Abstract
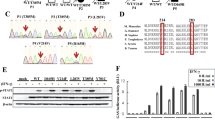
We evaluated the MYD88 L265P mutation in Waldenström’s macroglobulinemia (WM) and B-cell lymphoproliferative disorders by specific polymerase chain reaction (PCR) (sensitivity ∼10−3). No mutation was seen in normal donors, while it was present in 101/117 (86%) WM patients, 27/31 (87%) IgM monoclonal gammapathies of uncertain significance (MGUS), 3/14 (21%) splenic marginal zone lymphomas and 9/48 (19%) non-germinal center (GC) diffuse large B-cell lymphomas (DLBCLs). The mutation was absent in all 28 GC-DLBCLs, 13 DLBCLs not subclassified, 35 hairy cell leukemias, 39 chronic lymphocytic leukemias (16 with M-component), 25 IgA or IgG-MGUS, 24 multiple myeloma (3 with an IgM isotype), 6 amyloidosis, 9 lymphoplasmacytic lymphomas and 1 IgM-related neuropathy. Among WM and IgM-MGUS, MYD88 L265P mutation was associated with some differences in clinical and biological characteristics, although usually minor; wild-type MYD88 cases had smaller M-component (1.77 vs 2.72 g/dl, P=0.022), more lymphocytosis (24 vs 5%, P=0.006), higher lactate dehydrogenase level (371 vs 265 UI/L, P=0.002), atypical immunophenotype (CD23−CD27++FMC7++), less Immunoglobulin Heavy Chain Variable gene (IGHV) somatic hypermutation (57 vs 97%, P=0.012) and less IGHV3–23 gene selection (9 vs 27%, P=0.014). These small differences did not lead to different time to first therapy, response to treatment or progression-free or overall survival.

Similar content being viewed by others
References
Groves FD, Travis LB, Devesa SS, Ries LA, Fraumeni JF . Waldenstrom's macroglobulinemia: incidence patterns in the United States, 1988–1994. Cancer 1998; 82: 1078–1081.
Herrinton LJ, Weiss NS . Incidence of Waldenstrom's macroglobulinemia. Blood 1993; 82: 3148–3150.
Garcia-Sanz R, Montoto S, Torrequebrada A, de Coca AG, Petit J, Sureda A et al. Waldenstrom macroglobulinaemia: presenting features and outcome in a series with 217 cases. Br J Haematol 2001; 115: 575–582.
Phekoo KJ, Jack RH, Davies E, M°ller H, Schey SA . The incidence and survival of Waldenstrom's macroglobulinaemia in South East England. Leukemia Res 2008; 32: 55–59.
Braggio E, Philipsborn C, Novak A, Hodge L, Ansell S, Fonseca R . Molecular pathogenesis of Waldenström macroglobulinemia. Haematologica 2012; 97: 1281–1290.
Treon SP, Xu L, Yang G, Zhou Y, Liu X, Cao Y et al. MYD88 L265P somatic mutation in Waldenström's macroglobulinemia. N Engl J M 2012; 367: 826–833.
Ngo VN, Young RM, Schmitz R, Jhavar S, Xiao W, Lim KH et al. Oncogenically active MYD88 mutations in human lymphoma. Nature 2011; 470: 115–119.
Lohr JG, Stojanov P, Lawrence MS, Auclair D, Chapuy B, Sougnez C et al. Discovery and prioritization of somatic mutations in diffuse large B-cell lymphoma (DLBCL) by whole-exome sequencing. Proc Natl Acad Sci 2012; 109: 3879–3884.
Pasqualucci L, Trifonov V, Fabbri G, Ma J, Rossi D, Chiarenza A et al. Analysis of the coding genome of diffuse large B-cell lymphoma. Nat Genet 2011; 43: 830–837.
Pham-Ledard A, Cappellen D, Martinez F, Vergier B, Beylot-Barry M, Merlio JP . myd88 somatic mutation is a genetic feature of primary cutaneous diffuse large B-cell lymphoma, Leg type. J Invest Dermatol 2012; 132: 2118–2120.
