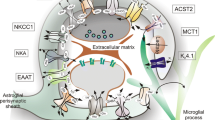
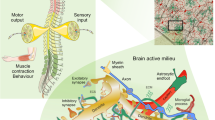
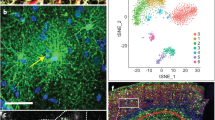
Summary
Reactive astrogliosis has long been recognized as a ubiquitous feature of CNS pathologies. Although its roles in CNS pathology are only beginning to be defined, genetic tools are enabling molecular dissection of the functions and mechanisms of reactive astrogliosis in vivo. It is now clear that reactive astrogliosis is not simply an all-or-nothing phenomenon but, rather, is a finely gradated continuum of molecular, cellular, and functional changes that range from subtle alterations in gene expression to scar formation. These changes can exert both beneficial and detrimental effects in a context-dependent manner determined by specific molecular signaling cascades. Dysfunction of either astrocytes or the process of reactive astrogliosis is emerging as an important potential source of mechanisms that might contribute to, or play primary roles in, a host of CNS disorders via loss of normal or gain of abnormal astrocyte activities. A rapidly growing understanding of the mechanisms underlying astrocyte signaling and reactive astrogliosis has the potential to open doors to identifying many molecules that might serve as novel therapeutic targets for a wide range of neurological disorders. This review considers general principles and examines selected examples regarding the potential of targeting specific molecular aspects of reactive astrogliosis for therapeutic manipulations, including regulation of glutamate, reactive oxygen species, and cytokines.
Article PDF
Similar content being viewed by others
References
Barres BA. The mystery and magic of glia: a perspective on their roles in health and disease. Neuron 2008;60: 430–440.
Nedergaard M, Ransom B, Goldman SA. New roles for astrocytes: redefining the functional architecture of the brain. Trends Neurosci 2003;26: 523–530.
Pellerin L, Bouzier-Sore AK, Aubert A, et al. Activity-dependent regulation of energy metabolism by astrocytes: an update. Glia 2007;55: 1251–1262.
Seifert G, Schilling K, Steinhäuser C. Astrocyte dysfunction in neurological disorders: a molecular perspective. Nat Rev Neurosci 2006;7: 194–206.
Sofroniew MV, Vinters HV. Astrocytes: biology and pathology. Acta Neuropathol 2010;119: 7–35.
Pekny M, Nilsson M. Astrocyte activation and reactive gliosis. Glia 2005;50: 427–434.
Sofroniew MV. Molecular dissection of reactive astrogliosis and glial scar formation. Trends Neurosci 2009;32: 638–647.
Sofroniew MV. Reactive astrocytes in neural repair and protection. Neuroscientist 2005;5: 400–407.
Darlington CL. Astrocytes as targets for neuroprotective drugs. Curr Opin Investig Drugs 2005;6: 700–703.
Fuller S, Münch G, Steele M. Activated astrocytes: a therapeutic target in Alzheimer’s disease? Expert Rev Neurother 2009;9: 1585–1594.
Sofroniew MV. Astrocyte failure as a cause of CNS dysfunction. Mol Psychiatry 2000;5: 230–232.
Rothstein JD, Dykes-Hoberg M, Pardo CA, et al. Knockout of glutamate transporters reveals a major role for astroglial transport in excitotoxicity and clearance of glutamate. Neuron 1996;16: 675–686.
Bush TG, Puvanachandra N, Horner CH, et al. Leukocyte infiltration, neuronal degeneration, and neurite outgrowth after ablation of scar-forming, reactive astrocytes in adult transgenic mice. Neuron 1999;23: 297–308.
Swanson RA, Ying W, Kauppinen TM. Astrocyte influences on ischemic neuronal death. Curr Mol Med 2004;4: 193–205.
Chen Y, Vartiainen NE, Ying W, Chan PH, Koistinaho J, Swanson RA. Astrocytes protect neurons from nitric oxide toxicity by a glutathione-dependent mechanism. J Neurochem 2001;77: 1601–1610.
Shih AY, Johnson DA, Wong G, et al. Coordinate regulation of glutathione biosynthesis and release by Nrf2-expressing glia potently protects neurons from oxidative stress. J Neurosci 2003;23: 3394–3406.
Vargas MR, Johnson DA, Sirkis DW, Messing A, Johnson JA. Nrf2 activation in astrocytes protects against neurodegeneration in mouse models of familial amyotrophic lateral sclerosis. J Neurosci 2008;28: 13574–13581.
Sarafian TA, Montes C, Imura T, et al. Disruption of astrocyte STAT3 signaling decreases mitochondrial function and increases oxidative stress in vitro. PLoS One 2010;5: e9532.
Lin JH, Lou N, Kang N, et al. A central role of connexin 43 in hypoxic preconditioning. J Neurosci 2008;28: 681–695.
Rao KV, Panickar KS, Jayakumar AR, Norenberg MD. Astrocytes protect neurons from ammonia toxicity. Neurochem Res 2005;30: 1311–1318.
Koistinaho M, Lin S, Wu X, et al. Apolipoprotein E promotes astrocyte colocalization and degradation of deposited amyloid-β peptides. Nat Med 2004;10: 719–726.
Zador Z, Stiver S, Wang V, Manley GT. Role of aquaporin-4 in cerebral edema and stroke. Handb Exp Pharmacol 2009;190: 159–170.
Faulkner JR, Herrmann JE, Woo MJ, Tansey KE, Doan NB, Sofroniew MV. Reactive astrocytes protect tissue and preserve function after spinal cord injury. J Neurosci 2004;24: 2143–2155.
