Abstract
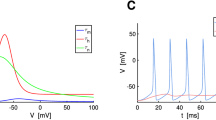
In our study, we represent the theoretical and numerical analysis of a stochastic version of the Hodgkin–Huxley model applied to a two-dimensional spatial cylindrical area simulating the neuronal somatic membrane. We characterized the spatiotemporal dynamics of the membrane potential by its local value V m (x, y, t) and the integral of this value with respect to time F(x, y, T) within an interval [0, T]. Analysis of the model showed that (i) there are nonzero gradients of F(x, y, T) at any distribution of ion channels; (ii) the maximum gradient F(x, y, T) decreases down to zero with the time T, if the channels are distributed homogeneously, and acquire some positive constant value, if the channels are distributed inhomogeneously; the gradient F(x, y, T) depends on the gradient of spatial distribution of the channels; and (iii) under conditions of spatial redistribution of the channels with preservation of their number, the dynamics of V m (x, y, t) does not change.
Similar content being viewed by others
REFERENCES
W. R. Holmes and W. B. Levy, “Quantifying the role of inhibition in associative long-term potentiation in granule cells with computational model,” J. Neurophysiol., 78, No. 3, 103–116 (1997).
J. C. Callaway and W. N. Ross, “Spatial distribution of synaptically activated sodium concentration changes in cerebellar Purkinje neurons,” J. Neurophysiol., 77, No. 1, 145–152 (1997).
P. Southan and B. Robertson, “Patch-clamp recording from cerebellar basket cell bodies and their presynaptic terminals reveals an asymmetric distribution of voltage-gated potassium channels, J. Neurosci., 18, No. 3, 948–955 (1998).
G. J. Stuart and B. Sakmann, “Active propagation of somatic action potentials into neocortical pyramidal cell dendrites,” Nature, 367, 69–72 (1994).
G. J. Stuart and B. Sakmann, “Amplification of EPSPs by axosomatic sodium channels in neocortical pyramidal neurons,” Neuron, 15, 1065–1076 (1995).
G. M. Shepherd, Neurobiology, Vol. 1, Oxford Univ. Press (1983).
G. P. Zhukova, Neuronal Structure and Neuronal Connections between the Encephalon and Spinal Cord [in Russian], Meditsina, Moscow (1977).
P. Gogan, I. Schmiedel-Jakob, Y. Chitti, and S. Tyč-Dumont, “Fluorescence imaging of local membrane electrical fields during the excitation of single neurons in culture,” Biophys. J., 69, 299–310 (1995).
D. Zecevic, “Multiple spike-initiation zones in single neurons revealed by voltage-sensitive dyes,” Nature, 381, 322–325 (1996).
P. Fromgerz, K. H. Dambacher, H. Ephardt, et al., “Fluorescent dyes as probes of voltage transients in neuron membranes. Progress report,” Ber. Bunsenges. Phys. Chem., 95, 1333–1345 (1991).
R. F. Fox, “Stochastic versions of the Hodgkin-Huxley equations,” Biophys. J., 72, 2068–2074 (1997).
B. Hille, Ionic Channels of Excitable Membranes, Sinauer Ass., Sunderland, Mass. (1992).
G. Major, A. U. Larkman, P. Jonas, et al., “Detailed passive cable models of whole-cell recorded CA3 pyramidal neurons in rat hippocampal slices,” J. Neurosci., 14, No. 8, 4613–4638 (1994).
M. Correa and F. Bezanilla, “Gating of the squid sodium channel at positive potentials. II. Single channels reveal two open states,” Biophys. J., 66, No. 6, 1864–1878 (1994).
J. R. Clay and L. J. DeFelice, “Relationship between membrane excitability and single channel open-close kinetics,” Biophys. J., 42, 151–157 (1983).
L. J. DeFelice and A. Isaac, “Chaotic states in random world: Relationship between the nonlinear differential equations of excitability and the stochastic properties of ion channels,” J. Stat. Phys., 70, 339–354 (1993).
C. C. Chow and A. W. John, “Spontaneous action potential due to channel fluctuation,” Biophys. J., 71, 3013–3021 (1996).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Savchenko, L.P. Spatial Distribution of the Potential on a Cylindrical Surface Simulating the Somatic Membrane: Model Studies. Neurophysiology 32, 291–299 (2000). https://doi.org/10.1023/A:1010333820815
Issue Date:
DOI: https://doi.org/10.1023/A:1010333820815