Abstract
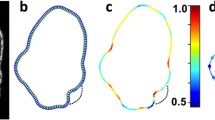
Current models for protrusive motility in animal cells focus on cytoskeleton-based mechanisms, where localized protrusion is driven by local regulation of actin biochemistry1,2,3. In plants and fungi, protrusion is driven primarily by hydrostatic pressure4,5,6. For hydrostatic pressure to drive localized protrusion in animal cells7,8, it would have to be locally regulated, but current models treating cytoplasm as an incompressible viscoelastic continuum9 or viscous liquid10 require that hydrostatic pressure equilibrates essentially instantaneously over the whole cell. Here, we use cell blebs as reporters of local pressure in the cytoplasm. When we locally perfuse blebbing cells with cortex-relaxing drugs to dissipate pressure on one side, blebbing continues on the untreated side, implying non-equilibration of pressure on scales of approximately 10 µm and 10 s. We can account for localization of pressure by considering the cytoplasm as a contractile, elastic network infiltrated by cytosol. Motion of the fluid relative to the network generates spatially heterogeneous transients in the pressure field, and can be described in the framework of poroelasticity11,12.




Similar content being viewed by others
References
Mahadevan, L. & Matsudaira, P. Motility powered by supramolecular springs and ratchets. Science 288, 95–100 (2000)
Rafelski, S. M. & Theriot, J. A. Crawling toward a unified model of cell mobility: spatial and temporal regulation of actin dynamics. Annu. Rev. Biochem. 73, 209–239 (2004)
Mogilner, A. & Oster, G. Polymer motors: pushing out the front and pulling up the back. Curr. Biol. 13, R721–R733 (2003)
Messerli, M. A. & Robinson, K. R. Ionic and osmotic disruptions of the lily pollen tube oscillator: testing proposed models. Planta 217, 147–157 (2003)
Money, N. P. & Harold, F. M. Extension growth of the water mold Achlya: interplay of turgor and wall strength. Proc. Natl Acad. Sci. USA 89, 4245–4249 (1992)
Harold, F. M. Force and compliance: rethinking morphogenesis in walled cells. Fungal Genet. Biol. 37, 271–282 (2002)
Tilney, L. G. & Inoue, S. Acrosomal reaction of the Thyone sperm. III. The relationship between actin assembly and water influx during the extension of the acrosomal process. J. Cell Biol. 100, 1273–1283 (1985)
Condeelis, J. Life at the leading edge: the formation of cell protrusions. Annu. Rev. Cell Biol. 9, 411–444 (1993)
Boal, D. H. Mechanics of the Cell (Cambridge Univ. Press, Cambridge, UK, 2002)
Drury, J. L. & Dembo, M. Hydrodynamics of micropipette aspiration. Biophys. J. 76, 110–128 (1999)
Biot, M. General theory of three-dimensional consolidation. J. Appl. Phys. 12, 155–164 (1941)
Wang, H. Theory of Linear Poroelasticity with Applications to Geomechanics and Hydrogeology (Princeton Univ. Press, Princeton, New Jersey, 2000)
Mills, J. C., Stone, N. L., Erhardt, J. & Pittman, R. N. Apoptotic membrane blebbing is regulated by myosin light chain phosphorylation. J. Cell Biol. 140, 627–636 (1998)
Fishkind, D. J., Cao, L. G. & Wang, Y. L. Microinjection of the catalytic fragment of myosin light chain kinase into dividing cells: effects on mitosis and cytokinesis. J. Cell Biol. 114, 967–975 (1991)
Burton, K. & Taylor, D. L. Traction forces of cytokinesis measured with optically modified elastic substrata. Nature 385, 450–454 (1997)
Trinkaus, J. P. Surface activity and locomotion of Fundulus deep cells during blastula and gastrula stages. Dev. Biol. 30, 69–103 (1973)
Friedl, P. & Wolf, K. Tumour-cell invasion and migration: diversity and escape mechanisms. Nature Rev. Cancer 3, 362–374 (2003)
Albrecht-Buehler, G. Does blebbing reveal the convulsive flow of liquid and solutes through the cytoplasmic meshwork? Cold Spring Harb. Symp. Quant. Biol. 46, 45–49 (1982)
Cunningham, C. C. Actin polymerization and intracellular solvent flow in cell surface blebbing. J. Cell Biol. 129, 1589–1599 (1995)
Cunningham, C. C. et al. Actin-binding protein requirement for cortical stability and efficient locomotion. Science 255, 325–327 (1992)
Cheung, A. et al. A small-molecule inhibitor of skeletal muscle myosin II. Nature Cell Biol. 4, 83–88 (2002)
