Abstract
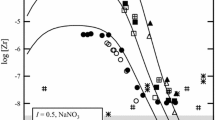
The enthalpies of dissolution of ZrCl4, ZrBr4, HfCl4 and HfBr4 in water in weakly acidic and alkaline solutions were measured at 25°C in a calorimeter provided with an isothermal cover. The standard enthalpies of formation of Zr(OH)4 and Hf(OH)4 in solution were measured. The thermodynamic characteristics of the reactions which resulted in the formation of tetrahydroxy complexes of Zr and Hf in aqueous solution were also determined.
Similar content being viewed by others
References
Spravochnik 'Termicheskie konstanty veschestv', Pod red. Glouchko V. P. M.: Nauka, 1965–1974.
M. Beck, Z. Anorg. Allgem. Chem., 174 (1928) 31.
A. G. Turnbull, J. Phys. Chem., 65 (1961) 1652.
R. A. Cooley and H. O. Banks, J. Amer. Chem. Soc., 73 (1951) 4022.
A. S. Solovkin and E. N. Tsvetkov, Uspehi Khimii, 31 (1962) 1394.
V. A. Nazarenko, V. P. Antonovich and E. M. Nevskaya, Gidroliz ionov metallov v rastvorah. M.: Atomizdat, 1973.
V. A. Nazarenko and O. V. Mangaladze, J. Neorg. Khimii, 14 (1969) 1219.
V. A. Nazarenko, V. P. Antonovich and E. M. Nevskaya, J. Neorg. Khimii, 16 (1971) 2387.
V. P. Vasil'ev and A. I. Lytkin, J. Neorg. Khimii, 21 (1976) 3037.
V. P. Vasil'ev, V. A. Borodin and E. V. Kozlovskiy, Primenenie IBC v himico-analiticheskih raschetah. M.: Vyschaya shkola, 1993, p. 81.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Vasil'ev, V.P., Lytkin, A.I. & Chernyavskaya, N.V. Thermodynamic Characteristics of Zirconium and Hafnium Hydroxides in Aqueous Solution. Journal of Thermal Analysis and Calorimetry 55, 1003–1009 (1999). https://doi.org/10.1023/A:1010114723995
Issue Date:
DOI: https://doi.org/10.1023/A:1010114723995