Abstract
Purpose
This review aimed to discuss the conflicting findings from resistin research in rodents and humans as well as recent advances in our understanding of resistin’s role in obesity and insulin resistance.
Methods
A comprehensive review and synthesis of resistin’s role in obesity and insulin resistance as well as conflicting findings from resistin research in rodents and humans.
Results
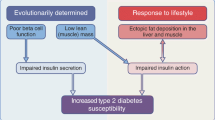
In rodents, resistin is increased in high-fat/high-carbohydrate-fed, obese states characterized by impaired glucose uptake and insulin sensitivity. Resistin plays a causative role in the development of insulin resistance in rodents via 5′ AMP-activated protein kinase (AMPK)-dependent and AMPK-independent suppressor of cytokine signaling-3 (SOCS-3) signaling. In contrast to rodents, human resistin is primarily secreted by peripheral-blood mononuclear cells (PBMCs) as opposed to white adipocytes. Circulating resistin levels have been positively associated with central/visceral obesity (but not BMI) as well as insulin resistance, while other studies show no such association. Human resistin has a role in pro-inflammatory processes that have been conclusively associated with obesity and insulin resistance. PBMCs, as well as vascular cells, have been identified as the primary targets of resistin’s pro-inflammatory activity via nuclear factor-κB (NF-κB, p50/p65) and other signaling pathways.
Conclusion
Mounting evidence reveals a continuing disconnect between resistin’s role in rodents and humans due to significant differences between these two species with respect to resistin’s gene and protein structure, differential gene regulation, tissue-specific distribution, and insulin resistance induction as well as a paucity of evidence regarding the resistin receptor and downstream signaling mechanisms of action.
Similar content being viewed by others
References
A’t Hart B, Amor S, Jonker M (2004) Evaluating the validity of animal models for research into therapies for immune-based disorders. Drug Discov Today 9:517–524
Abate N, Sallam HS, Rizzo M, Nikolic D, Obradovic M, Bjelogrlic P et al (2014) Resistin: an inflammatory cytokine. Role in cardiovascular diseases, diabetes and the metabolic syndrome. Curr Pharm Des 20:4961–4969
Abdul-Ghani MA, Tripathy D, DeFronzo RA (2006) Contributions of β-cell dysfunction and insulin resistance to the pathogenesis of impaired glucose tolerance and impaired fasting glucose. Diab Care 29:1130–1139
Al Hannan F, Culligan KG (2015) Human resistin and the RELM of Inflammation in diabesity. Diabetol Metab Syndr 7:1–11
Amal S, Pasha HF, Rashad NM (2013) Association of resistin gene polymorphisms with insulin resistance in Egyptian obese patients. Gene 515:233–238
Amato MC, Pizzolanti G, Torregrossa V, Misiano G, Milano S, Giordano C (2014) Visceral adiposity index (VAI) is predictive of an altered adipokine profile in patients with type 2 diabetes. PLoS One 9:e91969
Asano H, Izawa H, Nagata K, Nakatochi M, Kobayashi M, Hirashiki A et al (2010) Plasma resistin concentration determined by common variants in the resistin gene and associated with metabolic traits in an aged Japanese population. Diabetologia 53:234–246
Asensio C, Cettour-Rose P, Theander-Carrillo C, Rohner-Jeanrenaud F, Muzzin P (2004) Changes in glycemia by leptin administration or high-fat feeding in rodent models of obesity/type 2 diabetes suggest a link between resistin expression and control of glucose homeostasis. Endocrinology 145:2206–2213
Azuma K, Katsukawa F, Oguchi S, Murata M, Yamazaki H, Shimada A et al (2003) Correlation between serum resistin level and adiposity in obese individuals. Obes Res 11:997–1001