Myer DJ, Gurkoff GG, Lee SM, Hovda DA, Sofroniew MV. Essential protective roles of reactive astrocytes in traumatic brain injury. Brain 2006;129: 2761–2772.
Herrmann JE, Imura T, Song B, et al. STAT3 is a critical regulator of astrogliosis and scar formation after spinal cord injury. J Neurosci 2008;28: 7231–7243.
Li L, Lundkvist A, Andersson D, et al. Protective role of reactive astrocytes in brain ischemia. J Cereb Blood Flow Metab 2008;28: 468–481.
Okada S, Nakamura M, Katoh H, et al. Conditional ablation of Stat3 or Socs3 discloses a dual role for reactive astrocytes after spinal cord injury. Nat Med 2006;12: 829–834.
Drogemuller K, Helmuth U, Brunn A, et al. Astrocyte gp130 expression is critical for the control of Toxoplasma encephalitis. J Immunol 2008;181: 2683–2693.
Voskuhl RR, Peterson RS, Song B, et al. Reactive astrocytes form scar-like perivascular barriers to leukocytes during adaptive immune inflammation of the CNS. J Neurosci 2009;29: 11511–11522.
Maragakis NJ, Rothstein JD. Mechanisms of disease: astrocytes in neurodegenerative disease. Nat Clin Pract Neurol 2006;2: 679–689.
Sattler R, Rothstein JD. Regulation and dysregulation of glutamate transporters. Handb Exp Pharmacol 2006;(175):277–303.
Dabir DV, Robinson MB, Swanson E, et al. Impaired glutamate transport in a mouse model of tau pathology in astrocytes. J Neurosci 2006;26: 644–654.
Li LB, Toan SV, Zelenaia O, et al. Regulation of astrocytic glutamate transporter expression by Akt: evidence for a selective transcriptional effect on the GLT-1/EAAT2 subtype. J Neurochem 2006;97: 759–771.
Rothstein JD, Martin LJ, Kuncl RW. Decreased glutamate transport by the brain and spinal cord in amyotrophic lateral sclerosis. N Engl J Med 1992;326: 1464–1468.
Fontana AC, de Oliveira Beleboni R, Wojewodzic MW, et al. Enhancing glutamate transport: mechanism of action of Parawixin1, a neuroprotective compound from Parawixia bistriata spider venom. Mol Pharmacol 2007;72: 1228–1237.
Fontana AC, Guizzo R, de Oliveira Beleboni R, et al. Purification of a neuroprotective component of Parawixia bistriata spider venom that enhances glutamate uptake. Br J Pharmacol 2003;139: 1297–1309.
Rothstein JD, Patel S, Regan MR, et al. β-Lactam antibiotics offer neuroprotection by increasing glutamate transporter expression. Nature 2005;433: 73–77.
Pascuzzi RM, Shefner J, Chappell AS, et al. A phase II trial of talampanel in subjects with amyotrophic lateral sclerosis. Amyotroph Lateral Scler 2010;11: 266–271.
Cornell-Bell AH, Finkbeiner SM, Cooper MS, Smith SJ. Glutamate induces calcium waves in cultured astrocytes: long-range glial signaling. Science 1990;247: 470–473.
Wang X, Takano T, Nedergaard M. Astrocytic calcium signaling: mechanism and implications for functional brain imaging. Methods Mol Biol 2009;489: 93–109.
Shigetomi E, Kracun S, Sofroniew MV, Khakh BS. A genetically targeted optical sensor to monitor calcium signals in astrocyte processes. Nat Neurosci 2010;13: 759–766.
Halassa MM, Haydon PG. Integrated brain circuits: astrocytic networks modulate neuronal activity and behavior. Annu Rev Physiol 2010;72: 335–355.
Henneberger C, Papouin T, Oliet SH, Rusakov DA. Long-term potentiation depends on release of D-serine from astrocytes. Nature 2010;463: 232–236.
Shigetomi E, Bowser DN, Sofroniew MV, Khakh BS. Two forms of astrocyte calcium excitability have distinct effects on NMDA receptor-mediated slow inward currents in pyramidal neurons. J Neurosci 2008;28: 6659–6663.
Hamby ME, Hewett JA, Hewett SJ. TGF-β1 reduces the heterogeneity of astrocytic cyclooxygenase-2 and nitric oxide synthase-2 gene expression in a stimulus-independent manner. Prostaglandins Other Lipid Mediat 2008;85: 115–124.
Hewett SJ, Csemansky CA, Choi DW. Selective potentiation of NMDA-induced neuronal injury following induction of astrocytic iNOS. Neuron 1994;13: 487–494.
Katsuse O, Iseki E, Kosaka K. Immunohistochemical study of the expression of cytokines and nitric oxide synthases in brains of patients with dementia with Lewy bodies. Neuropathology 2003;23: 9–15.
Lieb K, Engels S, Fiebich BL. Inhibition of LPS-induced iNOS and NO synthesis in primary rat microglial cells. Neurochem Int 2003;42: 131–137.
Tran EH, Hardin-Pouzet H, Verge G, Owens T. Astrocytes and microglia express inducible nitric oxide synthase in mice with experimental allergic encephalomyelitis. J Neuroimmunol 1997;74: 121–129.
Liberatore GT, Jackson-Lewis V, Vukosavic S, et al. Inducible nitric oxide synthase stimulates dopaminergic neurodegeneration in the MPTP model of Parkinson disease. Nat Med 1999;5: 1403–1409.
Pozza M, Bettelli C, Aloe L, Giardino L, Calzà L. Further evidence for a role of nitric oxide in experimental allergic encephalomyelitis: aminoguanidine treatment modifies its clinical evolution. Brain Res 2000;855: 39–46.
