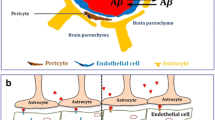
Summary
The circuitry of the human brain is formed by neuronal networks embedded into astroglial syncytia. The astrocytes perform numerous functions, providing for the overall brain homeostasis, assisting in neurogenesis, determining the micro-architecture of the grey matter, and defending the brain through evolutionary conserved astrogliosis programs.
Astroglial cells are engaged in neurological diseases by determining the progression and outcome of neuropathological process. Astrocytes are specifically involved in various neurodegenerative diseases, including Alzheimer’s disease, amyotrophic lateral sclerosis, Parkinson’s disease, and various forms of dementia. Recent evidence suggest that early stages of neurodegenerative processes are associated with atrophy of astroglia, which causes disruptions in synaptic connectivity, disbalance in neurotransmitter homeostasis, and neuronal death through increased excitotoxicity. At the later stages, astrocytes become activated and contribute to the neuroinflammatory component of neurodegeneration.
Article PDF
Similar content being viewed by others
References
Case RM, Eisner D, Gurney A, Jones O, Muallem S, Verkhratsky A. Evolution of calcium homeostasis: from birth of the first cell to an omnipresent signalling system. Cell Calcium 2007;42: 345–350.
Durell SR, Guy HR. A putative prokaryote voltage-gated Ca2+ channel with only one 6TM motif per subunit. Biochem Biophys Res Commun 2001;281: 741–746.
Matsushita T, Hirata H, Kusaka I. Calcium channels in bacteria. Purification and characterization. Ann N Y Acad Sci 1989;560: 426–429.
Shemarova IV, Nesterov VP. Evolution of mechanisms of calcium signaling: the role of calcium ions in signal transduction in prokaryotes. Zh Evol Biokhim Fiziol 2005;41: 12–17.
Tisa LS, Sekelsky JJ, Adler J. Effects of organic antagonists of Ca2+, Na+, and K+ on chemotaxis and motility of Escherichia coli J Bacteriol 2000;182: 4856–4861.
Eckert R, Brehm P. Ionic mechanisms of excitation in Paramecium. Annu Rev Biophys Bioeng 1979;8: 353–383.
Franciolini F, Petris A. Evolution of ionic channels of biological membranes. Mol Biol Evol 1989;6: 503–513.
Koishi R, Xu H, Ren D, et al. A superfamily of voltage-gated sodium channels in bacteria. J Biol Chem 2004;279: 9532–9538.
Shemarova IV, Nesterov VP. Evolution of Ca2+ signaling mechanisms. Role of calcium ions in signal transduction in lower eukaryotes. Zh Evol Biokhim Fiziol 2005;41: 303–313.
Ladenburger EM, Sehring IM, Kom I, Plattner H. Novel types of Ca2+ release channels participate in the secretory cycle of Paramecium cells. Mol Cell Biol 2009;29: 3605–3622.
Matt H, Plattner H, Reichel K, Lefort-Tran M, Beisson J. Genetic dissection of the final exocytosis steps in Paramecium tetraurelia cells: trigger analyses. J Cell Sci 1980;46: 41–60.
Plattner H, Reichel K, Matt H, Beisson J, Lefort-Tran M, Pouphile M. Genetic dissection of the final exocytosis steps in Paramecium tetraurelia cells: cytochemical determination of Ca2+-ATPase activity over performed exocytosis sites. J Cell Sci 1980;46: 17–40.
Burnstock G, Verkhratsky A. Evolutionary origins of the purinergic signalling system. Acta Physiol (Oxf) 2009;195: 415–447.
Sakaguchi M, Mizusina A, Kobayakawa Y. Structure, development, and maintenance of the nerve net of the body column in Hydra. J Comp Neurol 1996;373: 41–54.
Bacaj T, Tevlin M, Lu Y, Shaham S. Glia are essential for sensory organ function in C. elegans. Science 2008;322: 744–747.
Reichenbach A, Pannicke T. Neuroscience. A new glance at glia. Science 2008;322: 693–694.
Verkhratsky A, Butt A. Glial neurobiology. A textbook. Chichester: John Wiley & Sons, 2007.
Oberheim NA, Takano T, Han X, et al. Uniquely hominid features of adult human astrocytes. J Neurosci 2009;29: 3276–3287.
Oberheim NA, Wang X, Goldman S, Nedergaard M. Astrocytic complexity distinguishes the human brain. Trends Neurosci 2006;29: 547–553.
Bushong EA, Martone ME, Ellisman MH. Maturation of astrocyte morphology and the establishment of astrocyte domains during postnatal hippocampal development. Int J Dev Neurosci 2004;22: 73–86.
Bushong EA, Martone ME, Jones YZ, Ellisman MH. Protoplasmic astrocytes in CA1 stratum radiatum occupy separate anatomical domains. J Neurosci 2002;22: 183–192.
Nedergaard M, Ransom B, Goldman SA. New roles for astrocytes: redefining the functional architecture of the brain. Trends Neurosci 2003;26: 523–530.