Dai, J. & Sheetz, M. P. Membrane tether formation from blebbing cells. Biophys. J. 77, 3363–3370 (1999)
Evans, E. & Leung, A. Adhesivity and rigidity of erythrocyte membrane in relation to wheat germ agglutinin binding. J. Cell Biol. 98, 1201–1208 (1984)
Popov, S., Brown, A. & Poo, M. M. Forward plasma membrane flow in growing nerve processes. Science 259, 244–246 (1993)
O'Connell, C. B., Warner, A. K. & Wang, Y. Distinct roles of the equatorial and polar cortices in the cleavage of adherent cells. Curr. Biol. 11, 702–707 (2001)
Yarrow, J. C., Totsukawa, G., Charras, G. T. & Mitchison, T. J. Screening for cell migration inhibitors via automated microscopy reveals a rho-kinase inhibitor. Chem. Biol. 12, 385–395 (2005)
Takayama, S. et al. Selective chemical treatment of cellular microdomains using multiple laminar streams. Chem. Biol. 10, 123–130 (2003)
Zicha, D. et al. Rapid actin transport during cell protrusion. Science 300, 142–145 (2003)
Baumgartner, M., Patel, H. & Barber, D. L. Na(+ )/H(+ ) exchanger NHE1 as plasma membrane scaffold in the assembly of signaling complexes. Am. J. Physiol. Cell Physiol. 287, C844–C850 (2004)
Acknowledgements
The authors would like to acknowledge the Nikon Imaging Centre at Harvard Medical School and, in particular, J. Waters. The authors would also like to acknowledge J. Horn at the HMS machine shop for manufacturing the perfusion chamber. G.T.C. was in receipt of a Wellcome Trust Overseas Fellowship. M.A.H. is supported by a Programme Grant from the Wellcome Trust. L.M. was supported by NSF-MRSEC at Harvard University. This work was supported by a grant from the NIH to T.J.M.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Notes
This contains Supplementary Discussion, Supplementary Methods, Supplementary Table 1 and Legends to accompany the Supplementary Figures S1-S4 and Supplementary Videos S1-S12. (DOC 202 kb)
Supplementary Figure S1
Dose response of bleb diameter and frequency with increasing osmotic pressure (PDF 164 kb)
Supplementary Figure S2
Distribution of maximum bleb expansion velocities and minimum bleb retraction velocities. (PDF 168 kb)
Supplementary Figure S3
Dissassembly of the actin cortex posterior to bleb expansion (PDF 150 kb)
Supplementary Figure S4
Volume fluctuations in a blebbing microplast (PDF 121 kb)
Supplementary Video S1
Bath treatment of cells with high concentration sucrose (MOV 4519 kb)
Supplementary Video S2
Bath treatment of cells with WGA (MOV 4872 kb)
Supplementary Video S3
Local perfusion of WGA (MOV 3031 kb)
Supplementary Video S4
Local perfusion of Sucrose (MOV 3697 kb)
Supplementary Video S5
Local perfusion of Blebbistatin (MOV 4290 kb)
Supplementary Video S6
Local perfusion of 3-(4-pyridyl)indole (MOV 4789 kb)
Supplementary Video S7
Local perfusion of Latrunculin (MOV 4233 kb)
Supplementary Video S8
Local perfusion of Staurosporine (MOV 4858 kb)
Supplementary Video S9
Local perfusion of HA 1077 (MOV 5119 kb)
Supplementary Video S10
Colocalisation of actin and myosin II at the cell cortex (MOV 3470 kb)
Supplementary Video S11
MRLC localisation during blebbistatin treatment (MOV 4861 kb)
Supplementary Video S12
Dose response of bleb diameter and frequency with increasing osmotic pressure (MOV 401 kb)
Rights and permissions
About this article
Cite this article
Charras, G., Yarrow, J., Horton, M. et al. Non-equilibration of hydrostatic pressure in blebbing cells. Nature 435, 365–369 (2005). https://doi.org/10.1038/nature03550
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature03550
- Springer Nature Limited
This article is cited by
-
Apoptotic bodies: bioactive treasure left behind by the dying cells with robust diagnostic and therapeutic application potentials
Journal of Nanobiotechnology (2023)
-
Membrane tension induces F-actin reorganization and flow in a biomimetic model cortex
Communications Biology (2023)
-
Drosophila motor neuron boutons remodel through membrane blebbing coupled with muscle contraction
Nature Communications (2023)
-
Membrane curvature governs the distribution of Piezo1 in live cells
Nature Communications (2022)
-
Heterologously secreted MbxA from Moraxella bovis induces a membrane blebbing response of the human host cell
Scientific Reports (2022)