Banerjee RR, Rangwala SM, Shapiro JS, Rich AS, Rhoades B, Qi Y et al (2004) Regulation of fasted blood glucose by resistin. Science 303:1195–1198
Barbosa-da-Silva S, Fraulob-Aquino JC, Lopes JR, Mandarim-de-Lacerda CA, Aguila MB (2012) Weight cycling enhances adipose tissue inflammatory responses in male mice. PLoS One 7:e39837
Benomar Y, Gertler A, De Lacy P, Crépin D, Hamouda HO, Riffault L et al (2013) Central resistin overexposure induces insulin resistance through Toll-like receptor 4. Diabetes 62:102–114
Bertolani C, Sancho-Bru P, Failli P, Bataller R, Aleffi S, DeFranco R et al (2006) Resistin as an intrahepatic cytokine: overexpression during chronic injury and induction of proinflammatory actions in hepatic stellate cells. Am J Pathol 169:2042–2053
Bielohuby M, Sisley S, Sandoval D, Herbach N, Zengin A, Fischereder M et al (2013) Impaired glucose tolerance in rats fed low-carbohydrate, high-fat diets. Am J Physiol Endocrinol Metab 305:E1059–E1070
Brunetti L, Orlando G, Recinella L, Michelotto B, Ferrante C, Vacca M (2004) Resistin, but not adiponectin, inhibits dopamine and norepinephrine release in the hypothalamus. Eur J Pharmacol 493:41–44
Castañeda T, Nogueiras R, Müller T, Krishna R, Grant E, Jones A et al (2011) Decreased glucose tolerance and plasma adiponectin: resistin ratio in a mouse model of post-traumatic stress disorder. Diabetologia 54:900–909
Chen Y-H, Lee M-J, Chang H-H, Hung P-F, Kao Y-H (2006) 17β-Estradiol stimulates resistin gene expression in 3T3-L1 adipocytes via the estrogen receptor, extracellularly regulated kinase, and CCAAT/enhancer binding protein-α pathways. Endocrinology 147:4496–4504
Cherneva RV, Georgiev OB, Petrova DS, Mondeshki TL, Ruseva SR, Cakova AD et al. (2013) Resistin-the link between adipose tissue dysfunction and insulin resistance in patients with obstructive sleep apnea. J Diabetes Metab Disord 12
Cho YM, Youn B-S, Chung SS, Kim KW, Lee H, Yu K-Y et al (2004) Common genetic polymorphisms in the promoter of resistin gene are major determinants of plasma resistin concentrations in humans. Diabetologia 47:559–565
Daquinag AC, Zhang Y, Amaya-Manzanares F, Simmons PJ, Kolonin MG (2011) An isoform of decorin is a resistin receptor on the surface of adipose progenitor cells. Cell Stem Cell 9:74–86
De Luis D, Terroba M, Cuellar L, Conde R, Primo D, Aller R et al (2011) Resistin levels in morbid obese patients following the biliopancreatic diversion surgery. Horm Metab Res 43:205–208
Felipe F, Bonet ML, Ribot J, Palou A (2004) Modulation of resistin expression by retinoic acid and vitamin A status. Diabetes 53:882–889
Friese MA, Montalban X, Willcox N, Bell JI, Martin R, Fugger L (2006) The value of animal models for drug development in multiple sclerosis. Brain 129:1940–1952
Gan A-M, Butoi ED, Manea A, Simion V, Stan D, Parvulescu M-M et al (2013) Inflammatory effects of resistin on human smooth muscle cells: up-regulation of fractalkine and its receptor, CX3CR1 expression by TLR4 and Gi-protein pathways. Cell Tissue Res 351:161–174
Gerber M, Boettner A, Seidel B, Lammert A, Bar J, Schuster E et al (2005) Serum resistin levels of obese and lean children and adolescents: biochemical analysis and clinical relevance. J Clin Endocrinol Metab 90:4503–4509
Ghosh S, Singh AK, Aruna B, Mukhopadhyay S, Ehtesham NZ (2003) The genomic organization of mouse resistin reveals major differences from the human resistin: functional implications. Gene 305:27–34