Iadecola C, Nedergaard M. Glial regulation of the cerebral microvasculature. Nat Neurosci 2007;10: 1369–1376.
Mulligan SJ, MacVicar BA. Calcium transients in astrocyte end-feet cause cerebrovascular constrictions. Nature 2004;431: 195–199.
Takano T, Tian GF, Peng W, et al. Astrocyte-mediated control of cerebral blood flow. Nat Neurosci 2006;9: 260–267.
Zonta M, Angulo MC, Gobbo S, et al. Neuron-to-astrocyte signaling is central to the dynamic control of brain microcirculation. Nat Neurosci 2003;6: 43–50.
Magistretti PJ. Neuron-glia metabolic coupling and plasticity. J Exp Biol 2006;209: 2304–2311.
Magistretti PJ. Role of glutamate in neuron-glia metabolic coupling. Am J Clin Nute 2009;90: 875S-880S.
Kofuji P, Newman EA. Potassium buffering in the central nervous system. Neuroscience 2004;129: 1045–1056.
Newman EA, Frambach DA, Odette LL. Control of extracellular potassium levels by retinal glial cell K+ siphoning. Science 1984;225: 1174–1175.
Simard M, Nedergaard M. The neurobiology of glia in the context of water and ion homeostasis. Neuroscience 2004;129: 877–896.
Swanson RA. Astrocyte neurotransmitter uptake. In: Neuroglia. Kettenmann H, Ransom B, eds. Oxford: Oxford University Press, 2005: 346–354.
Verkhratsky A, Kirchhoff F. Glutamate-mediated neuronal-glial transmission. J Anat 2007;210: 651–660.
Danbolt NC. Glutamate uptake. Progr Neurobiol 2001;65: 1–105.
Zerangue N, Kavanaugh MP. Flux coupling in a neuronal glutamate transporter. Nature 1996;383: 634–637.
Kirischuk S, Kettenmann H, Verkhratsky A. Membrane currents and cytoplasmic sodium transients generated by glutamate transport in Bergmann glial cells. Pflugers Arch 2007;454: 245–252.
Kirischuk S, Kettenmann H, Verkhratsky A. Na+/Ca2+ exchanger modulates kainate-triggered Ca2+ signaling in Bergmann glial cells in situ. Faseb J 1997;11: 566–572.
Minelli A, Castaldo P, Gobbi P, Salucci S, Magi S, Amoroso S. Cellular and subcellular localization of Na+-Ca2+ exchanger protein isoforms, NCX1, NCX2, and NCX3 in cerebral cortex and hippocampus of adult rat. Cell Calcium 2007;41: 221–234.
Martinez-Hernandez A, Bell KP, Norenberg MD. Glutamine synthetase: glial localization in brain. Science 1977;195: 1356–1358.
Bowman CL, Kimelberg HK. Excitatory amino acids directly depolarize rat brain astrocytes in primary culture. Nature 1984;311: 656–659.
Kettenmann H, Backus KH, Schachner M. Aspartate, glutamate and gamma-aminobutyric acid depolarize cultured astrocytes. Neurosci Lett 1984;52: 25–29.
Kettenmann H, Gilbert P, Schachner M. Depolarization of cultured oligodendrocytes by glutamate and GABA. Neurosci Lett 1984;47: 271–276.
Bevan S, Chiu SY, Gray PT, Ritchie JM. The presence of voltage-gated sodium, potassium and chloride channels in rat cultured astrocytes. Proc R Soc Lond B Biol Sci 1985;225: 299–313.
Blankenfeld GV, Verkhratsky AN, Kettenmann H. Ca2+ channel expression in the oligodendrocyte lineage. Eur J Neurosci 1992;4: 1035–1048.
Dave V, Gordon GW, McCarthy KD. Cerebral type 2 astroglia are heterogeneous with respect to their ability to respond to neuroligands linked to calcium mobilization. Glia 1991;4: 440–447.
Enkvist MO, Holopainen I, Akerman KE. Glutamate receptor-linked changes in membrane potential and intracellular Ca2+ in primary rat astrocytes. Glia 1989;2: 397–402.
Glaum SR, Holzwarth JA, Miller RJ. Glutamate receptors activate Ca2+ mobilization and Ca2+ influx into astrocytes. Proc Natl Acad Sci U S A 1990;87: 3454–3458.
McCarthy KD, Salm AK. Pharmacologically-distinct subsets of astroglia can be identified by their calcium response to neuroligands. Neuroscience 1991;41: 325–333.
Pearce B, Murphy S, Jeremy J, Morrow C, Dandona P. ATP-evoked Ca2+ mobilisation and prostanoid release from astrocytes: P2-purinergic receptors linked to phosphoinositide hydrolysis. J Neurochem 1989;52: 971–977.
Sontheimer H, Ransom BR, Cornell Bell AH, Black JA, Waxman SG. Na+-current expression in rat hippocampal astrocytes in vitro: alterations during development. J Neurophysiol 1991;65: 3–19.
Verkhratsky AN, Trotter J, Kettenmann H. Cultured glial precursor cells from mouse cortex express two types of calcium currents. Neurosci Lett 1990;112: 194–198.