Smith KJ, Lassmann H. The role of nitric oxide in multiple sclerosis. Lancet Neurol 2002;1: 232–241.
Iadecola C, Zhang F, Casey R, Nagayama M, Ross ME. Delayed reduction of ischemic brain injury and neurological deficits in mice lacking the inducible nitric oxide synthase gene. J Neurosci 1997;17: 9157–9164.
Iadecola C, Zhang F, Xu X. Inhibition of inducible nitric oxide synthase ameliorates cerebral ischemic damage. Am J Physiol 1995;268: R286-R292.
Shin T, Kim S, Moon C, Wie M, Kim H. Aminoguanidine-induced amelioration of autoimmune encephalomyelitis is mediated by reduced expression of inducible nitric oxide synthase in the spinal cord. Immunol Invest 2000;29: 233–241.
Shin T, Tanuma N, Kim S, et al. An inhibitor of inducible nitric oxide synthase ameliorates experimental autoimmune myocarditis in Lewis rats. J Neuroimmunol 1998;92: 133–138.
Iadecola C, Li J, Ebner TJ, Xu X. Nitric oxide contributes to functional hyperemia in cerebellar cortex. Am J Physiol 1995;268: R1153-R1162.
Nathan C, Calingasan N, Nezezon J, et al. Protection from Alzheimer’s-like disease in the mouse by genetic ablation of inducible nitric oxide synthase. J Exp Med 2005;202: 1163–1169.
Lei SZ, Pan ZH, Aggarwal SK, et al. Effect of nitric oxide production on the redox modulatory site of the NMDA receptor-channel complex. Neuron 1992;8: 1087–1099.
Colton CA, Vitek MP, Wink DA, et al. NO synthase 2 (NOS2) deletion promotes multiple pathologies in a mouse model of Alzheimer’s disease. Proc Natl Acad Sci U S A 2006;103: 12867–12872.
Lipton SA, Choi YB, Pan ZH, et al. A redox-based mechanism for the neuroprotective and neurodestructive effects of nitric oxide and related nitroso-compounds. Nature 1993;364: 626–632.
Vidwans AS, Kim S, Coffin DO, Wink DA, Hewett SJ. Analysis of the neuroprotective effects of various nitric oxide donor compounds in murine mixed cortical cell culture. J Neurochem 1999;72: 1843–1852.
Szabo C, Zingarelli B, O’Connor M, Salzman AL. DNA strand breakage, activation of poly (ADP-ribose) synthetase, and cellular energy depletion are involved in the cytotoxicity of macrophages and smooth muscle cells exposed to peroxynitrite. Proc Natl Acad Sci U S A 1996;93: 1753–1758.
Zingarelli B, O’Connor M, Wong H, Salzman AL, Szabó C. Peroxynitrite-mediated DNA strand breakage activates poly-adenosine diphosphate ribosyl synthetase and causes cellular energy depletion in macrophages stimulated with bacterial lipopolysaccharide. J Immunol 1996;156: 350–358.
Hewett SJ, Muir JK, Lobner D, Symons A, Choi DW. Potentiation of oxygen-glucose deprivation-induced neuronal death after induction of iNOS. Stroke 1996;27: 1586–1591.
Vidwans AS, Hewett SJ. Enhanced release of synaptic glutamate underlies the potentiation of oxygen-glucose deprivation-induced neuronal injury after induction of NOS-2. Exp Neurol 2004;190: 91–101.
Koehler RC, Roman RJ, Harder DR. Astrocytes and the regulation of cerebral blood flow. Trends Neurosci 2009;32: 160–169.
Barber SC, Shaw PJ. Oxidative stress in ALS: key role in motor neuron injury and therapeutic target. Free Radic Biol Med 2010;48: 629–641.
Ilieva H, Polymenidou M, Cleveland DW. Non-cell autonomous toxicity in neurodegenerative disorders: ALS and beyond. J Cell Biol 2009;187: 761–772.
Yamanaka K, Chun SJ, Boillee S, et al. Astrocytes as determinants of disease progression in inherited amyotrophic lateral sclerosis. Nat Neurosci 2008;11: 251.
Rothstein JD, Van Kammen M, Levey AI, Martin LJ, Kuncl RW. Selective loss of glial glutamate transporter GLT-1 in amyotrophic lateral sclerosis. Ann Neurol 1995;38: 73–84.
Bruijn LI, Becher MW, Lee MK, et al. ALS-linked SOD1 mutant G85R mediates damage to astrocytes and promotes rapidly progressive disease with SOD1-containing inclusions. Neuron 1997;18: 327–338.
Lepore AC, Rauck B, Dejea C, et al. Focal transplantation-based astrocyte replacement is neuroprotective in a model of motor neuron disease. Nat Neurosci 2008;11: 1294–1301.
Lleo A, Galea E, Sastre M. Molecular targets of non-steroidal anti-inflammatory drugs in neurodegenerative diseases. Cell Mol Life Sci 2007;64: 1403–1418.
Asanuma M, Miyazaki I. Nonsteroidal anti-inflammatory drugs in Parkinson’s disease: possible involvement of quinone formation. Expert Rev Neurotherapeutics 2006;6: 1313–1325.
Heneka MT, Sastre M, Dumitrescu-Ozimek L, et al. Acute treatment with the PPARγ agonist pioglitazone and ibuprofen reduces glial inflammation and Aβ1-42 levels in APPV717I transgenic mice. Brain 2005;128: 1442–1453.
Drachman DB, Frank K, Dykes-Hoberg M, et al. Cyclooxygenase 2 inhibition protects motor neurons and prolongs survival in a transgenic mouse model of ALS. Ann Neurol 2002;52: 771–778.