Graveleau C, Zaha VG, Mohajer A, Banerjee RR, Dudley-Rucker N, Steppan CM et al (2005) Mouse and human resistins impair glucose transport in primary mouse cardiomyocytes, and oligomerization is required for this biological action. J Biol Chem 280:31679–31685
Haugen F, Jørgensen A, Drevon CA, Trayhurn P (2001) Inhibition by insulin of resistin gene expression in 3T3-L1 adipocytes. FEBS Lett 507:105–108
Heilbronn L, Rood J, Janderova L, Albu J, Kelley D, Ravussin E et al (2004) Relationship between serum resistin concentrations and insulin resistance in non-obese, obese, and obese diabetic subjects. J Clin Endocrinol Metab 89:1844–1848
Higashida K, Fujimoto E, Higuchi M, Terada S (2013) Effects of alternate-day fasting on high-fat diet-induced insulin resistance in rat skeletal muscle. Life Sci 93:208–213
Hivert M-F, Sullivan LM, Fox CS, Nathan DM, D’Agostino RB Sr, Wilson PW et al (2008) Associations of adiponectin, resistin, and tumor necrosis factor-α with insulin resistance. J Clin Endocrinol Metab 93:3165–3172
Hsu WY, Chao YW, Tsai YL, Lien CC, Chang CF, Deng MC et al (2011) Resistin induces monocyte–endothelial cell adhesion by increasing ICAM-1 and VCAM-1 expression in endothelial cells via p38MAPK-dependent pathway. J Cell Physiol 226:2181–2188
Ikeda Y, Tsuchiya H, Hama S, Kajimoto K, Kogure K (2013) Resistin affects lipid metabolism during adipocyte maturation of 3T3-L1 cells. FEBS J 280:5884–5895
Ikeda Y, Tsuchiya H, Hama S, Kajimoto K, Kogure K (2014) Resistin regulates the expression of plasminogen activator inhibitor-1 in 3T3-L1 adipocytes. Biochem Biophys Res Commun 448:129–133
Jain SH, Massaro JM, Hoffmann U, Rosito GA, Vasan RS, Raji A et al (2009) Cross-sectional associations bet ween abdominal and thoracic adipose tissue compartments and adiponectin and resistin in the Framingham heart study. Diab Care 32:903–908
Johnson AM, Olefsky JM (2013) The origins and drivers of insulin resistance. Cell 152:673–684
Juan C-C, Au L-C, Fang VS, Kang S-F, Ko Y-H, Kuo S-F et al (2001) Suppressed gene expression of adipocyte resistin in an insulin-resistant rat model probably by elevated free fatty acids. Biochem Biophys Res Commun 289:1328–1333
Kahn BB, Flier JS (2000) Obesity and insulin resistance. J Clin Invest 106:473
Kalueff A, Wheaton M, Murphy D (2007) What’s wrong with my mouse model?: advances and strategies in animal modeling of anxiety and depression. Behav Brain Res 179:1–18
Kamigaki M, Sakaue S, Tsujino I, Ohira H, Ikeda D, Itoh N et al (2006) Oxidative stress provokes atherogenic changes in adipokine gene expression in 3T3-L1 adipocytes. Biochem Biophys Res Commun 339:624–632
Kaser S, Kaser A, Sandhofer A, Ebenbichler C, Tilg H, Patsch J (2003) Resistin messenger-RNA expression is increased by proinflammatory cytokines in vitro. Biochem Biophys Res Commun 309:286–290
Kim K-H, Lee K, Moon YS, Sul HS (2001) A cysteine-rich adipose tissue-specific secretory factor inhibits adipocyte differentiation. J Biol Chem 276:11252–11256
Kim K-H, Zhao L, Moon Y, Kang C, Sul HS (2004) Dominant inhibitory adipocyte-specific secretory factor (ADSF)/resistin enhances adipogenesis and improves insulin sensitivity. Proc Natl Acad Sci USA 101:6780–6785
Kusminski CM, Mcternan PG, Kumar S (2005) Role of resistin in obesity, insulin resistance and Type II diabetes. Clin Sci 109:243–256
Kwon H, Pessin JE (2013) Adipokines mediate inflammation and insulin resistance Front Endocrinol 4
Lazar M (2007) Resistin-and obesity-associated metabolic diseases. Horm Metab 39:710–716
Le Lay S, Boucher J, Rey A, Castan-Laurell I, Krief S, Ferré P et al (2001) Decreased resistin expression in mice with different sensitivities to a high-fat diet. Biochem Biophys Res Commun 289:564–567