Verkhratsky A, Shmigol A. Calcium-induced calcium release in neurones. Cell Calcium 1996;19: 1–14.
Kirischuk S, Scherer J, Moller T, Verkhratsky A, Kettenmann H. Subcellular heterogeneity of voltage-gated Ca2+ channels in cells of the oligodendrocyte lineage. Glia 1995;13: 1–12.
Verkhratsky A, Steinhauser C. Ion channels in glial cells. Brain Res Brain Res Rev 2000;32: 380–412.
Rodriguez JJ, Mackie K, Pickel VM. Ultrastructural localization of the CB1 cannabinoid receptor in mu-opioid receptor patches of the rat Caudate putamen nucleus. J Neurosci 2001;21: 823–833.
Kirischuk S, Moller T, Voitenko N, Kettenmann H, Verkhratsky A. ATP-induced cytoplasmic calcium mobilization in Bergmann glial cells. J Neurosci 1995;15: 7861–7871.
Kirischuk S, Tuschick S, Verkhratsky A, Kettenmann H. Calcium signalling in mouse Bergmann glial cells mediated by a1-adreno-receptors and H1 histamine receptors. Eur J Neurosci 1996;8: 1198–1208.
Kirischuk S, Matiash V, Kulik A, Voitenko N, Kostyuk P, Verkhratsky A. Activation of P2-purino-, a1-adreno and H1-histamine receptors triggers cytoplasmic calcium signalling in cerebellar Purkinje neurons. Neuroscience 1996;73: 643–647.
Verkhratsky A, Orkand RK, Kettenmann H. Glial calcium: homeostasis and signaling function. Physiol Rev 1998;78: 99–141.
Kirischuk S, Kirchhoff F, Matyash V, Kettenmann H, Verkhratsky A. Glutamate-triggered calcium signalling in mouse Bergmann glial cells in situ: role of inositol-1,4,5-trisphosphate-mediated intracellular calcium release. Neuroscience 1999;92: 1051–1059.
Verkhratsky A, Krishtal OA, Burnstock G. Purinoceptors on neuroglia. Mol Neurobiol 2009;39: 190–208.
Verkhratsky A, Kirchhoff F. NMDA Receptors in Glia. Neuroscientist 2007;13: 28–37.
Karadottir R, Cavelier P, Bergeisen LH, Attwell D. NMDA receptors are expressed in oligodendrocytes and activated in ischaemia. Nature 2005;438: 1162–1166.
Lalo U, Pankratov Y, Kirchhoff F, North RA, Verkhratsky A. NMDA receptors mediate neuron-to-glia signaling in mouse cortical astrocytes. J Neurosci 2006;26: 2673–2683.
Lipton SA. NMDA receptors, glial cells, and clinical medicine. Neuron 2006;50: 9–11.
Araque A, Parpura V, Sanzgiri RP, Haydon PG. Tripartite synapses: glia, the unacknowledged partner. Trends Neurosci 1999;22: 208–215.
Halassa MM, Fellin T, Haydon PG. The tripartite synapse: roles for gliotransmission in health and disease. Trends Mol Med 2007;13: 54–63.
Giaume C, Venance L. Intercellular calcium signaling and gap junctional communication in astrocytes. Glia 1998;24: 50–64.
Giaume C, Maravall M, Welker E, Bonvento G. The barrel cortex as a model to study dynamic neuroglial interaction. Neuroscientist 2009;15: 351–366.
Houades V, Koulakoff A, Ezan P, Seif I, Giaume C. Gap junction-mediated astrocytic networks in the mouse barrel cortex. J Neurosci 2008;28: 5207–5217.
Berridge MJ. The endoplasmic reticulum: a multifunctional signaling organelle. Cell Calcium 2002;32: 235–249.
Berridge MJ, Irvine RF. Inositol phosphates and cell signalling. Nature 1989;341: 197–205.
Verkhratsky A, Petersen OH. The endoplasmic reticulum as an integrating signalling organelle: from neuronal signalling to neuronal death. Eur J Pharmacol 2002;447: 141–154.
Verkhratsky A. Physiology and pathophysiology of the calcium store in the endoplasmic reticulum of neurons. Physiol Rev 2005;85: 201–279.
Kostyuk P, Verkhratsky A. Calcium stores in neurons and glia. Neuroscience 1994;63: 381–404.
Petersen OH, Michalak M, Verkhratsky A. Calcium signalling: Past, present and future. Cell Calcium 2005;38: 161–169.
Verkhratsky A. Calcium ions and integration in neural circuits. Acta Physiol (Oxf) 2006;187: 357–369.
Burdakov D, Petersen OH, Verkhratsky A. Intraluminal calcium as a primary regulator of endoplasmic reticulum function. Cell Calcium 2005;38: 303–310.
Verkhratsky A. The endoplasmic reticulum and neuronal calcium signalling. Cell Calcium 2002;32: 393–404.
Wuytack F, Raeymaekers L, Missiaen L. Molecular physiology of the SERCA and SPCA pumps. Cell Calcium 2002;32: 279–305.
