Abstract
Objectives
Co-administration of posaconazole (PSZ) and vincristine (VCR) in the treatment of patients with acute lymphoblastic leukemia increases the neurotoxicity of VCR. Our aim is to study the effect of increased lipoprotein levels on the pharmacokinetics of PSZ and VCR upon co-administration in rats.
Methods
Rats were assigned to three groups, normolipidemic (NL), intermediate hyperlipidemic (IHL), and extreme hyperlipidemic (HL) groups. All rats were administered PSZ orally followed by VCR intravenously 4 h later. For the pharmacokinetic study, serial plasma samples were collected over 96 h and for tissue distribution study; plasma, lung, and liver tissues were collected over 48 h post oral dosing.
Results
Posaconazole showed higher plasma concentrations than VCR at all time points. Co-administration of VCR with PSZ reduced PSZ weight normalized oral clearance, increased PSZ area under the plasma concentration–time curve (AUC) from time zero to infinity, showed higher PSZ liver concentrations, and increased VCR volume of distribution of the central compartment. Upon increasing the lipoprotein levels, PSZ showed higher plasma availability and delayed tissue distribution, whereas VCR had shown a significant decrease in PSZ AUC0-24h, AUC0-tlast, and AUCo-inf (NL = IHL > HL) and a significant increase in the volume of distribution (NL = IHL < HL). Vincristine has shown higher tissue uptake and concentrations.
Conclusion
Monitoring cholesterol and triglyceride levels in patients with acute lymphoblastic leukemia is advisable to decrease VCR neurological side effect incidences and delay the activity of both PSZ and VCR.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Hyperlipidimia alone resulted in alterations of PSZ PK and delayed its onset of activity but had no effect on VCR PK when each drug was administered alone. |
Coadministration of both drugs in NL rats decreased PSZ Cl/f and increased its plasma and liver availability whereas, it increased VCR unbound fraction and tissue distribution. |
Coadministration of both drugs in HL rats further delayed PSZ tissue distribution and thus its assumed onset of action and decreased the plasma concentrations of VCR and increased its tissue uptake. |
It is important to monitor cholesterol and triglyceride levels in ALL patients to avoid aggrevated neurological side effects of VCR and the delay or lack of activity of PSZ. |
1 Introduction
Acute lymphoblastic leukemia (ALL) is a highly malignant disease characterized by immature lymphoid cells in the bone marrow, peripheral blood, and other organs [20, 36]. Acute lymphoblastic leukemia has a high incidence in USA where it is responsible for approximately 20,000 cancer diagnoses and 10,000 deaths annually [11, 36]. It has a median diagnosis age of 14 years [20]. Vincristine (VCR) is an antineoplastic agent that serves as one of the cornerstones in the induction, intensification, and consolidation of ALL treatment phases [39]. VCR is a key treatment for rhabdomyosarcoma, neuroblastoma, Wilms’ tumor, Hodgkin’s disease, and non-Hodgkin’s lymphomas [20, 29]. It acts through the inhibition of dividing cells at the metaphase stage, thus VCR may lead to neurological adverse effects [14, 19]. The dose and the duration of exposure to VCR greatly affect its clinical efficacy as well as its neurological toxicity [36]. VCR neurotoxicity is dose limiting, characterized by reversible symptoms such as paresthesia, sensory inadequacy, and seizures [14, 35]. Consequently, the safety and efficacy of VCR may be enormously affected by minor changes in its plasma concentration.
The reported clinical pharmacokinetic data for VCR exhibit a wide variability in the elimination phase half-life (t 1/2) (range 155–5100 min), volume of distribution (V d) (range 57–420 L/m2), and clearance (range 82–482 mL/min/m2) among tested patients [36]. VCR demonstrated linear low affinity binding to plasma proteins, mainly AAG, with a reported unbound fraction of 0.5 [9]. Its short distribution t 1/2, long elimination t 1/2, and large V d suggest a decreased tumor tissue exposure in vivo because of its high capability to bind to tissues [36]. In general, VCR has a considerable tendency to accumulate in most organs and tissues except in fat, eyes, and brain [36]. Additionally, VCR undergoes metabolism in liver by cytochrome P450 (CYP)3A (mainly CYP3A4 and CYP3A5) and is eliminated mainly in bile with approximately 8–15% excreted in urine [9, 14, 36]. In rats, VCR has demonstrated a similar pharmacokinetic pattern, exhibiting a biphasic plasma drug concentration decay with a rapid distribution phase t 1/2 (42–73 min) followed by a slower elimination phase t 1/2 of 14.3 ± 6.3 h [20]. VCR also presented a relatively large V d and a clearance of 0.12 ± 0.08 L/h/kg [20, 42].