Gonzalez-Aguilar A, Idbaih A, Boisselier B, Habbita N, Rossetto M, Laurenge A et al. Recurrent mutations of MYD88 and TBL1XR1 in primary central nervous system lymphomas. Clin Cancer Res 2012; 18: 5203–5211.
Montesinos-Rongen M, Godlewska E, Brunn A, Wiestler OD, Siebert R, Deckert M . Activating L265P mutations of the MYD88 gene are common in primary central nervous system lymphoma. Acta Neuropathol 2011; 122: 791–792.
Ocio EM, Del CD, Caballero A, Alonso J, Paiva B, Pesoa R et al. Differential diagnosis of IgM MGUS and WM according to B-lymphoid infiltration by morphology and flow cytometry. Clin Lymphoma Myeloma Leuk 2011; 11: 93–95.
Billadeau D, Quam L, Thomas W, Kay N, Greipp P, Kyle R et al. Detection and quantitation of malignant cells in the peripheral blood of multiple myeloma patients. Blood 1992; 80: 1818–1824.
Campo E, Swerdlow SH, Harris NL, Pileri S, Stein H, Jaffe ES . The 2008 WHO classification of lymphoid neoplasms and beyond: evolving concepts and practical applications. Blood 2011; 117: 5019–5032.
Owen RG, Treon SP, Al Katib A, Fonseca R, Greipp PR, McMaster ML et al. Clinicopathological definition of Waldenstrom's macroglobulinemia: consensus panel recommendations from the Second International Workshop on Waldenstrom's Macroglobulinemia. Semin Oncol 2003; 30: 110–115.
Paiva B, Vidriales MB, Perez JJ, Mateo G, Montalban MA, Mateos MV et al. Multiparameter flow cytometry quantification of bone marrow plasma cells at diagnosis provides more prognostic information than morphological assessment in myeloma patients. Haematologica 2009; 94: 1599–1602.
Garcia-Sanz R, Ocio E, Caballero A, Magalhaes RJ, Alonso J, Lopez-Anglada L et al. Post-treatment bone marrow residual disease>5% by flow cytometry is highly predictive of short progression-free and overall survival in patients with Waldenstrom's macroglobulinemia. Clin Lymphoma Myeloma Leuk 2011; 11: 168–171.
Rawstron AC, Orfao A, Beksac M, Bezdickova L, Brooimans RA, Bumbea H et al. Report of the European Myeloma Network on multiparametric flow cytometry in multiple myeloma and related disorders. Haematologica 2008; 93: 431–438.
van Dongen JJ, Lhermitte L, Bottcher S, Almeida J, van dV V, Flores-Montero J et al. EuroFlow antibody panels for standardized n-dimensional flow cytometric immunophenotyping of normal, reactive and malignant leukocytes. Leukemia 2012; 26: 1908–1975.
Pedreira CE, Costa ES, Almeida J, Fernandez C, Quijano S, Flores J et al. A probabilistic approach for the evaluation of minimal residual disease by multiparameter flow cytometry in leukemic B-cell chronic lymphoproliferative disorders. Cytometry A 2008; 73A: 1141–1150.
Ocio EM, Schop RF, Gonzalez B, Van Wier SA, Hernandez-Rivas JM, Gutierrez NC et al. 6q deletion in Waldenstrom macroglobulinemia is associated with features of adverse prognosis. Br J Haematol 2007; 136: 80–86.
van Dongen JJ, Langerak AW, Bruggemann M, Evans PA, Hummel M, Lavender FL et al. Design and standardization of PCR primers and protocols for detection of clonal immunoglobulin and T-cell receptor gene recombinations in suspect lymphoproliferations: report of the BIOMED-2 Concerted Action BMH4-CT98-3936. Leukemia 2003; 17: 2257–2317.