Solovyova N, Verkhratsky A. Monitoring of free calcium in the neuronal endoplasmic reticulum: an overview of modern approaches. J Neurosci Methods 2002;122: 1–12.
Solovyova N, Veselovsky N, Toescu EC, Verkhratsky A. Ca2+ dynamics in the lumen of the endoplasmic reticulum in sensory neurons: direct visualization of Ca2+-induced Ca2+ release triggered by physiological Ca2+ entry. Embo J 2002;21: 622–630.
Petersen OH, Tepikin A, Park MK. The endoplasmic reticulum: one continuous or several separate Ca2+ stores? Trends Neurosci 2001;24: 27127–6.
Solovyova N, Verkhratsky A. Neuronal endoplasmic reticulum acts as a single functional Ca2+ store shared by ryanodine and inositol-1,4,5-trisphosphate receptors as revealed by intra-ER [Ca2+] recordings in single rat sensory neurones. Pflugers Arch 2003;446: 447–454.
Petersen OH, Verkhratsky A. Endoplasmic reticulum calcium tunnels integrate signalling in polarised cells. Cell Calcium 2007;42: 373–378.
Bezprozvanny I. The inositol 1,4,5-trisphosphate receptors. Cell Calcium 2005;38: 261–272.
Hamilton SL. Ryanodine receptors. Cell Calcium 2005;38: 253–260.
Deitmer JW, Verkhratsky AJ, Lohr C. Calcium signalling in glial cells. Cell Calcium 1998;24: 405–416.
Grosche J, Matyash V, Moller T, Verkhratsky A, Reichenbach A, Kettenmann H. Microdomains for neuron-glia interaction: parallel fiber signaling to Bergmann glial cells. Nat Neurosci 1999;2: 139–143.
Cornell Bell AH, Finkbeiner SM, Cooper MS, Smith SJ. Glutamate induces calcium waves in cultured astrocytes: long-range glial signaling. Science 1990;247: 470–473.
Dani JW, Chemjavsky A, Smith SJ. Neuronal activity triggers calcium waves in hippocampal astrocyte networks. Neuron 1992;8: 429–440.
Scemes E, Giaume C. Astrocyte calcium waves: what they are and what they do. Glia 2006;54: 716–725.
Anderson CM, Bergher JP, Swanson RA. ATP-induced ATP release from astrocytes. J Neurochem 2004;88: 246–256.
Arcuino G, Lin JH, Takano T, Liu C, Jiang L, Gao Q, et al. Intercellular calcium signaling mediated by point-source burst release of ATP. Proc Natl Acad Sci U S A 2002;99: 9840–9845.
Bennett MR, Famell L, Gibson WG. A quantitative model of purinergic junctional transmission of calcium waves in astrocyte networks. Biophys J 2005;89: 2235–2250.
Suadicani SO, Brosnan CF, Scemes E. P2X7 receptors mediate ATP release and amplification of astrocytic intercellular Ca2+ signaling. J Neurosci 2006;26: 1378–1385.
Angulo MC, Le Meur K, Kozlov AS, Charpak S, Audinat E. GABA, a forgotten gliotransmitter. Prog Neurobiol 2008;86: 297–303.
Bezzi P, Carmignoto G, Pasti L, et al. Prostaglandins stimulate calcium-dependent glutamate release in astrocytes. Nature 1998;391: 281–285.
Bezzi P, Gundersen V, Galbete JL, et al. Astrocytes contain a vesicular compartment that is competent for regulated exocytosis of glutamate. Nat Neurosci 2004;7: 613–620.
Fellin T, Pascual O, Gobbo S, Pozzan T, Haydon PG, Carmignoto G. Neuronal synchrony mediated by astrocytic glutamate through activation of extrasynaptic NMDA receptors. Neuron 2004;43: 729–743.
Jourdain P, Bergeisen LH, Bhaukaurally K, et al. Glutamate exocytosis from astrocytes controls synaptic strength. Nat Neurosci 2007;10: 331–339.
Oliet SH, Mothet JP. Regulation of N-methyl-D-aspartate receptors by astrocytic D-serine. Neuroscience 2009;158: 275–283.
Volterra A, Meldolesi J. Astrocytes, from brain glue to communication elements: the revolution continues. Nat Rev Neurosci 2005;6: 626–640.
Montana V, Malaikey EB, Verderio C, Matteoli M, Parpura V. Vesicular transmitter release from astrocytes. Glia 2006;54: 700–715.
Parpura V, Mohideen U. Molecular form follows function: (un-)snaring the SNAREs. Trends Neurosci 2008;31: 435–443.
Reyes RC, Parpura V. The trinity of Ca2+ sources for the exocytotic glutamate release from astrocytes. Neurochem Int 2009: 55: 2–8.
Cotrina ML, Lin JH, Alves-Rodrigues A, et al. Connexins regulate calcium signaling by controlling ATP release. Proc Natl Acad Sci U S A 1998;95: 15735–15740.
Duan S, Anderson CM, Keung EC, Chen Y, Swanson RA. P2X7 receptor-mediated release of excitatory amino acids from astrocytes. J Neurosci 2003;23: 1320–1328.
