Abstract
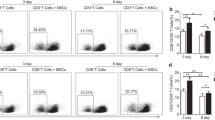
Mesenchymal stromal cells (MSCs) possess broad immunomodulatory capacities that are currently investigated for potential clinical application in treating autoimmune disorders. Third-party MSCs suppress alloantigen-induced proliferation of peripheral blood mononuclear cells providing the rationale for clinical use in graft-versus-host disease (GvHD). We confirmed that MSCs strongly inhibited proliferation of CD8+ T cells in a mixed lymphocyte reaction. However, MSCs also suppressed proliferation of T cells specifically recognizing cytomegalovirus (CMV) and influenza virus. Inhibition was dose dependent, but independent of the culture medium. MSCs inhibited proliferation of specific CD8+ T cells and the release of IFN-γ by specific CD8+ T cells for immunodominant HLA-A2- and HLA-B7- restricted antigen epitopes derived from CMV phosphoprotein 65 and influenza matrix protein. This is in contrast to a recently reported scenario where MSCs exert differential effects on alloantigen and virus-specific T cells potentially having an impact on surveillance and prophylaxis of patients treated by MSCs.





Similar content being viewed by others
References
Friedenstein AJ, Deriglasova UF, Kulagina NN, Panasuk AF, Rudakowa SF, Luria EA et al. Precursors for fibroblasts in different populations of hematopoietic cells as detected by the in vitro colony assay method. Exp Hematol 1974; 2: 83–92.
Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD et al. Multilineage potential of adult human mesenchymal stem cells. Science 1999; 284: 143–147.
Di Nicola M, Carlo-Stella C, Magni M, Milanesi M, Longoni PD, Matteucci P et al. Human bone marrow stromal cells suppress T-lymphocyte proliferation induced by cellular or nonspecific mitogenic stimuli. Blood 2002; 99: 3838–3843.
Tse WT, Pendleton JD, Beyer WM, Egalka MC, Guinan EC . Suppression of allogeneic T-cell proliferation by human marrow stromal cells: implications in transplantation. Transplantation 2003; 75: 389–397.
Le Blanc K, Frassoni F, Ball L, Locatelli F, Roelofs H, Lewis I et al. Mesenchymal stem cells for treatment of steroid-resistant, severe, acute graft-versus-host disease: a phase II study. Lancet 2008; 371: 1579–1586.
Le Blanc K, Rasmusson I, Sundberg B, Gotherstrom C, Hassan M, Uzunel M et al. Treatment of severe acute graft-versus-host disease with third party haploidentical mesenchymal stem cells. Lancet 2004; 363: 1439–1441.
Galipeau J . The mesenchymal stromal cells dilemma—does a negative phase III trial of random donor mesenchymal stromal cells in steroid-resistant graft-versus-host disease represent a death knell or a bump in the road? Cytotherapy 2013; 15: 2–8.
Abdelrazik H, Spaggiari GM, Chiossone L, Moretta L . Mesenchymal stem cells expanded in human platelet lysate display a decreased inhibitory capacity on T- and NK-cell proliferation and function. Eur J Immunol 2011; 41: 3281–3290.
Doucet C, Ernou I, Zhang Y, Llense JR, Begot L, Holy X et al. Platelet lysates promote mesenchymal stem cell expansion: a safety substitute for animal serum in cell-based therapy applications. J Cell Physiol 2005; 205: 228–236.
Bernardo ME, Avanzini MA, Perotti C, Cometa AM, Moretta A, Lenta E et al. Optimization of in vitro expansion of human multipotent mesenchymal stromal cells for cell-therapy approaches: further insights in the search for a fetal calf serum substitute. J Cell Physiol 2007; 211: 121–130.
Perez-Ilzarbe M, Diez-Campelo M, Aranda P, Tabera S, Lopez T, del Canizo C et al. Comparison of ex vivo expansion culture conditions of mesenchymal stem cells for human cell therapy. Transfusion 2009; 49: 1901–1910.
Ludwig A, Saffrich R, Eckstein V, Bruckner T, Wagner W, Ho AD et al. Functional potentials of human hematopoietic progenitor cells are maintained by mesenchymal stromal cells and not impaired by plerixafor. Cytotherapy 2014; 16: 111–121.
Jin N, Malcherek G, Mani J, Zurleit R, Schmitt A, Chen B et al. Suppression of cytomegalovirus-specific CD8+ T cells by everolimus. Leuk Lymphoma 2014; 55: 1144–1150.
Karlsson H, Samarasinghe S, Ball LM, Sundberg B, Lankester AC, Dazzi F et al. Mesenchymal stem cells exert differential effects on alloantigen and virus-specific T-cell responses. Blood 2008; 112: 532–541.
Sundin M, Orvell C, Rasmusson I, Sundberg B, Ringden O, Le Blanc K . Mesenchymal stem cells are susceptible to human herpesviruses, but viral DNA cannot be detected in the healthy seropositive individual. Bone Marrow Transplant 2006; 37: 1051–1059.
Rasmusson I, Uhlin M, Le Blanc K, Levitsky V . Mesenchymal stem cells fail to trigger effector functions of cytotoxic T lymphocytes. J Leukoc Biol 2007; 82: 887–893.
Rasmusson I, Ringden O, Sundberg B, Le Blanc K . Mesenchymal stem cells inhibit the formation of cytotoxic T lymphocytes, but not activated cytotoxic T lymphocytes or natural killer cells. Transplantation 2003; 76: 1208–1213.