Martin-Jimenez P, Garcia-Sanz R, Balanzategui A, Alcoceba M, Ocio E, Sanchez ML et al. Molecular characterization of heavy chain immunoglobulin gene rearrangements in Waldenstrom's macroglobulinemia and IgM monoclonal gammopathy of undetermined significance. Haematologica 2007; 92: 635–642.
Palumbo A, Anderson K . Multiple myeloma. N Engl J Med 2011; 364: 1046–1060.
Treon SP . How I treat Waldenstrom macroglobulinemia. Blood 2009; 114: 2375–2385.
Merlini G, Stone MJ . Dangerous small B-cell clones. Blood 2006; 108: 2520–2530.
Kyle RA, Buadi F, Rajkumar SV . Management of monoclonal gammopathy of undetermined significance (MGUS) and smoldering multiple myeloma (SMM). Oncology (Williston Park) 2011; 25: 578–586.
Gachard N, Parrens M, Soubeyran I, Petit B, Marfak A, Rizzo D et al. IGHV gene features and MYD88 L265P mutation separate the three marginal zone lymphoma entities and Waldenstrom macroglobulinemia/lymphoplasmacytic lymphomas. Leukemia 2013; 27: 183–189.
Varettoni M, Arcaini L, Zibellini S, Boveri E, Rattotti S, Riboni R et al. Prevalence and clinical significance of the MYD88 (L265P) somatic mutation in Waldenstrm's macroglobulinemia and related lymphoid neoplasms. Blood 2013; e-pub ahead of print 25 January 2013.
Wang CZ, Lin J, Qian J, Shao R, Xue D, Qian W et al. Development of high-resolution melting analysis for the detection of the MYD88 L265P mutation. Clin Biochem 2012; 46: 385–387.
Xu L, Hunter ZR, Yang G, Zhou Y, Cao Y, Liu X et al. MYD88 L265P in Waldenström's macroglobulinemia, IgM monoclonal gammopathy, and other B-cell lymphoproliferative disorders using conventional and quantitative allele-specific PCR. Blood 2013; e-pub ahead of print 15 January 2013.
Landgren O, Staudt LM . MYD88 L265P Somatic Mutation in IgM MGUS. N Engl J Med 2012; 367: 2255–2257.
Je EM, Yoo NJ, Lee SH . Absence of MYD88 gene mutation in acute leukemias and multiple myelomas. Eur J Haematol 2012; 88: 273–274.
Puente XS, Pinyol M, Quesada V, Conde L, Ordonez GR, Villamor N et al. Whole-genome sequencing identifies recurrent mutations in chronic lymphocytic leukaemia. Nature 2011; 475: 101–105.
Xu Z, Zan H, Pone EJ, Mai T, Casali P . Immunoglobulin class-switch DNA recombination: induction, targeting and beyond. Nat Rev Immunol 2012; 12: 517–531.
San Miguel JF, Vidriales MB, Ocio E, Mateo G, Sanchez-Guijo F, Sanchez ML et al. Immunophenotypic analysis of Waldenstrom's macroglobulinemia. Semin Oncol 2003; 30: 187–195.
Ocio EM, Hernandez JM, Mateo G, Sanchez ML, Gonzalez B, Vidriales B et al. Immunophenotypic and cytogenetic comparison of Waldenstrom's macroglobulinemia with splenic marginal zone lymphoma. Clin Lymphoma 2005; 5: 241–245.
Sahota SS, Babbage G, Weston-Bell NJ . CD27 in defining memory B-cell origins in Waldenstrom's macroglobulinemia. Clin Lymphoma Myeloma 2009; 9: 33–35.
Kriangkum J, Taylor BJ, Treon SP, Mant MJ, Belch AR, Pilarski LM . Clonotypic IgM V/D/J sequence analysis in Waldenstrom macroglobulinemia suggests an unusual B-cell origin and an expansion of polyclonal B cells in peripheral blood. Blood 2004; 104: 2134–2142.