Bae MK, Kim SR, Lee HJ, et al. Aspirin-induced blockade of NF-κB activity restrains up-regulation of glial fibrillary acidic protein in human astroglial cells. Biochim Biophys Acta 2006;1763: 282–289.
Dringen R, Hirrlinger J. Glutathione pathways in the brain. Biol Chem 2003;384: 505–516.
Bush TG, Savidge TC, Freeman TC, et al. Fulminant jejunoileitis following ablation of enteric glia in adult transgenic mice. Cell 1998;93: 189–201.
Savidge TC, Newman P, Pothoulakis C, et al. Enteric glia regulate intestinal barrier function and inflammation via release of S-nitrosoglutathione. Gastroenterology 2007;132: 1344–1358.
Hamby ME, Hewett JA, Hewett SJ. TGF-β1 potentiates astrocytic nitric oxide production by expanding the population of astrocytes that express NOS-2. Glia 2006;54: 566–577.
Hewett SJ. Interferon-gamma reduces cyclooxygenase-2-mediated prostaglandin E2 production from primary mouse astrocytes independent of nitric oxide formation. J Neuroimmunol 1999;94: 134–143.
Hewett SJ, Corbett JA, McDaniel ML, Choi DW. Interferongamma and interleukin-1 β induce nitric oxide formation from primary mouse astrocytes. Neurosci Lett 1993;164: 229–232.
Hamby ME, Gragnolati AR, Hewett SJ, Hewett JA. TGFβ1 and TNFα potentiate nitric oxide production in astrocyte cultures by recruiting distinct subpopulations of cells to express NOS-2. Neurochem Int 2008;52: 962–971.
Minghetti L, Polazzi E, Nicolini A, Levi G. Opposite regulation of prostaglandin E2 synthesis by transforming growth factor-β1 and interleukin 10 in activated microglial cultures. J Neuroimmunol 1998;82: 31–39.
Saad B, Constam DB, Ortmann R, Moos M, Fontana A, Schachner M. Astrocyte-derived TGF-β2 and NGF differentially regulate neural recognition molecule expression by cultured astrocytes. J Cell Biol 1991;115: 473–484.
Krieglstein K, Reuss B, Maysinger D, Unsicker K. Short communication: transforming growth factor-β mediates the neurotrophic effect of fibroblast growth factor-2 on midbrain dopaminergic neurons. Eur J Neurosci 1998;10: 2746–2750.
Villoslada P, Genain CP. Role of nerve growth factor and other trophic factors in brain inflammation. Prog Brain Res 2004;146: 403–414.
Ridet JL, Malhotra SK, Privat A, Gage FH. Reactive astrocytes: cellular and molecular cues to biological function. Trends Neurosci 1997;20: 570–577.
Mena MA, Garcïa de Yébenes J. Glial cells as players in parkinsonism: the “good,” the “bad,” and the “mysterious” glia. Neuroscientist 2008;14: 544–560.
Ishibashi T, Dakin KA, Stevens B, et al. Astrocytes promote myelination in response to electrical impulses. Neuron 2006;49: 823–832.
Lindholm D, Castrén E, Kiefer R, Zafra F, Thoenen H. Transforming growth factor-β1 in the rat brain: increase after injury and inhibition of astrocyte proliferation. J Cell Biol 1992;117: 395–400.
Wang X, Yue TL, White RF, Barone FC, Feuerstein GZ. Transforming growth factor-β1 exhibits delayed gene expression following focal cerebral ischemia. Brain Res Bull 1995;36: 607–609.
Krupinski J, Kumar P, Kumar S, Kaluza J. Increased expression of TGF-β1 in brain tissue after ischemic stroke in humans. Stroke 1996;27: 852–857.
Ata AK, Funa K, Olsson Y. Expression of various TGF-β isoforms and type I receptor in necrotizing human brain lesions. Acta Neuropathol 1997;93: 326–333.
Ali C, Docagne F, Nicole O, et al. Increased expression of transforming growth factor-β after cerebral ischemia in the baboon: an endogenous marker of neuronal stress? J Cereb Blood Flow Metab 2001;21: 820–827.
Zetterberg H, Andreasen N, Blennow K. Increased cerebrospinal fluid levels of transforming growth factor-β1 in Alzheimer’s disease. Neurosci Lett 2004;367: 194–196.
Kiefer R, Streit WJ, Toyka KV, Kreutzberg GW, Hartung HP. Transforming growth factor-β1: a lesion-associated cytokine of the nervous system. Int J Dev Neurosci 1995;13: 331–339.
Kiefer R, Lindholm D, Kreutzberg GW. Interleukin-6 and transforming growth factor-β1 mRNAs are induced in rat facial nucleus following motoneuron axotomy. Eur J Neurosci 1993;5: 775–781.
Knuckey NW, Finch P, Palm DE, et al. Differential neuronal and astrocytic expression of transforming growth factor β isoforms in rat hippocampus following transient forebrain ischemia. Brain Res Mol Brain Res 1996;40: 1–14.
Morgan TE, Nichols NR, Pasinetti GM, Finch CE. TGF-β1 mRNA increases in macrophage/microglial cells of the hippocampus in response to deafferentation and kainic acid-induced neurodegeneration. Exp Neurol 1993;120: 291–301.
Wahl SM, Allen JB, McCartney-Francis N, et al. Macrophage- and astrocyte-derived transforming growth factor β as a mediator of central nervous system dysfunction in acquired immune deficiency syndrome. J Exp Med 1991;173: 981–991.
Krupinski J, Vodovotz Y, Li C, et al. Inducible nitric oxide production and expression of transforming growth factor-β1 in serum and CSF after cerebral ischaemic stroke in man. Nitric Oxide 1998;2: 442–453.