Owing to their immunocompromised state and intensive chemotherapy, the ALL patient population is highly susceptible to invasive fungal infections. Triazole antifungals including fluconazole, voriconazole, itraconazole, and posaconazole (PSZ) are among the common measures taken for the prophylaxis and treatment of invasive fungal infections [18, 40]. Posaconazole is widely used as prophylactic therapy in hematopoietic stem cell transplant recipients with graft-vs.-host disease, or in individuals with hematological malignancies experiencing prolonged neutropenia as a result of intensive chemotherapy [3, 18, 22].
After oral administration in humans, PSZ is erratically absorbed and follows linear pharmacokinetics. Posaconazole has a large V d (1774 L), and is considerably bound to plasma proteins with a proportion of 98% to albumin [18, 22, 25]. Posaconazole has a median t 1/2 between 15 and 35 h [7]. The time reported to reach maximum plasma concentration is between 6 and 8 h [33]. Posaconazole is excreted unchanged in feces, with only 16% undergoing glucuronidation through UGT1A4 and recovered in urine [10, 18]. It was also reported that PSZ undergoes enterohepatic circulation, presenting a second peak after 24 h [24, 33]. As a CYP3A4 inhibitor, PSZ has higher potential for drug–drug interactions with CYP3A4 substrates [18, 23, 25]. In rats, PSZ also follows linear pharmacokinetics, its absorption is slow with the maximum plasma concentration reached within 3–12 h in fed rats and an oral bioavailability of 6.7% [22]. Posaconazole is highly protein bound (97–99%) and demonstrates low clearance values between 0.103 and 0.207 L/h/kg. Enterohepatic recirculation was also reported with the use of PSZ, showing a second peak in rats and mice; however, this was not the case in dogs [22, 32].
Hyperlipidemia is a chronic disease characterized by high plasma lipoprotein levels [5, 16, 41]. The risk factors of hyperlipidemia include genetic makeup defects, unhealthy diet, sedentary lifestyle habits, and certain drugs or diseases [15]. The incidence of hyperlipidemia had remarkably increased in the last few years as a result of the increase in diabetes mellitus and obesity worldwide [15, 41]. Hyperlipidemia is accompanied by a consistent increase in low-density lipoprotein that is a key contributor to an increased risk of atherosclerosis, hypertension, and ischemic heart disease [17, 30]. Furthermore, hyperlipidemia was shown to result in alterations in the pharmacokinetic and pharmacodynamics pattern of some drugs [1, 16, 22, 34, 37]. This may be attributed to reduced unbound fraction, enhanced lipoprotein receptor-mediated drug tissue uptake, and/or altered metabolizing enzyme expression [15, 17, 22, 34]. Recently, patients who have undergone allogenic hematopoietic stem cell transplantation have shown a high incidence of hyperlipidemia [2]. In addition, acute and severe hypertriglyceridemia (>10,000 mg/L of the plasma triglyceride level) was shown to be associated with VCR/L-asparaginase treatment of patients with ALL and malignant lymphoma [20, 38].
Recent reports have demonstrated that hyperlipidemia resulted in significant changes in PSZ pharmacokinetics and tissue distribution in rodents with hyperlipidemia [8, 22]. However, it resulted in insignificant alterations in VCR pharmacokinetics and protein binding in rats [20]. Thus, hyperlipidemia alone may not pose a threat to the VCR clinical efficacy and toxicity [20]. Recent publications have demonstrated that co-administration of azoles including PSZ with VCR led to an increased VCR toxicity [12, 14, 27, 31]. Ketoconazole is another azole antifungal whose pharmacokinetic parameters were not affected by hyperlipidemia in rats. However, its liver uptake was altered and the severity of its drug interaction with midazolam was increased [16]. Whether hyperlipidemia could potentiate or reduce the severity of the drug interaction between PSZ and VCR is still unknown. Therefore, the aim of the current study is to test the impact of different levels of plasma lipoproteins on the disposition of PSZ and VCR regarding their plasma levels, protein binding, and tissue distribution in a polymer-induced hyperlipidemic rat model.