Kang J, Kang N, Lovatt D, et al. Connexin 43 hemichannels are permeable to ATP. J Neurosci 2008;28: 4702–4711.
Pankratov Y, Lalo U, Verkhratsky A, North RA. Vesicular release of ATP at central synapses. Pflugers Arch 2006;452: 589–597.
Perea G, Navarrete M, Araque A. Tripartite synapses: astrocytes process and control synaptic information. Trends Neurosci 2009;32: 421–431.
Alzheimer A. Beiträge zur Kenntnis der pathologischen Neuroglia und ihrer Beziehungen zu den Abbauvorgängen im Nervengewebe. In: Nissl F, Alzheimer A, eds. Histologische und Histopathologische Arbeiten über die Grosshirnrinde mit besonderer Berücksichtigung der pathologischen Anatomie der Geisteskrankheiten Jena: Verlag von Gustav Fischer 1910: 401–562.
Frommann C. Untersuchungen über die Gewebsveränderungen bei der Multiplen Sklerose des Gehirns und Rückenmarks. Jena: Verlag von Gustav Fischer, 1878.
Nissl F. Ueber einige Beziehungen zwischen Nervenzellerkrankungen und gliösen Erscheinungen bei verschiedenen Psychosen. Arch. Psychiat 1899;32: 1–21.
Giaume C, Kirchhoff F, Matute C, Reichenbach A, Verkhratsky A. Glia: the fulcrum of brain diseases. Cell Death Differ 2007;14: 1324–1335.
Nedergaard M, Rodriguez JJ, Verkhratsky A. Glial calcium and diseases of the nervous system. Cell Calcium 2010;47: 140–149
Nedergaard M, Dirnagl U. Role of glial cells in cerebral ischemia. Glia 2005;50: 281–286.
Li L, Lundkvist A, Andersson D, et al. Protective role of reactive astrocytes in brain ischemia. J Cereb Blood Flow Metab 2008;28: 468–481.
Pekny M, Nilsson M. Astrocyte activation and reactive gliosis. Glia 2005;50: 427–434.
Rossi D, Volterra A. Astrocytic dysfunction: Insights on the role in neurodegeneration. Brain Res Bull 2009;80: 224–232.
Jabs R, Seifert G, Steinhauser C. Astrocytic function and its alteration in the epileptic brain. Epilepsia 2008;49(suppl 2): 3–12.
Seifert G, Schilling K, Steinhauser C. Astrocyte dysfunction in neurological disorders: a molecular perspective. Nat Rev Neurosci 2006;7: 194–206.
Tsai G, Coyle JT. Glutamatergic mechanisms in schizophrenia. Annu Rev Pharmacol Toxicol 2002;42: 165–179.
Heneka MT, Rodriguez JJ, Verkhratsky A. Neuroglia in neurodegeneration. Brain Res Rev 2010;63: 189–211.
Kano M, Hashimoto K. Synapse elimination in the central nervous system. Curr Opin Neurobiol 2009;19: 154–161.
Scheff SW, Price DA, Schmitt FA, DeKosky ST, Mufson EJ. Synaptic alterations in CA1 in mild Alzheimer disease and mild cognitive impairment. Neurology 2007;68: 1501–1508.
Terry RD. Cell death or synaptic loss in Alzheimer disease. J Neuropathol Exp Neurol 2000;59: 1118–1119.
Charcot JM. Amyotrophic lateral sclerosis: symptomatology. In: Lectures on diseases of the nervous system London: New Sydenham Society, 1881: 192–204.
Charcot JM, Joffroy A. Deux cas d’atrophie musculaire progressive avec lesions de la substance grise et de faisceaux anterolateraux de la moelle epiniere. Arch Physiol Norm Pathol 1869;1: 354–367.
Turner BJ, Talbot K. Transgenics, toxicity and therapeutics in rodent models of mutant SOD1-mediated familial ALS. Prog Neurobiol 2008;85: 94–134.
Rossi D, Brambilla L, Valori CF, et al. Focal degeneration of astrocytes in amyotrophic lateral sclerosis. Cell Death Differ 2008;15: 1691–1700.
Yamanaka K, Chun SJ, Boillee S, et al. Astrocytes as determinants of disease progression in inherited amyotrophic lateral sclerosis. Nat Neurosci 2008;11: 251–253.
Johansson A, Engler H, Blomquist G, et al. Evidence for astrocytosis in ALS demonstrated by [11C](L)-deprenyl-D2 PET. J Neurol Sci 2007;255: 17–22.
McGeer PL, McGeer EG. Inflammatory processes in amyotrophic lateral sclerosis. Muscle Nerve 2002;26: 459–470.
Parkinson J. An Essay on the Shaking Palsy. London: Sherwood, Neely, and Jones, 1817.
McGeer PL, McGeer EG. Glial reactions in Parkinson’s disease. Mov Disord 2008;23: 474–483.
Mena MA, Garcia de Yebenes J. Glial cells as players in parkinsonism: the “good,” the “bad,” and the “mysterious” glia. Neuroscientist 2008;14: 544–560.