Morganti-Kossmann MC, Hans VH, Lenzlinger PM, et al. TGF-β is elevated in the CSF of patients with severe traumatic brain injuries and parallels blood-brain barrier function. J Neurotrauma 1999;16: 617–628.
Huang CC, Chang YC, Chow NH, Wang ST. Level of transforming growth factor β1 is elevated in cerebrospinal fluid of children with acute bacterial meningitis. J Neurol 1997;244: 634–638.
De Groot CJ, Montagne L, Barten AD, Sminia P, Van Der Valk P. Expression of transforming growth factor (TGF)-β1, -β2, and -β3 isoforms and TGF-β type I and type II receptors in multiple sclerosis lesions and human adult astrocyte cultures. J Neuropathol Exp Neurol 1999;58: 174–187.
Peress NS, Perillo E, Seidman RJ. Glial transforming growth factor (TGF)-β isotypes in multiple sclerosis: differential glial expression of TGF-β1, 2 and 3 isotypes in multiple sclerosis. J Neuroimmunol 1996;71: 115–123.
Vawter MP, Dillon-Carter O, Tourtellotte WW, Carvey P, Freed WJ. TGFβ1 and TGFβ2 concentrations are elevated in Parkinson’s disease in ventricular cerebrospinal fluid. Exp Neurol 1996;142: 313–322.
Miyazawa K, Shinozaki M, Hara T, Furuya T, Miyazono K. Two major Smad pathways in TGF-β superfamily signalling. Genes Cells 2002;7: 1191–1204.
Dhandapani KM, Brann DW. Transforming growth factor-β: a neuroprotective factor in cerebral ischemia. Cell Biochem Biophys 2003;39: 13–22.
Buisson A, Lesné S, Docagne F, et al. Transforming growth factor-β and ischemic brain injury. Cell Mol Neurobiol 2003;23: 539–550.
Boche D, Cunningham C, Gauldie J, Perry VH. Transforming growth factor-β1-mediated neuroprotection against excitotoxic injury in vivo. J Cereb Blood Flow Metab 2003;23: 1174–1182.
Henrich-Noack P, Prehn JH, Krieglstein J. TGF-β1 protects hippocampal neurons against degeneration caused by transient global ischemia. Dose-response relationship and potential neuroprotective mechanisms. Stroke 1996;27: 1609–1614; discussion 1615.
Prehn JH, Backhauss C, Krieglstein J. Transforming growth factor-β1 prevents glutamate neurotoxicity in rat neocortical cultures and protects mouse neocortex from ischemic injury in vivo. J Cereb Blood Flow Metab 1993;13: 521–525.
Brionne TC, Tesseur I, Masliah E, Wyss-Coray T. Loss of TGF-β1 leads to increased neuronal cell death and microgliosis in mouse brain. Neuron 2003;40: 1133–1145.
Wyss-Coray T, Borrow P, Brooker MJ, Mucke L. Astroglial overproduction of TGF-β1 enhances inflammatory central nervous system disease in transgenic mice. J Neuroimmunol 1997;77: 45–50.
Wyss-Coray T, Masliah E, Mallory M, et al. Amyloidogenic role of cytokine TGF-β1 in transgenic mice and in Alzheimer’s disease. Nature 1997;389: 603–606.
Wyss-Coray T, Feng L, Masliah E, et al. Increased central nervous system production of extracellular matrix components and development of hydrocephalus in transgenic mice overexpressing transforming growth factor-β1. Am J Pathol 1995;147: 53–67.
Lesné S, Docagne F, Gabriel C, et al. Transforming growth factor-β1 potentiates amyloid-β generation in astrocytes and in transgenic mice. J Biol Chem 2003;278: 18408–18418.
Burton T, Liang B, Dibrov A, Amara F. Transcriptional activation and increase in expression of Alzheimer’s β-amyloid precursor protein gene is mediated by TGF-β in normal human astrocytes. Biochem Biophys Res Commun 2002;295: 702–712.
Burton T, Liang B, Dibrov A, Amara F. Transforming growth factor-β-induced transcription of the Alzheimer β-amyloid precursor protein gene involves interaction between the CTCF-complex and Smads. Biochem Biophys Res Commun 2002;295: 713–723.
Johns LD, Flanders KC, Ranges GE, Sriram S. Successful treatment of experimental allergic encephalomyelitis with transforming growth factor-β1. J Immunol 1991;147: 1792–1796.
Amara FM, Junaid A, Clough RR, Liang B. TGF-β1, regulation of Alzheimer amyloid precursor protein mRNA expression in a normal human astrocyte cell line: mRNA stabilization. Brain Res Mol Brain Res 1999;71: 42–49.
Smith GM, Strunz C. Growth factor and cytokine regulation of chondroitin sulfate proteoglycans by astrocytes. Glia 2005;52: 209–218.
Wang Y, Moges H, Bharucha Y, Symes A. Smad3 null mice display more rapid wound closure and reduced scar formation after a stab wound to the cerebral cortex. Exp Neurol 2007;203: 168–184.
da Cunha A, Jefferson JA, Jackson RW, Vitković L. Glial cell-specific mechanisms of TGF-β1 induction by IL-1 in cerebral cortex. J Neuroimmunol 1993;42: 71–85.
Vivien D, Bernaudin M, Buisson A, Divoux D, MacKenzie ET, Nouvelot A. Evidence of type I and type II transforming growth factor-β receptors in central nervous tissues: changes induced by focal cerebral ischemia. J Neurochem 1998;70: 2296–2304.
Pratt BM, McPherson JM. TGF-β in the central nervous system: potential roles in ischemic injury and neurodegenerative diseases. Cytokine Growth Factor Rev 1997;8: 267–292.
