: a major role of the dimer interface. Pflugers Arch 463:247–56. doi:10.1007/s00424-011-1052-0
Mellman I, Fuchs R, Helenius A (1986) Acidification of the endocytic and exocytic pathways. Annu Rev Biochem 55:663–700. doi:10.1146/annurev.bi.55.070186.003311
Miesenbock G, De Angelis DA, Rothman JE (1998) Visualizing secretion and synaptic transmission with pH-sensitive green fluorescent proteins. Nature 394:192–5. doi:10.1038/28190
Nakamura M, Yamazaki O, Shirai A, Horita S, Satoh N, Suzuki M, Hamasaki Y, Noiri E, Kume H, Enomoto Y, Homma Y, Seki G (2015) Preserved Na/HCO3 cotransporter sensitivity to insulin may promote hypertension in metabolic syndrome. Kidney Int 87:535–42. doi:10.1038/ki.2014.351
Nguitragool W, Miller C (2006) Uncoupling of a CLC Cl−/H+ exchange transporter by polyatomic anions. J Mol Biol 362:682–90. doi:10.1016/j.jmb.2006.07.006
Novarino G, Weinert S, Rickheit G, Jentsch TJ (2010) Endosomal chloride-proton exchange rather than chloride conductance is crucial for renal endocytosis. Science 328:1398–401. doi:10.1126/science.1188070
Picollo A, Malvezzi M, Accardi A (2010) Proton block of the CLC-5 Cl−/H+ exchanger. J Gen Physiol 135:653–9. doi:10.1085/jgp.201010428
Picollo A, Pusch M (2005) Chloride/proton antiporter activity of mammalian CLC proteins ClC-4 and ClC-5. Nature 436:420–3. doi:10.1038/nature03720
Piwon N, Gunther W, Schwake M, Bosl MR, Jentsch TJ (2000) ClC-5 Cl−-channel disruption impairs endocytosis in a mouse model for Dent’s disease. Nature 408:369–73. doi:10.1038/35042597
Rahmati N, Kunzelmann K, Xu J, Barone S, Sirianant L, De Zeeuw CI, Soleimani M (2013) Slc26a11 is prominently expressed in the brain and functions as a chloride channel: expression in Purkinje cells and stimulation of V H+-ATPase. Pflugers Arch 465:1583–97. doi:10.1007/s00424-013-1300-6
Sabolic I, Burckhardt G (1986) Characteristics of the proton pump in rat renal cortical endocytotic vesicles. Am J Physiol 250:F817–26
Sakamoto H, Sado Y, Naito I, Kwon TH, Inoue S, Endo K, Kawasaki M, Uchida S, Nielsen S, Sasaki S, Marumo F (1999) Cellular and subcellular immunolocalization of ClC-5 channel in mouse kidney: colocalization with H+-ATPase. Am J Physiol 277:F957–65
Scheel O, Zdebik AA, Lourdel S, Jentsch TJ (2005) Voltage-dependent electrogenic chloride/proton exchange by endosomal CLC proteins. Nature 436:424–7. doi:10.1038/nature03860
Schodel J, Klanke B, Weidemann A, Buchholz B, Bernhardt W, Bertog M, Amann K, Korbmacher C, Wiesener M, Warnecke C, Kurtz A, Eckardt KU, Willam C (2009) HIF-prolyl hydroxylases in the rat kidney: physiologic expression patterns and regulation in acute kidney injury. Am J Pathol 174:1663–74. doi:10.2353/ajpath.2009.080687
Seki G, Fromter E (1990) The chloride/base exchanger in the basolateral cell membrane of rabbit renal proximal tubule S3 segment requires bicarbonate to operate. Pflugers Arch 417:37–41
Sekine T, Komoda F, Miura K, Takita J, Shimadzu M, Matsuyama T, Ashida A, Igarashi T (2014) Japanese Dent disease has a wider clinical spectrum than Dent disease in Europe/USA: genetic and clinical studies of 86 unrelated patients with low-molecular-weight proteinuria. Nephrol Dial Transplant 29:376–84. doi:10.1093/ndt/gft394
Shirakabe K, Priori G, Yamada H, Ando H, Horita S, Fujita T, Fujimoto I, Mizutani A, Seki G, Mikoshiba K (2006) IRBIT, an inositol 1,4,5-trisphosphate receptor-binding protein, specifically binds to and activates pancreas-type Na+/HCO3 − cotransporter 1 (pNBC1). Proc Natl Acad Sci U S A 103:9542–7. doi:10.1073/pnas.0602250103
Smith AJ, Lippiat JD (2010) Direct endosomal acidification by the outwardly rectifying CLC-5 Cl−/H+ exchanger. J Physiol 588:2033–45. doi:10.1113/jphysiol.2010.188540
Smith AJ, Reed AA, Loh NY, Thakker RV, Lippiat JD (2009) Characterization of Dent’s disease mutations of CLC-5 reveals a correlation between functional and cell biological consequences and protein structure. Am J Physiol Renal Physiol 296:F390–7. doi:10.1152/ajprenal.90526.2008
