Abstract
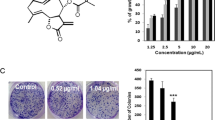
Cleistanthin B is a potential anticancer agent isolated from the tropical plant Cleistanthus collinus. We have previously shown that cleistanthin B is clastogenic and induces micronuclei formation and chromosomal aberrations. We now show that this compound inhibits DNA synthesis in Chinese hamster ovary (CHO) cells and induces apoptosis in cervical carcinoma (SiHa) cells. Flow cytometric analysis of cleistanthin treated CHO cells revealed that they were blocked in G1. Cervical carcinoma (SiHa) cells exposed to cleistanthin B shrank, rounded up and had condensed chromatin and fragmented nuclei. DNA isolated from cleistanthin treated cells exhibited the characteristic apoptotic ladder when electrophoresed in agarose gels. These results were confirmed by flow cytometry. Etoposide, a structurally similar compound also induced apoptosis in these cells although with a difference. Etoposide induced apoptosis after permitting cells to enter into S phase, while cleistanthin B stopped entry of cells into S phase and subsequently drove them to apoptosis.
Similar content being viewed by others
References
Pradheep Kumar CP, Panneerselvam N, Rajesh S, Shanmugam G. Cytotoxic and genotoxic effects of cleistanthin B in normal and tumour cells. Mutagenesis 1996; 11: 543–547.
Bhakuni DS, Dhar ML, Dhar MM, Dhawan BN, Mehrota BN. Screening of Indian plants for biological activity part II. Indian Exp Bio 1969; 7: 20.
Pradheepkumar CP. Regulation of cell proliferation: effects of cleistanthins on the proliferation of normal and tumour cells. Ph D Thesis. Madurai Kamaraj University: 1998.
Kerr JFR, Wyllie AH, Currie AR. Apoptosis: A basic biological phenomenon with wide-ranging implication in disease kinetics. Br J Cancer 1972; 26: 239–257.
Sen S, D'Incalci M. Apoptosis: Biological events and relevance to cancer chemotherapy. FEBS Lett 1992; 307: 122–127.
Fawthrop DJ, Boobis AR, Davies DS. Mechanisms of cell death. Arch Toxicol 1991; 65: 437–444.
Barry MA, Behnke CA, Eastman A. Activation of programmed cell death (Apoptosis) by cisplatin, other anticancer drugs, toxins and hyperthermia. Biochem Pharmacol 1990; 40: 2353– 2362.
Ormerod MG, Orr RM, Peacock JH. The role of apoptosis in cell killing by cisplatin: a flow cytometric study. Br J Cancer 1994; 69: 93–100.
Kastan MB, Zhan Q, El-Diery WS, et al. A mammalian cell cycle checkpoint pathway utilizing p53 and GADD45 is defective in ataxia-telangiectasia. Cell 1992; 71: 587–597.
Waldman T, Lengauer C, Kinzler KW, Vogelstein B. Uncoupling of S phase and mitosis induced by anticancer agents in cells lacking P21. Nature 1996; 381: 713–716.
Nasmyth K, Hunt T. Dams and sluice. Nature 1994; 366: 634–635.
Meikrantz W, Gisselbrecht S, Tam SW, Schlegel R. Activation of cyclin-A dependent protein kinases during apoptosis. Proc Natl Acad Sci USA 1994; 91: 3754–3758.
Kondo S. Apoptosis by antitumor agents and other factors in relation to cell cycle checkpoints. J Radiat Res 1995; 36: 56– 62.
Afanas'ev VN, Korol' BA, Mantsygin YA, Nelipovich PA, Pechtnikov VA, Umansky SR. Flow cytometry and biochemical analysis of DNA degradation characteristic of two types of cell death. FEBS Lett 1986; 194: 347–350.
Frankfurt OS, Byrnes JJ, Seckinge D, Sugarbaker EV. Apoptosis (programmed cell death) and the evaluation of chemosensitivity in chronic lymphocytic leukaemia and lymphoma. Oncology Res 1993; 5: 37–42.
Wrasidlo W, Johnson DS, Boger DL. Induction of endonucleolytic DNA fragmentation and apoptosis by the Duocarmycins. Bioorg Medicn Chem Letters 1994; 4: 631–636.
Nicolaou KC, Dai WM, Tsay SC, Estevez VA, Wrasidlo W. Designed enediynes: a new class of DNA-cleaving molecules with potent and selective anticancer activity. Science 1992; 256: 1172–1178.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pradheep Kumar, C.P., Pande, G. & Shanmugam, G. Cleistanthin B causes G1 arrest and induces apoptosis in mammalian cells. Apoptosis 3, 413–419 (1998). https://doi.org/10.1023/A:1009658518998
Issue Date:
DOI: https://doi.org/10.1023/A:1009658518998