Lee JH, Bullen JW, Stoyneva VL, Mantzoros CS (2005) Circulating resistin in lean, obese, and insulin-resistant mouse models: lack of association with insulinemia and glycemia. Am J Physiol Endocrinol Metab 288:E625–E632
Lee JH, Chan JL, Yiannakouris N, Kontogianni M, Estrada E, Seip R et al (2003) Circulating resistin levels are not associated with obesity or insulin resistance in humans and are not regulated by fasting or leptin administration: cross-sectional and interventional studies in normal, insulin-resistant, and diabetic subjects. J Clin Endocrinol Metab 88:4848–4856
Lee M-J, Lin H, Liu C-W, Wu M-H, Liao W-J, Chang H-H et al (2008) Octylphenol stimulates resistin gene expression in 3T3-L1 adipocytes via the estrogen receptor and extracellular signal-regulated kinase pathways. Am J Physiol Cell Physiol 294:C1542–C1551
Lehrke M, Reilly MP, Millington SC, Iqbal N, Rader DJ, Lazar MA (2004) An inflammatory cascade leading to hyperresistinemia in humans. PLoS Med 1:e45
Li Y, Ding L, Hassan W, Abdelkader D, Shang J (2013) Adipokines and hepatic insulin resistance. J Diab Res 2013
Liu C-W, Yang S-Y, Lin C-K, Liu H-S, Ho L-T, Wu L-Y et al (2014) The forkhead transcription factor FOXO1 stimulates the expression of the adipocyte resistin gene. Gen Comp Endocrinol 196:41–51
Lobo TF, Torloni MR, Gueuvoghlanian-Silva BY, Mattar R, Daher S (2013) Resistin concentration and gestational diabetes: a systematic review of the literature. J Reprod Immunol 97:120–127
Luo Z, Zhang Y, Li F, He J, Ding H, Yan L et al (2009) Resistin induces insulin resistance by both AMPK-dependent and AMPK-independent mechanisms in HepG2 cells. Endocrine 36:60–69
Manduteanu I, Pirvulescu M, Gan AM, Stan D, Simion V, Dragomir E et al (2010) Similar effects of resistin and high glucose on P-selectin and fractalkine expression and monocyte adhesion in human endothelial cells. Biochem Biophys Res Commun 391:1443–1448
Mantovani J, Roy R (2011) Re-evaluating the general (ized) roles of AMPK in cellular metabolism. FEBS Lett 585:967–972
Masuzaki H, Paterson J, Shinyama H, Morton NM, Mullins JJ, Seckl JR et al (2001) A transgenic model of visceral obesity and the metabolic syndrome. Science 294:2166–2170
McTernan CL, McTernan PG, Harte AL, Levick P, Barnett A, Kumar S (2002) Resistin, central obesity, and type 2 diabetes. Lancet 359:46–47
Menzaghi C, Coco A, Salvemini L, Thompson R, De Cosmo S, Doria A et al (2006) Heritability of serum resistin and its genetic correlation with insulin resistance-related features in nondiabetic Caucasians. J Clin Endocrinol Metab 91:2792–2795
Milan G, Granzotto M, Scarda A, Calcagno A, Pagano C, Federspil G et al (2002) Resistin and adiponectin expression in visceral fat of obese rats: effect of weight loss. Obes Res 10:1095–1103
Minn AH, Patterson NB, Pack S, Hoffmann SC, Gavrilova O, Vinson C et al (2003) Resistin is expressed in pancreatic islets. Biochem Biophys Res Commun 310:641–645
Monzillo LU, Hamdy O, Horton ES, Ledbury S, Mullooly C, Jarema C et al (2003) Effect of lifestyle modification on adipokine levels in obese subjects with insulin resistance. Obes Res 11:1048–1054
Morash BA, Willkinson D, Ur E, Wilkinson M (2002) Resistin expression and regulation in mouse pituitary. FEBS Lett 526:26–30
Moschen AR, Molnar C, Wolf AM, Weiss H, Graziadei I, Kaser S et al (2009) Effects of weight loss induced by bariatric surgery on hepatic adipocytokine expression. J Hepatol 51:765–777
Muse ED, Lam TK, Scherer PE, Rossetti L (2007) Hypothalamic resistin induces hepatic insulin resistance. J Clin Invest 117:1670