Sonawane ND, Thiagarajah JR, Verkman AS (2002) Chloride concentration in endosomes measured using a ratioable fluorescent Cl− indicator: evidence for chloride accumulation during acidification. J Biol Chem 277:5506–13. doi:10.1074/jbc.M110818200
Stauber T, Jentsch TJ (2013) Chloride in vesicular trafficking and function. Annu Rev Physiol 75:453–77. doi:10.1146/annurev-physiol-030212-183702
Stauber T, Weinert S, Jentsch TJ (2012) Cell biology and physiology of CLC chloride channels and transporters. Compr Physiol 2:1701–44. doi:10.1002/cphy.c110038
Steinmeyer K, Schwappach B, Bens M, Vandewalle A, Jentsch TJ (1995) Cloning and functional expression of rat CLC-5, a chloride channel related to kidney disease. J Biol Chem 270:31172–7
Suzuki M, Vaisbich MH, Yamada H, Horita S, Li Y, Sekine T, Moriyama N, Igarashi T, Endo Y, Cardoso TP, de Sa LC, Koch VH, Seki G, Fujita T (2008) Functional analysis of a novel missense NBC1 mutation and of other mutations causing proximal renal tubular acidosis. Pflugers Arch 455:583–93. doi:10.1007/s00424-007-0319-y
Thakker RV (2000) Pathogenesis of Dent’s disease and related syndromes of X-linked nephrolithiasis. Kidney Int 57:787–93. doi:10.1046/j.1523-1755.2000.00916.x
Wang SS, Devuyst O, Courtoy PJ, Wang XT, Wang H, Wang Y, Thakker RV, Guggino S, Guggino WB (2000) Mice lacking renal chloride channel, CLC-5, are a model for Dent’s disease, a nephrolithiasis disorder associated with defective receptor-mediated endocytosis. Hum Mol Genet 9:2937–45
Weinert S, Jabs S, Supanchart C, Schweizer M, Gimber N, Richter M, Rademann J, Stauber T, Kornak U, Jentsch TJ (2010) Lysosomal pathology and osteopetrosis upon loss of H+-driven lysosomal Cl- accumulation. Science 328:1401–3. doi:10.1126/science.1188072
Wrong OM, Norden AG, Feest TG (1994) Dent’s disease; a familial proximal renal tubular syndrome with low-molecular-weight proteinuria, hypercalciuria, nephrocalcinosis, metabolic bone disease, progressive renal failure and a marked male predominance. QJM 87:473–93
Wu F, Roche P, Christie PT, Loh NY, Reed AA, Esnouf RM, Thakker RV (2003) Modeling study of human renal chloride channel (hCLC-5) mutations suggests a structural-functional relationship. Kidney Int 63:1426–32. doi:10.1046/j.1523-1755.2003.00859.x
Yamazaki O, Yamada H, Suzuki M, Horita S, Shirai A, Nakamura M, Satoh N, Fujita T, Seki G (2013) Identification of dominant negative effect of L522P mutation in the electrogenic Na+-HCO3 − cotransporter NBCe1. Pflugers Arch 465:1281–91. doi:10.1007/s00424-013-1277-1
Yamazaki O, Yamada H, Suzuki M, Horita S, Shirai A, Nakamura M, Seki G, Fujita T (2011) Functional characterization of nonsynonymous single nucleotide polymorphisms in the electrogenic Na+-HCO3 − cotransporter NBCe1A. Pflugers Arch 461:249–59. doi:10.1007/s00424-010-0918-x
Zifarelli G, De Stefano S, Zanardi I, Pusch M (2012) On the mechanism of gating charge movement of ClC-5, a human Cl−/H+ antiporter. Biophys J 102:2060–9. doi:10.1016/j.bpj.2012.03.067
Zifarelli G, Pusch M (2009) Conversion of the 2 Cl−/1 H+ antiporter ClC-5 in a NO3 −/H+ antiporter by a single point mutation. EMBO J 28:175–82. doi:10.1038/emboj.2008.284
Zoncu R, Bar-Peled L, Efeyan A, Wang S, Sancak Y, Sabatini DM (2011) mTORC1 senses lysosomal amino acids through an inside-out mechanism that requires the vacuolar H+-ATPase. Science 334:678–83. doi:10.1126/science.1207056
Acknowledgments
This study was supported in part by grants from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Daisuke Yamamoto deceased.
Parts of this paper were taken from the thesis written in English by Nobuhiko Satoh. The title of the thesis, which is in Japanese, is as follows: “CLC-5の2Cl/H交換輸送機能はV-ATPaseを介する効率的エンドゾーム酸性化に必要である”. The summary (in Japanese) of the thesis is accessible at http://repository.dl.itc.u-tokyo.ac.jp/index_e.html.
Rights and permissions
About this article
Cite this article
Satoh, N., Yamada, H., Yamazaki, O. et al. A pure chloride channel mutant of CLC-5 causes Dent’s disease via insufficient V-ATPase activation. Pflugers Arch - Eur J Physiol 468, 1183–1196 (2016). https://doi.org/10.1007/s00424-016-1808-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-016-1808-7