Broe M, Kril J, Halliday GM. Astrocytic degeneration relates to the severity of disease in frontotemporal dementia. Brain 2004;127: 2214–2220.
Kersaitis C, Halliday GM, Kril JJ. Regional and cellular pathology in frontotemporal dementia: relationship to stage of disease in cases with and without Pick bodies. Acta Neuropathol 2004;108: 515–523.
Potts R, Leech RW. Thalamic dementia: an example of primary astroglial dystrophy of Seitelberger. Clin Neuropathol 2005;24: 271–275.
Thompson KA, McArthur JC, Wesselingh SL. Correlation between neurological progression and astrocyte apoptosis in HIV-associated dementia. Ann Neurol 2001;49: 745–752.
Vanzani MC, Iacono RF, Caccuri RL, Troncoso AR, Berria MI. Regional differences in astrocyte activation in HIV-associated dementia. Medicina (B Aires) 2006;66: 108–112.
Dabir DV, Trojanowski JQ, Richter-Landsberg C, Lee VM, Forman MS. Expression of the small heat-shock protein aB-crystallin in tauopathies with glial pathology. Am J Pathol 2004;164: 155–166.
Forman MS, Lal D, Zhang B, et al. Transgenic mouse model of tau pathology in astrocytes leading to nervous system degeneration. J Neurosci 2005;25: 3539–3550.
Hazell AS. Astrocytes are a major target in thiamine deficiency and Wemicke’s encephalopathy. Neurochem Int 2009;55: 129–135.
Hazell AS, Sheedy D, Oanea R, et al. Loss of astrocytic glutamate transporters in Wemicke encephalopathy. Glia 2009;58: 148–156.
Nagele RG, D’Andrea MR, Lee H, Venkataraman V, Wang HY. Astrocytes accumulate A b 42 and give rise to astrocytic amyloid plaques in Alzheimer disease brains. Brain Res 2003;971: 197–209.
Nagele RG, Wegiel J, Venkataraman V, Imaki H, Wang KC. Contribution of glial cells to the development of amyloid plaques in Alzheimer’s disease. Neurobiol Aging 2004;25: 663–674.
Rodriguez JJ, Olabarria M, Chvatal A, Verkhratsky A. Astroglia in dementia and Alzheimer’s disease. Cell Death Differ 2009;16: 378–385.
Olabarria M, Noristani HN, Verkhratsky A, Rodriguez JJ. Concomitant astroglial atrophy and astrogliosis in a triple transgenic animal model of Alzheimer’s disease. Glia 2010;58: 831–838.
Beach TG, McGeer EG. Lamina-specific arrangement of astrocytic gliosis and senile plaques in Alzheimer’s disease visual cortex. Brain Res 1988;463: 357–361.
Griffin WS, Stanley LC, Ling C, et al. Brain interleukin 1 and S-100 immunoreactivity are elevated in Down syndrome and Alzheimer disease. Proc Natl Acad Sci U S A 1989;86: 7611–7615.
Kashon ML, Ross GW, O’Callaghan JP, et al. Associations of cortical astrogliosis with cognitive performance and dementia status. J Alzheimers Dis 2004;6: 595–604.
Mrak RE, Griffin WS. Glia and their cytokines in progression of neurodegeneration. Neurobiol Aging 2005;26: 349–354.
Sheng JG, Mrak RE, Rovnaghi CR, Kozlowska E, Van Eldik LJ, Griffin WS. Human brain S100 beta and S100 beta mRNA expression increases with age: pathogenic implications for Alzheimer’s disease. Neurobiol Aging 1996;17: 359–363.
Meda L, Baron P, Scarlato G. Glial activation in Alzheimer’s disease: the role of Abeta and its associated proteins. Neurobiol Aging 2001;22: 885–893.
Simpson JE, Ince PG, Lace G, et al. Astrocyte phenotype in relation to Alzheimer-type pathology in the ageing brain. Neurobiol Aging 2010;31: 578–590.
Wharton SB, O’Callaghan JP, Savva GM, et al. Population variation in glial fibrillary acidic protein levels in brain aging: relationship to Alzheimer-type pathology and dementia. Dement Geriatr Cogn Disord 2009;27: 465–473.
Mansour H, Chamberlain CG, Weible MW 2nd, Hughes S, Chu Y, Chan-Ling T. Aging-related changes in astrocytes in the rat retina: imbalance between cell proliferation and cell death reduces astrocyte availability. Aging Cell 2008;7: 526–540.
Ramirez JM, Ramirez AI, Salazar JJ, de Hoz R, Trivino A. Changes of astrocytes in retinal ageing and age-related macular degeneration. Exp Eye Res 2001;73: 601–615.
Mouton PR, Long JM, Lei DL, et al. Age and gender effects on microglia and astrocyte numbers in brains of mice. Brain Res 2002;956: 30–35.
Amenta F, Bronzetti E, Sabbatini M, Vega JA. Astrocyte changes in aging cerebral cortex and hippocampus: a quantitative immunohistochemical study. Microsc Res Tech 1998;43: 29–33.
Peinado MA, Quesada A, Pedrosa JA, et al. Quantitative and ultrastructural changes in glia and pericytes in the parietal cortex of the aging rat. Microsc Res Tech 1998;43: 34–42.
