Abstract
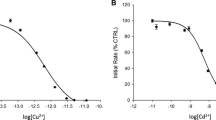
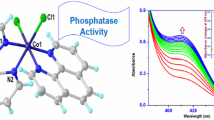
MANY enzymes require the presence of metal ions for activity1. Kinetic studies on such systems indicate the existence of an equilibrium between the enzyme protein, the activating metal ion and a complex between metal ion and enzyme. By assuming the complex to be the only species catalytically active, a hypothetical dissociation constant can be determined from activity measurements. Considerations of this kind have led Smith2 to formulate a theory of peptidase action according to which the metal ion acts as a binding group bringing enzyme and substrate together. Already in 1942, the same type of mechanism was suggested by Warburg and Christian3 for the activation of enolase by magnesium, manganous and zinc ions. Since, however, no direct measurements of the interaction of activating metal ions with well-characterized enzyme preparations have been published, the present study was undertaken.
Similar content being viewed by others
References
Lehninger, A. L., Physiol. Rev., 30, 393 (1950).
Smith, E. L., “Adv. Enzymol.”, 12, 191 (1951).
Warburg, O., and Christian, W., Biochem. Z., 310, 384 (1942).
Klotz, I. M., Walker, F. M., and Pivan, R. B., J. Amer. Chem. Soc., 68, 1486 (1946).
Monk, C. B., Trans. Farad. Soc., 47, 297 (1950).
Albert, A., Biochem. J., 47, 531 (1950); 50, 690 (1952).
Kroll, H., J. Amer. Chem. Soc., 74, 2034 (1952).
Cohn, E. J., Surgenor, D. M., Schmid, K., Batchelor, W. H., and Isliker, H. C., Farad. Soc. Discuss. (August 1952).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
MALMSTRÖM, B. Interaction of Manganous Ions with Enolase. Nature 171, 392–393 (1953). https://doi.org/10.1038/171392a0
Issue Date:
DOI: https://doi.org/10.1038/171392a0
- Springer Nature Limited