Walsh SH, Laurell A, Sundstrom G, Roos G, Sundstrom C, Rosenquist R . Lymphoplasmacytic lymphoma/Waldenstrom's macroglobulinemia derives from an extensively hypermutated B cell that lacks ongoing somatic hypermutation. Leuk Res 2005; 29: 729–734.
Owen RG, Kyle RA, Stone MJ, Rawstron AC, Leblond V, Merlini G et al. Response assessment in Waldenstrom macroglobulinaemia: update from the VIth International Workshop. Br J Haematol 2013; 160: 171–176.
Kimby E, Treon SP, Anagnostopoulos A, Dimopoulos M, Garcia-Sanz R, Gertz MA et al. Update on recommendations for assessing response from the Third International Workshop on Waldenstrom's Macroglobulinemia. Clin Lymphoma Myeloma 2006; 6: 380–383.
van der Velden V, Cazzaniga G, Schrauder A, Hancock J, Bader P, Panzer-Grumayer ER et al. Analysis of minimal residual disease by Ig/TCR gene rearrangements: guidelines for interpretation of real-time quantitative PCR data. Leukemia 2007; 21: 604–611.
Acknowledgements
We thank Alicia Antón and Rebeca Maldonado for their technical assistance and Phill Mason for checking the English usage and grammar of the manuscript. This work has been partially supported by the grant number PS09/01450 from the Spanish ‘Fondo de Investigaciones Sanitarias’ and the grant reference HUS412A12-1 from the ‘Consejería de Educación de la Junta de Castilla y León’.
Authors contributions
Cristina Jiménez and Ramón García-Sanz were the initial designers of the study. Cristina Jiménez, Elena Sebastián, María del Carmen Chillón and Ana Balanzategui carried out all molecular studies and prepared the database for the final analysis. Ramón García-Sanz desgined the database, and Miguel Alcoceba helped to handle it and to develop the initial analysis. Ramón García-Sanz helped by Luis A Marín developed the statistical analysis. Pilar Giraldo, José Mariano Hernández, Fernando Escalante, Tomás J González-López, Carmen Aguilera, Alfonso García de Coca, Ilda Murillo and Marcos González were clinicians responsible for the patients and those who took care of the protocols’ accomplishment, sampling and collection of clinical data. Rocío Corral and María Eugenia Sarasquete helped in the molecular analysis and data collection. Bruno Paiva, Enrique M Ocio and Norma C Gutiérrez were responsible for the immunophenotyping and ctyogenetic analysis of the patients included in this series. Ramón García-Sanz and Cristina Jiménez prepared the initial version of the paper. Jesús F San Miguel and Marcos González were the main responsible of the global and molecular groups, respectively, and they were the persons responsible of the final revision of the draft, as well as the ones who gave the final approval of the version to be published. Ramón García-Sanz reviewed the conception and design of most of the work, made the database and supervised the statistical analysis. He rewrote the paper and made the final upload of the paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Leukemia website
Rights and permissions
About this article
Cite this article
Jiménez, C., Sebastián, E., Chillón, M. et al. MYD88 L265P is a marker highly characteristic of, but not restricted to, Waldenström’s macroglobulinemia. Leukemia 27, 1722–1728 (2013). https://doi.org/10.1038/leu.2013.62
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2013.62
- Springer Nature Limited
Keywords
This article is cited by
-
Clinical, biological, electrophysiological and therapeutic profile of patients with anti-MAG neuropathy according to MYD88L265P and CXCR4 mutations and underlying haemopathy
Journal of Neurology (2024)
-
Single-cell transcriptome analysis reveals stem cell-like subsets in the progression of Waldenström’s macroglobulinemia
Experimental Hematology & Oncology (2023)
-
A comparison of the International Consensus and 5th World Health Organization classifications of mature B-cell lymphomas
Leukemia (2023)
-
Diagnostics in Waldenström’s macroglobulinemia: a consensus statement of the European Consortium for Waldenström’s Macroglobulinemia
Leukemia (2023)
-
CXCR4 in Waldenström’s Macroglobulinema: chances and challenges
Leukemia (2021)