Flanders KC, Ren RF, Lippa CF. Transforming growth factor-betas in neurodegenerative disease. Prog Neurobiol 1998;54: 71–85.
König HG, Kögel D, Rami A, Prehn JH. TGF-β1 activates two distinct type I receptors in neurons: implications for neuronal NF-κB signaling. J Cell Biol 2005;168: 1077–1086.
Lux A, Salway F, Dressman HK, et al. ALK1 signalling analysis identifies angiogenesis related genes and reveals disparity between TGF-β and constitutively active receptor induced gene expression. BMC Cardiovasc Disord 2006;6: 13.
Le Y, Iribarren P, Gong W, Cui Y, Zhang X, Wang JM. TGF-β1 disrupts endotoxin signaling in microglial cells through Smad3 and MAPK pathways. J Immunol 2004;173: 962–968.
Hamby ME, Hewett JA, Hewett SJ. Smad3-dependent signaling underlies the TGF-β1-mediated enhancement in astrocytic iNOS expression. Glia 2010;58: 1282–1891.
Spooren A, Kooijman R, Lintermans B, et al. Cooperation of NFκB and CREB to induce synergistic IL-6 expression in astrocytes. Cell Signal 2010;22: 871–881.
Fogal B, Hewett SJ. Interleukin-1β: a bridge between inflammation and excitotoxicity? J Neurochem 2008;106: 1–23.
John GR, Lee SC, Song X, Rivieccio M, Brosnan CF. IL-1-regulated responses in astrocytes: Relevance to injury and recovery. Glia 2005;49: 161–176.
Brambilla R, Bracchi-Ricard V, Hu W-H, et al. Inhibition of astroglial nuclear factor κB reduces inflammation and improves functional recovery after spinal cord injury. J Exp Med 2005;202: 145–156.
Saccani S, Pantano S, Natoli G. p38-Dependent marking of inflammatory genes for increased NF-κB recruitment. Nat Immunol 2002;3: 69–75.
Rahman I, Marwick J, Kirkham P. Redox modulation of chromatin remodeling: impact on histone acetylation and deacetylation, NF-κB and pro-inflammatory gene expression. Biochem Pharmacol 2004;68: 1255–1267.
Muegge K. Preparing the target for the bullet. Nat Immunol 2002;3: 16–17.
Saccani S, Pantano S, Natoli G. Two waves of nuclear factor κB recruitment to target promoters. J Exp Med 2001;193: 1351–1359.
Natoli G, Saccani S, Bosisio D, Marazzi I. Interactions of NF-κB with chromatin: the art of being at the right place at the right time. Nat Immunol 2005;6: 439–445.
Ershler WB, Sun WH, Binkley N. The role of interleukin-6 in certain age-related diseases. Drugs Aging 1994;5: 358–365.
Taga T, Kishimoto T. gp130 and the interleukin-6 family of cytokines. Annu Rev Immunol 1997;15: 797–819.
Penkowa M, Giralt M, Lago N, et al. Astrocyte-targeted expression of IL-6 protects the CNS against a focal brain injury. Exp Neurol 2003;181: 130–148.
Penkowa M, Camats J, Hadberg H, et al. Astrocyte-targeted expression of interleukin-6 protects the central nervous system during neuroglial degeneration induced by 6-aminonicotinamide. J Neurosci Res 2003;73: 481–496.
Swartz KR, Liu F, Sewell D, et al. Interleukin-6 promotes posttraumatic healing in the central nervous system. Brain Res 2001: 896: 86–95.
Campbell IL, Abraham CR, Masliah E, et al. Neurologic disease induced in transgenic mice by cerebral overexpression of interleukin 6. Proc Natl Acad Sci U S A 1993;90: 10061–10065.
Quintana A, Muller M, Frausto RF, et al. Site-specific production of IL-6 in the central nervous system retargets and enhances the inflammatory response in experimental autoimmune encephalomyelitis. J Immunol 2009;183: 2079–2088.
Sriram K, Benkovic SA, Hebert MA, Miller DB, O’Callaghan JP. Induction of gp130-related cytokines and activation of JAK2/STAT3 pathway in astrocytes precedes up-regulation of glial fibrillary acidic protein in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine model of neurodegeneration: key signaling pathway for astrogliosis in vivo? J Biol Chem 2004;279: 19936–19947.
Nakashima K, Yanagisawa M, Arakawa H, et al. Synergistic signaling in fetal brain by STAT3—Smadl complex bridged by p300. Science 1999;284: 479–482.
Fan G, Martinowich K, Chin MH, et al. DNA methylation controls the timing of astrogliogenesis through regulation of JAK-STAT signaling. Development 2005;132: 3345–3356.
Herrmann JE, Imura T, Song B, et al. STAT3 is a critical regulator of astrogliosis and scar formation after spinal cord injury. J Neurosci 2008;28: 7231–7243.
Su Z, Yuan Y, Cao L, et al. Triptolide promotes spinal cord repair by inhibiting astrogliosis and inflammation. Glia 2010: 58: 901–915.
Gong Y, Xue B, Jiao J, Jing L, Wang X. Triptolide inhibits COX-2 expression and PGE2 release by suppressing the activity of NF-κB and JNK in LPS-treated microglia. J Neurochem 2008;107: 779–788.
Levison SW, Jiang FJ, Stoltzfus OK, Ducceschi MH. IL-6-type cytokines enhance epidermal growth factor-stimulated astrocyte proliferation. Glia 2000;32: 328–337.
Gadea A, Schinelli S, Gallo V. Endothelin-1 regulates astrocyte proliferation and reactive gliosis via a JNK/c-Jun signaling pathway. J Neurosci 2008;28: 2394–2408.