Muse ED, Obici S, Bhanot S, Monia BP, McKay RA, Rajala MW et al (2004) Role of resistin in diet-induced hepatic insulin resistance. J Clin Invest 114:232
Nagaev I, Smith U (2001) Insulin resistance and type 2 diabetes are not related to resistin expression in human fat cells or skeletal muscle. Biochem Biophys Res Commun 285:561–564
Nieva-Vazquez A, Pérez-Fuentes R, Torres-Rasgado E, López-López JG, Romero JR (2014) Serum resistin levels are associated with adiposity and insulin sensitivity in obese hispanic subjects. Metab Syndr Relat Disord 12:143–148
Niles LP, Lobb DK, Kang NH, Armstrong KJ (2012) Resistin expression in human granulosa cells. Endocrine 42:742–745
Nogueiras R, Gualillo O, Caminos JE, Garcı́ T, Casanueva FF, Diéguez C (2003) Resistin is expressed in different rat tissues and is regulated in a tissue-and gender-specific manner. FEBS Lett 548:21–27
Oliveira MC, Menezes-Garcia Z, Henriques MC, Soriani FM, Pinho V, Faria A et al (2013) Acute and sustained inflammation and metabolic dysfunction induced by high refined carbohydrate-containing diet in mice. Obesity 21:E396–E406
Oliver P, Ribot J, Rodriguez A, Sanchez J, Pico C, Palou A (2006) Resistin as a putative modulator of insulin action in the daily feeding/fasting rhythm. Pflügers Archiv 452:260–267
Onuma H, Tabara Y, Kawamura R, Ohashi J, Nishida W, Takata Y et al (2013) Plasma resistin is associated with single nucleotide polymorphisms of a possible resistin receptor, the decorin gene, in the general Japanese population. Diabetes 62:649–652
Ort T, Arjona AA, MacDougall JR, Nelson PJ, Rothenberg ME, Wu F et al (2005) Recombinant human FIZZ3/resistin stimulates lipolysis in cultured human adipocytes, mouse adipose explants, and normal mice. Endocrinology 146:2200–2209
Osawa H, Onuma H, Ochi M, Murakami A, Yamauchi J, Takasuka T et al (2005) Resistin SNP-420 determines its monocyte mRNA and serum levels inducing type 2 diabetes. Biochem Biophys Res Commun 335:596–602
Osawa H, Tabara Y, Kawamoto R, Ohashi J, Ochi M, Onuma H et al (2007) Plasma resistin, associated with single nucleotide polymorphism −420, is correlated with insulin resistance, lower HDL cholesterol, and high-sensitivity C-reactive protein in the Japanese general population. Diab Care 30:1501–1506
Owecki M, Miczke A, Nikisch E, Pupek-Musialik D, Sowiński J (2011) Serum resistin concentrations are higher in human obesity but independent from insulin resistance. Exp Clin Endocrinol Diab Off J Ger Soc Endocrinol Ger Diab Assoc 119:117–121
Pajvani UB, Hawkins M, Combs TP, Rajala MW, Doebber T, Berger JP et al (2004) Complex distribution, not absolute amount of adiponectin, correlates with thiazolidinedione-mediated improvement in insulin sensitivity. J Biol Chem 279:12152–12162
Palanivel R, Maida A, Liu Y, Sweeney G (2006) Regulation of insulin signalling, glucose uptake and metabolism in rat skeletal muscle cells upon prolonged exposure to resistin. Diabetologia 49:183–190
Park HK, Ahima RS (2013) Resistin in rodents and humans. Diab Metab J 37:404–414
Patel L, Buckels AC, Kinghorn IJ, Murdock PR, Holbrook JD, Plumpton C et al (2003) Resistin is expressed in human macrophages and directly regulated by PPARγ activators. Biochem Biophys Res Commun 300:472–476
Qasim AN, Metkus TS, Tadesse M, Lehrke M, Restine S, Wolfe ML et al (2009) Resistin gene variation is associated with systemic inflammation but not plasma adipokine levels, metabolic syndrome or coronary atherosclerosis in nondiabetic Caucasians. Clin Endocrinol 70:698–705
Qatanani M, Szwergold NR, Greaves DR, Ahima RS, Lazar MA (2009) Macrophage-derived human resistin exacerbates adipose tissue inflammation and insulin resistance in mice. J Clin Investig 119:531