Bartholomew A, Sturgeon C, Siatskas M, Ferrer K, McIntosh K, Patil S et al. Mesenchymal stem cells suppress lymphocyte proliferation in vitro and prolong skin graft survival in vivo. Exp Hematol 2002; 30: 42–48.
Le Blanc K, Tammik L, Sundberg B, Haynesworth SE, Ringden O . Mesenchymal stem cells inhibit and stimulate mixed lymphocyte cultures and mitogenic responses independently of the major histocompatibility complex. Scand J Immunol 2003; 57: 11–20.
Aggarwal S, Pittenger MF . Human mesenchymal stem cells modulate allogeneic immune cell responses. Blood 2005; 105: 1815–1822.
Corcione A, Benvenuto F, Ferretti E, Giunti D, Cappiello V, Cazzanti F et al. Human mesenchymal stem cells modulate B-cell functions. Blood 2006; 107: 367–372.
Kim N, Im KI, Lim JY, Jeon EJ, Nam YS, Kim EJ et al. Mesenchymal stem cells for the treatment and prevention of graft-versus-host disease: experiments and practice. Ann Hematol 2013; 92: 1295–1308.
Jiang XX, Zhang Y, Liu B, Zhang SX, Wu Y, Yu XD et al. Human mesenchymal stem cells inhibit differentiation and function of monocyte-derived dendritic cells. Blood 2005; 105: 4120–4126.
Copland IB, Galipeau J . Death and inflammation following somatic cell transplantation. Semin Immunopathol 2011; 33: 535–550.
Chinnadurai R, Copland IB, Patel SR, Galipeau J . IDO-independent suppression of T cell effector function by IFN-gamma-licensed human mesenchymal stromal cells. J Immunol 2014; 192: 1491–1501.
Liu Y, Wang L, Kikuiri T, Akiyama K, Chen C, Xu X et al. Mesenchymal stem cell-based tissue regeneration is governed by recipient T lymphocytes via IFN-gamma and TNF-alpha. Nat Med 2011; 17: 1594–1601.
Krampera M . Mesenchymal stromal cell ‘licensing’: a multistep process. Leukemia 2011; 25: 1408–1414.
Hagmann S, Moradi B, Frank S, Dreher T, Kammerer PW, Richter W et al. Different culture media affect growth characteristics, surface marker distribution and chondrogenic differentiation of human bone marrow-derived mesenchymal stromal cells. BMC Musculoskelet Disord 2013; 14: 223.
Introna M, Lucchini G, Dander E, Galimberti S, Rovelli A, Balduzzi A et al. Treatment of graft versus host disease with mesenchymal stromal cells: a phase I study on 40 adult and pediatric patients. Biol Blood Marrow Transplant 2014; 20: 375–381.
von Bahr L, Sundberg B, Lonnies L, Sander B, Karbach H, Hagglund H et al. Long-term complications, immunologic effects, and role of passage for outcome in mesenchymal stromal cell therapy. Biol Blood Marrow Transplant 2012; 18: 557–564.
Acknowledgements
We acknowledge the NIH Tetramer Core Facility (contract HHSN272201300006C), for provision of HLA-A*0201/pp65 (495–503)-tetramer and TC Metrix for HLA-A*0201/IMP (58–66)- and HLA-B*0702 tetramers. This work was supported by the German Ministry of Education and Research (BMBF) as part of the supporting program ‘Cell-Based Regenerative Medicine’ (START-MSC2; funding code 01GN0940 to PW and ADH) and as part of the collaborative research project ‘Systems Biology of Erythropoietin’ (SBEpo; funding code 0316182D to PW and ADH). This work was also supported by the Thiele-Stiftung (funding to ADH) and the German Research Foundation (SFB 873; SP B7 to ADH).
Author Contributions
GM designed and supervised the original research and wrote the original manuscript; NJ performed most of the research and analyzed the data; AH designed new experiments, analyzed data and wrote parts of the resubmitted manuscript; JM, LW and UG performed some research; AS discussed and analyzed flow cytometry data; PW established MSC culture protocols and discussed the manuscript, AD supplied MSC cultures; ADH and BC discussed the manuscript; MS initiated the study, discussed the research and wrote parts of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
PW received honorarium for lectures from Sanofi and consulting fee or honorarium from ETICHO. The remaining authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Leukemia website
Supplementary information
Rights and permissions
About this article
Cite this article
Malcherek, G., Jin, N., Hückelhoven, A. et al. Mesenchymal stromal cells inhibit proliferation of virus-specific CD8+ T cells. Leukemia 28, 2388–2394 (2014). https://doi.org/10.1038/leu.2014.273
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2014.273
- Springer Nature Limited
This article is cited by
-
Transfer of mesenchymal stem cell mitochondria to CD4+ T cells contributes to repress Th1 differentiation by downregulating T-bet expression
Stem Cell Research & Therapy (2023)
-
Stem cell therapy: a novel approach against emerging and re-emerging viral infections with special reference to SARS-CoV-2
Molecular Biology Reports (2023)
-
Multipotent Stromal Cells and Viral Interaction: Current Implications for Therapy
Stem Cell Reviews and Reports (2022)
-
Nanoconjugates-Based Stem Cell Therapy for the Management of COVID-19
Stem Cell Reviews and Reports (2021)
-
Stem cell therapy: a potential approach for treatment of influenza virus and coronavirus-induced acute lung injury
Stem Cell Research & Therapy (2020)