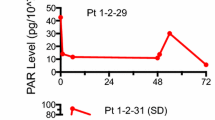
Abstract
Purpose: The objectives of this trial wereto assess the maximal tolerated dose andtoxicity of the combination of oraleniluracil and 5-fluorouracil andintravenous gemcitabine.
Patients and methods: Patients withhistologically confirmed, incurablemalignancy (solid tumor or lymphoma)refractory to standard therapy or for whichno standard therapy exists were enrolled. The treatment plan consisted of weeklygemcitabine for three weeks with twicedaily dosing of 5-FU and eniluracil for 21days beginning on day one of gemcitabine. Cycles repeated on an every four weekschedule. The initial cohort receivedgemcitabine 800 mg/m2, oral 5-FU 0.6 mg/m2and eniluracil 6.0 mg/m2.
Results: Twenty-six patients were enrolled. Eight patients received less than 2 cyclesof therapy. Hematologic andgastrointestinal toxicity predominated,with 48% of courses resulted in grade oneor two neutropenia. Hematologic toxicitywas dose limiting. One treatment relateddeath occurred.
Conclusions: The combination of eniluracil,5-fluorouracil and gemcitabine offers anoral alternative for 5-FU administration.The recommended phase II dose isgemcitabine 1000 mg/m2, 5FU 1.2 mg/m2 andeniluracil 12 mg/m2.
Similar content being viewed by others
References
Meta-Analysis Group in Cancer: Efficacy of intravenous continuous infusion of fluorouracil compared with bolus administration in advanced colorectal cancer. J Clin Oncol 16:301–308, 1998
Heggie GD, Sommadossi J-P, Cross DS, Huster WJ, Diasio RB: Clinical pharmacokinetics of 5-fluorouracil and its metabolites in plasma, urine, and bile. Cancer Res 47:2203–2206, 1987
Cohen JL, Irwin LE, Marshall GJ, Darvey H, Bateman JR: Clinical pharmacology of oral and intravenous 5-flourouracil (NSC-19893). Cancer Chemother Rep 58:723–731, 1974
Naguib FN, el Kouni MH, Cha S: Enzymes of uracil catabolism in normal and neoplastic human tissues. Cancer Res 45:5405–5412, 1985
De Bono JS, Twelves CJ: The oral fluorinated pyrimidines. Invest New Drugs 19:41–59, 2001
Fleming RA, Milano G, Gaspard MH, Bargnoux PJ, Thyss A, Plagne R, Renee N, Schneider M, Demard F: Dihydropyrimidine dehydrogenas activity in cancer patients. Eur J Cancer 29A:740–744, 1993
Lu Z, Shang R, Diasio RB: Dihydropyrimidine dehydrogenas activity in human peripheral blood mononuclear cells and liver: Population characteristics, newly identified deficient patients and clinical implications in 5-fluorouracil chemotherapy. Cancer Res 53:5433–5438, 1993
Tuchman M, Stoeckeler JS, Kiang DT, O'Dea RF, Ramnaraine ML, Mirkin BL: Familial pyrimidiemia and pyrimidinuria associated with severe fluorouracil toxicity. N Engl J Med 313:245–249, 1985
Diasio RB, Beavers TL, Carpenter JT: Familial deficiency of dihydropyrimidine dehydrogenase: Biochemical basis for familial pyridinemia and sever 5-fluorouracil-induced toxicity. J Clin Invest 81:47–51, 1988
Harris BE, Carpenter JT, Diasio RB: Severe 5-fluorouracil toxicity secondary to dihydropyrimidine dehydrogenase deficiency: A potentially more common pharmacogenetic syndrome. Cancer 68:499–501, 1991
Houyau P: Severe fluorouracil toxicity in a patient with dihydropyrimidine dehyrodenase deficiency. J Natl Cancer Inst 8:1602–1603, 1993
Porter DJT, Chestnut WG, Merrill BM, Spector T: Mechanism-based inactivation of dihdropyrimidine dehydrogenase by 5-ethynyluacil. J Biol Chem 267:5236–5242, 1992
Baker SD, Khor SP, Adjei AA, Doucette M, Spector T, Donehower RC, Grochow LB, Sartorius SE, Noe DA, Hohneker JA, Rowinsky EK: Pharmacokinetic, oral bioavailability, and safety study of fluorouracil in patients treated with 776C85, an inactivator of dihydropyrimidine dehydrogenase. J Clin Oncol 14:3085–3096, 1996
Schilsky RL, Hohneker J, Ratain MJ, Janisch L, Smetzer L, Lucas VS, Khor SP, Diasio R, Von Hoff DD, Burris HA 3rd: Phase I clinical and pharmacologic study of eniluracil plus fluorouracil in patients with advanced cancer. J Clin Oncol 16:1450–1457, 1998
Baker SD, Diasio RB, O'Reilly S, Lucus VS, Khor SP, Sartorius SE, Donehower RC, Grochow LB, Spector T, Hohneker JA, Rowinsky EK: Phase I and pharmacologic study of oral flourouracil on a chronic daily schedule in combination with the dihydropyrimidine dehydrogenase inactivator eniluracil. J Clin Oncol 18:915–926, 2000
Grem JL, Harold N, Shapiro J, Bi DQ, Quinn MG, Zentko S, Keith B, Hamilton JM, Monohan BP, Donovan S, Grollman F, Morrison G, Takimoto CH: Phase I and pharmacokinetic trial of weekly oral fluorouracil given with eniluracil and low-dose leucovorin to patients with solid tumors. J Clin Oncol 18:3952–3963, 2000