Khakh BS, North RA. P2X receptors as cell-surface ATP sensors in health and disease. Nature 2006;442: 527–532.
Bowser DN, Khakh BS. Vesicular ATP is the predominant cause of intercellular calcium waves in astrocytes. J Gen Physiol 2007: 129: 485–491.
Halassa MM, Fellin T, Haydon PG. Tripartite synapses: roles for astrocytic purines in the control of synaptic physiology and behavior. Neuropharmacology 2009;57: 343–346.
Neary JT, Kang Y, Shi YF. Signaling from nucleotide receptors to protein kinase cascades in astrocytes. Neurochem Res 2004: 29: 2037–2042.
Neary JT, Kang Y, Willoughby KA, Ellis EF. Activation of extracellular signal-regulated kinase by stretch-induced injury in astrocytes involves extracellular ATP and P2 purinergic receptors. J Neurosci 2003;23: 2348–2356.
Wanner IB, Deik A, Tones M, et al. A new in vitro model of the glial scar inhibits axon growth. Glia 2008;56: 1691–1709.
Wang X, Arcuino G, Takano T, et al. P2X7 receptor inhibition improves recovery after spinal cord injury. Nat Med 2004;10: 821–827.
Peng W, Cotrina ML, Han X, et al. Systemic administration of an antagonist of the ATP-sensitive receptor P2X7 improves recovery after spinal cord injury. Proc Natl Acad Sci U S A 2009;106: 12489–12493.
Urdinguio RG, Sanchez-Mut JV, Esteller M. Epigenetic mechanisms in neurological diseases: genes, syndromes, and therapies. Lancet Neurol 2009;8: 1056–1072.
Hamby ME, Coskun V, Sun YE. Transcriptional regulation of neuronal differentiation: the epigenetic layer of complexity. Biochim Biophys Acta 2008;1779: 432–437.
Camelo S, Iglesias AH, Hwang D, et al. Transcriptional therapy with the histone deacetylase inhibitor trichostatin A ameliorates experimental autoimmune encephalomyelitis. J Neuroimmunol 2005;164: 10–21.
Kim HJ, Rowe M, Ren M, Hong JS, Chen PS, Chuang DM. Histone deacetylase inhibitors exhibit anti-inflammatory and neuroprotective effects in a rat permanent ischemic model of stroke: multiple mechanisms of action. J Pharmacol Exp Ther 2007: 321: 892–901.
Faraco G, Pancani T, Formentini L, et al. Pharmacological inhibition of histone deacetylases by suberoylanilide hydroxamic acid specifically alters gene expression and reduces ischemic injury in the mouse brain. Mol Pharmacol 2006;70: 1876–1884.
Ren M, Leng Y, Jeong M, Leeds PR, Chuang DM. Valproic acid reduces brain damage induced by transient focal cerebral ischemia in rats: potential roles of histone deacetylase inhibition and heat shock protein induction. J Neurochem 2004;89: 1358–1367.
Chen PS, Peng GS, Li G, et al. Valproate protects dopaminergic neurons in midbrain neuron/glia cultures by stimulating the release of neurotrophic factors from astrocytes. Mol Psychiatry 2006;11: 1116–1125.
Faraco G, Pittelli M, Cavone L, et al. Histone deacetylase (HDAC) inhibitors reduce the glial inflammatory response in vitro and in vivo. Neurobiol Dis 2009;36: 269–279.
Suh HS, Choi S, Khattar P, Choi N, Lee SC. Histone deacetylase inhibitors suppress the expression of inflammatory and innate immune response genes in human microglia and astrocytes. J Neuroimmune Pharmacol 2010 Feb 17 [Epub ahead of print].
Morland C, Boldingh KA, Iversen EG, Hassel B. Valproate is neuroprotective against malonate toxicity in rat striatum: an association with augmentation of high-affinity glutamate uptake. J Cereb Blood Flow Metab 2004;24: 1226–1234.
Perisic T, Zimmermann N, Kirmeier T, et al. Valproate and amitriptyline exert common and divergent influences on global and gene promoter-specific chromatin modifications in rat primary astrocytes. Neuropsychopharmacology 2009;35: 792–805.
Trinka E. The use of valproate and new antiepileptic drugs in status epilepticus. Epilepsia 2007;48 Suppl 8: 49–51.
Michaelis M, Doerr HW, Cinatl J Jr. Valproic acid as anti-cancer drug. Curr Pharm Des 2007;13: 3378–3393.
Langley B, Gensert JM, Beal MF, Ratan RR. Remodeling chromatin and stress resistance in the central nervous system: histone deacetylase inhibitors as novel and broadly effective neuroprotective agents. Curr Drug Targets CNS Neurol Disord 2005;4: 41–50.
Kazantsev AG, Thompson LM. Therapeutic application of histone deacetylase inhibitors for central nervous system disorders. Nat Rev Drug Discov 2008;7: 854–868.
Balasubramanyam K, Varier RA, Altaf M, et al. Curcumin, a novel p300/CREB-binding protein-specific inhibitor of acetyl-transferase, represses the acetylation of histone/nonhistone proteins and histone acetyltransferase-dependent chromatin transcription. J Biol Chem 2004;279: 51163–51171.
Marcu MG, Jung YJ, Lee S, et al. Curcumin is an inhibitor of p300 histone acetyltransferase. Med Chem 2006;2: 169–174.
Natarajan C, Bright JJ. Curcumin inhibits experimental allergic encephalomyelitis by blocking IL-12 signaling through Janus kinase-STAT pathway in T lymphocytes. J Immunol 2002;168: 6506–6513.