Pilegaard K, Ladefoged O. Total number of astrocytes in the molecular layer of the dentate gyrus of rats at different ages. Anal Quant Cytol Histol 1996;18: 279–285.
Peters A, Verderosa A, Sethares C. The neuroglial population in the primary visual cortex of the aging rhesus monkey. Glia 2008;56: 1151–1161.
Pakkenberg B, Pelvig D, Marner L, et al. Aging and the human neocortex. Exp Gerontol 2003;38: 95–99.
Hinman JD, Abraham CR. What’s behind the decline? The role of white matter in brain aging. Neurochem Res 2007;32: 2023–2031.
Cotrina ML, Nedergaard M. Astrocytes in the aging brain. J Neurosci Res 2002;67: 1–10.
DeWitt DA, Perry G, Cohen M, Doller C, Silver J. Astrocytes regulate microglial phagocytosis of senile plaque cores of Alzheimer’s disease. Exp Neurol 1998;149: 329–340.
Abramov AY, Canevari L, Duchen MR. Changes in intracellular calcium and glutathione in astrocytes as the primary mechanism of amyloid neurotoxicity. J Neurosci 2003;23: 5088–5095.
Abramov AY, Canevari L, Duchen MR. b-Amyloid peptides induce mitochondrial dysfunction and oxidative stress in astrocytes and death of neurons through activation of NADPH oxidase. J Neurosci 2004;24: 565–575.
Kuchibhotla KV, Lattarulo CR, Hyman BT, Bacskai BJ. Synchronous hyperactivity and intercellular calcium waves in astrocytes in Alzheimer mice. Science 2009;323: 1211–1215.
Peters O, Schipke CG, Philipps A, et al. Astrocyte function is modified by Alzheimer’s disease-like pathology in aged mice. J Alzheimers Dis 2009;18: 177–189.
Matos M, Augusto E, Oliveira CR, Agostinho P. Amyloid-b peptide decreases glutamate uptake in cultured astrocytes: involvement of oxidative stress and mitogen-activated protein kinase cascades. Neuroscience 2008;156: 898–810.
Oddo S, Caccamo A, Shepherd JD, et al. Triple-transgenic model of Alzheimer’s disease with plaques and tangles: intracellular Ab and synaptic dysfunction. Neuron 2003;39: 409–421.
Kimelberg HK. The problem of astrocyte identity. Neurochem Int 2004;45: 191–202.
Senitz D, Reichenbach A, Smith TG Jr. Surface complexity of human neocortical astrocytic cells: changes with development, aging, and dementia. J Hirnforsch 1995;36: 531–537.
Rossner S, Lange-Dohna C, Zeitschel U, Perez-Polo JR. Alzheimer’s disease beta-secretase BACE1 is not a neuron-specific enzyme. J Neurochem 2005;92: 226–234.
Guenette SY. Astrocytes: a cellular player in Abeta clearance and degradation. Trends Mol Med 2003;9: 279–280.
Nicoll JA, Weller RO. A new role for astrocytes: beta-amyloid homeostasis and degradation. Trends Mol Med 2003;9: 281–282.
Apelt J, Ach K, Schliebs R. Aging-related down-regulation of neprilysin, a putative beta-amyloid-degrading enzyme, in transgenic Tg2576 Alzheimer-like mouse brain is accompanied by an astroglial upregulation in the vicinity of beta-amyloid plaques. Neurosci Lett 2003;339: 183–186.
Wyss-Coray T, Loike JD, Brionne TC, et al. Adult mouse astrocytes degrade amyloid-b in vitro and in situ. Nat Med 2003;9: 453–457.
Rossner S, Apelt J, Schliebs R, Perez-Polo JR, Bigl V. Neuronal and glial beta-secretase (BACE) protein expression in transgenic Tg2576 mice with amyloid plaque pathology. J Neurosci Res 2001;64: 437–446.
Hartlage-Rubsamen M, Zeitschel U, Apelt J, et al. Astrocytic expression of the Alzheimer’s disease b-secretase (BACE1) is stimulus-dependent. Glia 2003;41: 169–179.
Heneka MT, Sastre M, Dumitrescu-Ozimek L, et al. Focal glial activation coincides with increased BACE1 activation and precedes amyloid plaque deposition in APP[V717I] transgenic mice. J Neuroinflam 2005;2: 22.
Martins RN, Taddei K, Kendall C, Evin G, Bates KA, Harvey AR. Altered expression of apolipoprotein E, amyloid precursor protein and presenilin-1 is associated with chronic reactive gliosis in rat cortical tissue. Neuroscience 2001;106: 557–569.
Bell RD, Zlokovic BV. Neurovascular mechanisms and blood-brain barrier disorder in Alzheimer’s disease. Acta Neuropathol 2009;118: 103–113.
Iadecola C. Neurovascular regulation in the normal brain and in Alzheimer’s disease. Nat Rev Neurosci 2004;5: 347–360.
Zlokovic BV. The blood-brain barrier in health and chronic neurodegenerative disorders. Neuron 2008;57: 178–201.