Smith IE, Johnston MR, O'Brien ME, Hickish TF, de Boer RH, Norton A, Cirkel DT, Barton CM: Low-dose oral fluorouracil with eniluracil as first-line chemotherapy against advanced breast cancer: A phase II study. J Clin Oncol 18:2378–2384, 2000
Schilsky R, Bukowski R, Burris H 3rd, Hochster H, O'Rourke M, Wall JG, Mani S, Bonny T, Levin J, Hohneker J: A multicenter phase II study of a five-day regimen of oral 5-fluorouracil plus eniluracil with or without leucovorin in patients with metastatic colorectal cancer. Annal Oncol 11:415–420, 2000
Meropol NJ, Niedzwiecki D, Hollis D, Shilsky RL, Mayer RJ: Phase II study of oral eniluracil, 5-fluororacil and leucovorin in patients with advanced colorectal carcinoma. Cancer 91:1256–1263, 2001
Dorr RT, Van Hoff DD: Cancer Chemotherapy Handbook, Second Edition. Appleton and Lange, pp 500–514, pp 524-526, 1994
Schulz L, Schalhorm A, Wilmanns W, Heinemann V: Synergistic interaction of gemcitabine and 5-fluorouracil in colon cancer cells. Proc Amer Soc Clin Oncol 17:965, 1998
Berlin JD, Alberti DB, Arzoomanian RZ, Freierabend CA, Simon KJ, Binger KA, Marnocha RM, Wilding G: A phase I trial of gemcitabine, 5-fluororacil and leucovorin in patients with advanced, recurrent, and/or metastatic solid tumors. Invest New Drugs 16:325–330, 1999
Rinaldi DA, Lormand NA, Brierre JE, Cole JL, Stagg MP, Fontenot MF, Buller EJ, Rainey JM: A phase I trial of gemcitabine and infusional 5-fluorouacil (5-FU) in patients with refractory solid tumors: Louisiana Oncology Associates Protocol No. 1 (LOA-1) Am J Clin Oncol 23:78–82, 2000
Madajewicz S, Hentschel P, Burns P, Caruso R, Fiore J, Fried M, Malhotra H, Ostrow S, Sugarman S, Viola M: Phase I chemotherapy study of biochemical modulation of folinic acid and fluorouracil by gemcitabine in patients with solid tumor malignancies. J Clin Oncol 18:3553–3557, 2000
Mani S, Vogelzand N, Bertucci D, Stadler WM, Shilsky RL, Ratain MJ: Phase I study to evaluate multiple regimens of intravenous 5-fluorouracil administered in combination with weekly gemcitabine in patients with advanced solid tumors. Cancer 92:1567–1576, 2001
Ishikawa T, Utoh M, Sawada N, Nishida M, Fukase Y, Sekiguchi F, Ishitsuka H: Tumor selective delivery of 5-fluorouracil by capecitabine, a new oral fluoropyrimidine carbamate, in human cancer xenografts. Biochem Pharmacol 55(7):1091–1097, 1998
Mackean M, Phanting A, Twevles C, Schellens J, Allman D, Osterwalder B, Reigner B, Griffin T, Kaye S, Verweij J: Phase I and pharmacologic study of intermittent twice-daily oral therapy with capecitabine in patients with advanced and/or metastatic cancer. J Clin Oncol 16(9):2977–2985, 1998
Sulkes A, Benner SE, Canetta RM: Uracil-ftorafur: an oral fluoroupyrimidine active in colorectal cancer. J Clin Oncol 16(10):3461–3475, 1998
Takiuchi J, Ajani JA: Uracil-tegafur in gastric carcinoma: a comprehensive review. J Clin Oncol 16(8):2877–2885, 1998
Shirasaka T, Nakano K, Tadechi T, Satake H, Uchida J, Fujioka A, Siato H, Okabe H, Oyama K, Takeda S, Unemi N, Fukushima A: Antitumor activity of 1M tegafur-0.4 M 5-chloro-2,4-dihydroxypyridine-1 M potassium oxonate (S1) against human colon carcinoma orthotopically implanted into nude rats. Cancer Res 56(11):2602–2606, 1996
Shirasaka T, Shimarnoto Y, Fukushima M: Inhibition by oxonic acid of gastrointestinal toxicity of 5-fluorouracil without loss of its antitumor activity in rats. Cancer Res 53(17):4004–4009, 1993
Sakata Y, Ohtsu A, Horikoshi N, Sugimachi K, Mitachi Y, Taguchi T: Late phase II study of a novel oral fluoroupyrimidine anti-cancer drug 3-1 (1 M tegafur-0.4 M 5-chloro-2,4-dihydroxypyridine-1 M potassium oxonate) in advanced gastric cancer patients. Eur J Cancer 34(11):1715–1720, 1998
Baba J, Ohtsu A, Sakata Y, Mitachi Y, Sugimachi K, Taguchi T: Late phase II study of S-1 in patients with advanced colorectal cancer in Japan. Proc Am Soc Clin Oncol 17:1065a, 1998
Hidalgo M, Castellano D, Paz-Ares L, Gravalos C, Diaz-Puente M, Hitt R, Alonso S, Cortes-Funes H: Phase I-II study of gemcitabine and fluorouracil as a continuous infusion in patients with pancreatic cancer. J Clin Oncol 17:585–592, 1999
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Morgan-Meadows, S., Thomas, J.P., Mulkerin, D. et al. Phase I Study of Eniluracil, Oral 5-Fluororacil and Gemcitabine in Patients with Advanced Malignancy. Invest New Drugs 20, 377–382 (2002). https://doi.org/10.1023/A:1020673928704
Issue Date:
DOI: https://doi.org/10.1023/A:1020673928704