Qiu W, Chen N, Zhang Q, Zhuo L, Wang X, Wang D et al (2014) Resistin increases platelet P-selectin levels via p38 MAPK signal pathway. Diab Vasc Dis Res 11:121–124
Rajala MW, Obici S, Scherer PE, Rossetti L (2003) Adipose-derived resistin and gut-derived resistin-like molecule–β selectively impair insulin action on glucose production. J Clin Invest 111:225
Reilly MP, Lehrke M, Wolfe ML, Rohatgi A, Lazar MA, Rader DJ (2005) Resistin is an inflammatory marker of atherosclerosis in humans. Circulation 111:932–939
Romacho T, Elsen M, Röhrborn D, Eckel J (2014) Adipose tissue and its role in organ crosstalk. Acta Physiol 210:733–753
Sanchez-Solana B, Laborda J, Baladron V (2011) Mouse resistin modulates adipogenesis and glucose uptake in 3T3-L1 preadipocytes through the ROR1 receptor. Mol Endocrinol 26:110–127
Satoh H, Nguyen MA, Miles PD, Imamura T, Usui I, Olefsky JM (2004) Adenovirus-mediated chronic “hyper-resistinemia” leads to in vivo insulin resistance in normal rats. J Clin Invest 114:224
Savage DB, Sewter CP, Klenk ES, Segal DG, Vidal-Puig A, Considine RV et al (2001) Resistin/Fizz3 expression in relation to obesity and peroxisome proliferator–activated receptor-γ action in humans. Diabetes 50:2199–2202
Schaalan M, El-Abhar HS, Barakat M, El-Denshary ES (2009) Westernized-like-diet-fed rats: effect on glucose homeostasis, lipid profile, and adipocyte hormones and their modulation by rosiglitazone and glimepiride. J Diab Complicat 23:199–208
Schwartz DR, Lazar MA (2011) Human resistin: found in translation from mouse to man. Trends Endocrinol Metab 22:259–265
Seo JB, Noh MJ, Yoo EJ, Park SY, Park J, Lee IK et al (2003) Functional characterization of the human resistin promoter with adipocyte determination-and differentiation-dependent factor 1/sterol regulatory element binding protein 1c and CCAAT enhancer binding protein-α. Mol Endocrinol 17:1522–1533
Sheng CH, Du ZW, Song Y, Wu XD, Zhang YC, Wu M, et al. (2013) Human resistin inhibits myogenic differentiation and induces insulin resistance in myocytes. BioMed Res Int 2013
Shetty GK, Economides PA, Horton ES, Mantzoros CS, Veves A (2004) Circulating adiponectin and resistin levels in relation to metabolic factors, inflammatory markers, and vascular reactivity in diabetic patients and subjects at risk for diabetes. Diab Care 27:2450–2457
Shulman GI, Rothman DL, Jue T, Stein P, DeFronzo RA, Shulman RG (1990) Quantitation of muscle glycogen synthesis in normal subjects and subjects with non-insulin-dependent diabetes by 13C nuclear magnetic resonance spectroscopy. N Engl J Med 322:223–228
Silswal N, Singh AK, Aruna B, Mukhopadhyay S, Ghosh S, Ehtesham NZ (2005) Human resistin stimulates the pro-inflammatory cytokines TNF-α and IL-12 in macrophages by NF-κB-dependent pathway. Biochem Biophys Res Commun 334:1092–1101
Singhal NS, Lazar MA, Ahima RS (2007) Central resistin induces hepatic insulin resistance via neuropeptide Y. J Neurosci 27:12924–12932
Song H, Shojima N, Sakoda H, Ogihara T, Fujishiro M, Katagiri H et al (2002) Resistin is regulated by C/EBPs, PPARs, and signal-transducing molecules. Biochem Biophys Res Commun 299:291–298
Song R, Wang X, Mao Y, Li H, Li Z, Xu W et al (2013) Resistin disrupts glycogen synthesis under high insulin and high glucose levels by down-regulating the hepatic levels of GSK3β. Gene 529:50–56
Steppan CM, Bailey ST, Bhat S, Brown EJ, Banerjee RR, Wright CM et al (2001) The hormone resistin links obesity to diabetes. Nature 409:307–312