Farkas E, Luiten PG. Cerebral microvascular pathology in aging and Alzheimer’s disease. Prog Neurobiol 2001;64: 575–611.
Abbott NJ, Ronnback L, Hansson E. Astrocyte-endothelial interactions at the blood-brain barrier. Nat Rev Neurosci 2006;7: 41–53.
Mosconi L, Pupi A, De Leon MJ. Brain glucose hypometabolism and oxidative stress in preclinical Alzheimer’s disease. Ann N Y Acad Sci 2008;1147: 180–195.
Parpura-Gill A, Beitz D, Uemura E. The inhibitory effects of beta-amyloid on glutamate and glucose uptakes by cultured astrocytes. Brain Res 1997;754: 65–71.
Schubert D, Soucek T, Blouw B. The induction of HIF-1 reduces astrocyte activation by amyloid beta peptide. Eur J Neurosci 2009;29: 1323–1334.
Soucek T, Cumming R, Dargusch R, Maher P, Schubert D. The regulation of glucose metabolism by HIF-1 mediates a neuroprotective response to amyloid beta peptide. Neuron 2003;39: 43–56.
Allaman I, Gavillet M, Belanger M, Laroche T, Viertl D, Lashuel HA, et al. Amyloid-b aggregates cause alterations of astrocytic metabolic phenotype: impact on neuronal viability. J Neurosci 2010;30: 3326–3338.
Blass JP, Sheu RK, Gibson GE. Inherent abnormalities in energy metabolism in Alzheimer disease. Interaction with cerebrovascular compromise. Ann N Y Acad Sci 2000;903: 204–221.
Liang WS, Reiman EM, Valla J, et al. Alzheimer’s disease is associated with reduced expression of energy metabolism genes in posterior cingulate neurons. Proc Natl Acad Sci U S A 2008;105: 4441–4446.
Bigl M, Bruckner MK, Arendt T, Bigl V, Eschrich K. Activities of key glycolytic enzymes in the brains of patients with Alzheimer’s disease. J Neural Transm 1999;106: 499–511.
Coleman P, Federoff H, Kurlan R. A focus on the synapse for neuroprotection in Alzheimer disease and other dementias. Neurology 2004;63: 1155–1162.
Elias MF, Beiser A, Wolf PA, Au R, White RF, D’Agostino RB. The preclinical phase of alzheimer disease: A 22-year prospective study of the Framingham Cohort. Arch Neurol 2000;57: 808–813.
DeKosky ST, Scheff SW. Synapse loss in frontal cortex biopsies in Alzheimer’s disease: correlation with cognitive severity. Ann Neurol 1990;27: 457–464.
Samuel W, Masliah E, Hill LR, Butters N, Terry R. Hippocampal connectivity and Alzheimer’s dementia: effects of synapse loss and tangle frequency in a two-component model. Neurology 1994;44: 2081–2088.
Terry RD, Masliah E, Salmon DP, et al. Physical basis of cognitive alterations in Alzheimer’s disease: synapse loss is the major correlate of cognitive impairment. Ann Neurol 1991;30: 572–580.
Alle H, Roth A, Geiger JR. Energy-efficient action potentials in hippocampal mossy fibers. Science 2009;325: 1405–1408.
Magistretti PJ. Neuroscience. Low-cost travel in neurons. Science 2009;325: 1349–1351.
Bilan Y, Masters CL, Barnham KJ, Bush AI, Adlard PA. Pharmacotherapeutic targets in Alzheimer’s disease. J Cell Mol Med 2009;13: 61–86.
McCall MA, Gregg RG, Behringer RR, et al. Targeted deletion in astrocyte intermediate filament (Gfap) alters neuronal physiology. Proc Natl Acad Sci U S A 1996;93: 6361–6366.
Shibuki K, Gomi H, Chen L, et al. Deficient cerebellar long-term depression, impaired eyeblink conditioning, and normal motor coordination in GFAP mutant mice. Neuron 1996;16: 587–599.
Guarente L, Kenyon C. Genetic pathways that regulate ageing in model organisms. Nature 2000;408: 255–262.
Doble A. The pharmacology and mechanism of action of riluzole. Neurology 1996;47: S233-S241.
Frizzo ME, Dall’Onder LP, Dalcin KB, Souza DO. Riluzole enhances glutamate uptake in rat astrocyte cultures. Cell Mol Neurobiol 2004;24: 123–128.
Ji HF, Shen L, Zhang HY. Beta-lactam antibiotics are multipotent agents to combat neurological diseases. Biochem Biophys Res Commun 2005;333: 661–663.
Blackburn D, Sargsyan S, Monk PN, Shaw PJ. Astrocyte function and role in motor neuron disease: a future therapeutic target? Glia 2009;57: 1251–1264.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Verkhratsky, A., Olabarria, M., Noristani, H.N. et al. Astrocytes in Alzheimer’s disease. Neurotherapeutics 7, 399–412 (2010). https://doi.org/10.1016/j.nurt.2010.05.017
Issue Date:
DOI: https://doi.org/10.1016/j.nurt.2010.05.017