Laird MD, Sukumari-Ramesh S, Swift AEB, Meiler SE, Vender JR, Dhandapani KM. Curcumin attenuates cerebral edema following traumatic brain injury in mice: a possible role for aquaporin-4? J Neurochem 2010;113: 637–648.
Lin MS, Lee YH, Chiu WT, Hung KS. Curcumin provides neuroprotection after spinal cord injury. J Surg Res 2010 Aug 5 [Epub ahead of print].
Zador Z, Bloch O, Yao X, Manley GT. Aquaporins: role in cerebral edema and brain water balance. Prog Brain Res 2007;161: 185–194.
Kim JH, Lee YW, Park KA, Lee WT, Lee JE. Agmatine attenuates brain edema through reducing the expression of aquaporin-1 after cerebral ischemia. J Cereb Blood Flow Metab 2010;30: 943–949.
Filippidis AS, Kalani MY, Rekate HL. Hydrocephalus and aquaporins: lessons learned from the bench. Childs Nerv Syst 2010 Jul 13 [Epub ahead of print].
Benarroch EE. Neuron-astrocyte interactions: partnership for normal function and disease in the central nervous system. Mayo Clinic Proceedings 2005;80: 1326–1338.
Nag S, Manias J, Stewart D. Pathology and new players in the pathogenesis of brain edema. Acta Neuropathologica 2009;118: 197–217.
Lennon VA, Kryzer TJ, Pittock SJ, Verkman AS, Hinson SR. IgG marker of optic-spinal multiple sclerosis binds to the aquaporin-4 water channel. J Exp Med 2005;202: 473–477.
Hinson SR, McKeon A, Lennon VA. Neurological autoimmunity targeting aquaporin-4. Neuroscience 2010;168: 1009–1018.
Roemer SF, Parisi JE, Lennon VA, et al. Pattern-specific loss of aquaporin-4 immunoreactivity distinguishes neuromyelitis optica from multiple sclerosis. Brain 2007;130: 1194–1205.
Hu W, Lucchinetti CF. The pathological spectrum of CNS inflammatory demyelinating diseases. Semin Immunopathol 2009;31: 439–453.
Sharma R, Fischer MT, Bauer J, et al. Inflammation induced by innate immunity in the central nervous system leads to primary astrocyte dysfunction followed by demyelination. Acta Neuropathol 2010;120: 223–236.
Voskuhl RR, Palaszynski K. Sex hormones in experimental autoimmune encephalomyelitis: implications for multiple sclerosis. Neuroscientist 2001;7: 258–270.
Orthmann-Murphy J, Abrams C, Scherer S. Gap junctions couple astrocytes and oligodendrocytes. J Mol Neurosci 2008;35: 101–116.
Dermietzel R, Hertberg EL, Kessler JA, Spray DC. Gap junctions between cultured astrocytes: immunocytochemical, molecular, and electrophysiological analysis. J Neurosci 1991;11: 1421–1432.
Duffy HS, John GR, Lee SC, Brosnan CF, Spray DC. Reciprocal regulation of the junctional proteins claudin-1 and connexin43 by interleukin-1β in primary human fetal astrocytes. J Neurosci 2000;20: RC114.
Blomstrand F, Aberg ND, Eriksson PS, Hansson E, Rönnbäck L. Extent of intercellular calcium wave propagation is related to gap junction permeability and level of connexin-43 expression in astrocytes in primary cultures from four brain regions. Neuroscience 1999;92: 255–265.
Giaume C, Koulakoff A, Roux L, Holcman D, Rouach N. Astroglial networks: a step further in neuroglial and gliovascular interactions. Nat Rev Neurosci 2010;11: 87–89.
Rouach N, Avignone E, Même W, et al. Gap junctions and connexin expression in the normal and pathological central nervous system. Biol Cell 2002;94: 457–475.
Kielian T. Glial connexins and gap junctions in CNS inflammation and disease. J Neurochem 2008;106: 1000–1016.
Olsen ML, Sontheimer H. Functional implications for Kir4.1 channels in glial biology: from K+ buffering to cell differentiation. J Neurochem 2008;107: 589–601.
Mori T, Town T, Tan J, et al. Arundic acid ameliorates cerebral amyloidosis and gliosis in Alzheimer transgenic mice. J Pharmacol Exp Ther 2006;318: 571–578.
Oki C, Watanabe Y, Yokoyama H, Shimoda T, Kato H, Araki T. Delayed treatment with arundic acid reduces the MPTP-induced neurotoxicity in mice. Cell Mol Neurobiol 2008;28: 417–430.
Tateishi N, Mori T, Kagamiishi Y, et al. Astrocytic activation and delayed infarct expansion after permanent focal ischemia in rats. Part II: suppression of astrocytic activation by a novel agent (R)-(−)-2-propyloctanoic acid (ONO-2506) leads to mitigation of delayed infarct expansion and early improvement of neurologic deficits. J Cereb Blood Flow Metab 2002;22: 723–734.
Behrstock S, Ebert AD, Klein S, Schmitt M, Moore JM, Svendsen CN. Lesion-induced increase in survival and migration of human neural progenitor cells releasing GDNF. Cell Transplant 2008;17: 753–762.
Suzuki M, McHugh J, Tork C, et al. GDNF secreting human neural progenitor cells protect dying motor neurons, but not their projection to muscle, in a rat model of familial ALS. PLoS One 2007;2: e689.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hamby, M.E., Sofroniew, M.V. Reactive astrocytes as therapeutic targets for CNS disorders. Neurotherapeutics 7, 494–506 (2010). https://doi.org/10.1016/j.nurt.2010.07.003
Issue Date:
DOI: https://doi.org/10.1016/j.nurt.2010.07.003