Talmadge JE, Singh RK, Fidler IJ, Raz A (2007) Murine models to evaluate novel and conventional therapeutic strategies for cancer. Am J Pathol 170:793–804
Tang Y-C, Liu C-W, Chang H-H, Juan C-C, Kuo Y-C, Kao C-C et al (2014) Endothelin-1 stimulates resistin gene expression. Endocrinology 155:854–864
Tarkowski A, Bjersing J, Shestakov A, Bokarewa MI (2010) Resistin competes with lipopolysaccharide for binding to toll-like receptor 4. J Cell Mol Med 14:1419–1431
Tomaru T, Steger DJ, Lefterova MI, Schupp M, Lazar MA (2009) Adipocyte-specific expression of murine resistin is mediated by synergism between peroxisome proliferator-activated receptor γ and CCAAT/enhancer-binding proteins. J Biol Chem 284:6116–6125
Tontonoz P, Spiegelman BM (2008) Fat and beyond: the diverse biology of PPARγ. Annu Rev Biochem 77:289–312
Tovar S, Nogueiras R, Tung LY, Castañeda TR, Vázquez MJ, Morris A et al (2005) Central administration of resistin promotes short-term satiety in rats. Eur J Endocrinol 153:R1–R5
Ukkola O, Kunnari A, Kesäniemi YA (2008) Genetic variants at the resistin locus are associated with the plasma resistin concentration and cardiovascular risk factors. Regul Pept 149:56–59
Vazquez MJ, González CR, Varela L, Lage R, Tovar S, Sangiao-Alvarellos S et al (2008) Central resistin regulates hypothalamic and peripheral lipid metabolism in a nutritional-dependent fashion. Endocrinology 149:4534–4543
Way JM, Görgün CZ, Tong Q, Uysal KT, Brown KK, Harrington WW et al (2001) Adipose tissue resistin expression is severely suppressed in obesity and stimulated by peroxisome proliferator-activated receptor γ agonists. J Biol Chem 276:25651–25653
Wolfe BE, Jimerson DC, Orlova C, Mantzoros CS (2004) Effect of dieting on plasma leptin, soluble leptin receptor, adiponectin and resistin levels in healthy volunteers. Clin Endocrinol 61:332–338
Xu JY, Sham PC, Xu A, Tso AW, Wat N, Cheng KY et al (2007) Resistin gene polymorphisms and progression of glycaemia in southern Chinese: a 5-year prospective study. Clin Endocrinol 66:211–217
Yang R-Z, Huang Q, Xu A, McLenithan JC, Eison JA, Shuldiner AR et al (2003) Comparative studies of resistin expression and phylogenomics in human and mouse. Biochem Biophys Res Commun 310:927–935
Yannakoulia M, Yiannakouris N, Blüher S, Matalas A-L, Klimis-Zacas D, Mantzoros CS (2003) Body fat mass and macronutrient intake in relation to circulating soluble leptin receptor, free leptin index, adiponectin, and resistin concentrations in healthy humans. J Clin Endocrinol Metab 88:1730–1736
Youn B-S, Yu K-Y, Park HJ, Lee NS, Min SS, Youn MY et al (2004) Plasma resistin concentrations measured by enzyme-linked immunosorbent assay using a newly developed monoclonal antibody are elevated in individuals with type 2 diabetes mellitus. J Clin Endocrinol Metab 89:150–156
Zhang J, Qin Y, Zheng X, Qiu J, Gong L, Mao H et al (2002) The relationship between human serum resistin level and body fat content, plasma glucose as well as blood pressure. Zhonghua Yi Xue Za Zhi 82:1609–1612
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors state that they have no conflict of interest to declare.
Ethical approval
This article does not contain any studies with human participants or animal performed by any of the authors.
Informed consent
For this type of study formal consent is not required.
Rights and permissions
About this article
Cite this article
Huang, X., Yang, Z. Resistin’s, obesity and insulin resistance: the continuing disconnect between rodents and humans. J Endocrinol Invest 39, 607–615 (2016). https://doi.org/10.1007/s40618-015-0408-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-015-0408-2