2 Methodology
2.1 Materials and Reagents
Vincristine and posaconazole powders (purity >99%) were purchased from Selleckchem (Houston, TX, USA). Itraconazole was a kind gift from Nifty Labs Pvt Ltd (Hyderabad, India). High-performance liquid chromatography grade methanol and acetonitrile (Fisher Scientific UK Limited, Loughborough, UK), analytical grade potassium dihydrogen orthophosphate (Riedel-de-Haën, Germany), and high-purity distilled water were used. Vinracine® intravenous injection vials labeled to contain 1 mg/mL of VCR sulfate (EIMC United Pharmaceuticals, Cairo, Egypt), Aerraine® isoflurane USP, normal saline, and heparin sodium for injection 5000 U/mL were purchased from the Egyptian local market. Noxafil® oral suspension labeled to contain 40 mg/mL of PSZ (Pathe 103 on Inc., ON, Canada) was purchased from Schering-Plough S.A., Alexandria, Egypt.
2.2 Animals and Pre-Experimental Procedures
Experimental protocols involving animals were approved by the Ethics Committee of the Faculty of Pharmacy, Alexandria University. Sprague–Dawley rats were used in the studies. All rats weighed between 150 and 250 g and were housed in temperature-controlled rooms with 12 h of light per day. The animals were fed a standard commercial rat chow containing 4.5% fat (Tanta Oil and Soap JSC, Tanta, Egypt). Free access to food and water was permitted prior to experimentation. Rats were allocated into normolipidemic (NL), intermediate hyperlipidemic (IHL), or extreme hyperlipidemic (HL) groups. Hyperlipidemia was induced by a single intraperitoneal injection of 1 g/kg of P407 (0.13 g/mL solution in normal saline). To ensure the proper injection of P407, the animals were lightly anesthetized using isoflurane, and then allowed to recover. The rats were prepared for intravenous dosing by jugular vein cannulation. For each rat, a single jugular vein was catheterized with Silastic Laboratory Tubing (Dow Corning Corporation, Midland, MI, USA) under isoflurane/O2 anesthesia. The cannula was filled with locking solution containing 100 U/mL of heparin in 0.9% saline. After implantation, the rats were transferred to their regular holding cages and allowed free access to water, but food was withheld overnight so that the drug would be administered in the fasted state. The next morning, NL rats were transferred to the metabolic cages and dosed with PSZ orally; 4 h later, the rats were given VCR intravenously via the cannulated jugular vein. As for the HL and IHL groups, the dosing was started 36 and 72 h after the P407 injection, respectively [6, 22].
2.3 Drug Administration and Sample Collection
For the pharmacokinetic study, the rat groups (n = 6–10 each) received PSZ 40 mg/kg by oral gavage. Noxafil® oral suspension (40 mg/mL) was used for the administration of PSZ. Four hours later, the rat groups received 0.15 mg/kg VCR intravenously via the jugular vein cannula, immediately followed by an injection of normal saline. The intravenous solution was prepared by diluting the Vinracine® (1 mg/mL) injection vials with normal saline to reach a final concentration of 0.1 mg/mL. At the time of the first sample withdrawal post-intravenous dosing, the first 0.2 mL of blood was discarded. Food was provided to animals 5 h after the dose administration.
Serial blood samples (0.15–0.25 mL) were collected using the jugular vein cannula at 0.5, 1, 1.5, 2, 4.083, 4.25, 4.5, 5, 6, 8, 10, 24, 48, 72, and 96 h after the PSZ oral dose for each group. Plasma was separated by centrifugation of the blood at 4000g for 5 min and transferred into clean glass test tubes. The samples were kept at −20 °C until assayed.
2.4 Protein-Binding Study
The unbound fractions of PSZ and VCR in plasma were determined using ultrafiltration (Centrifree®; Amicon, Beverly, MA, USA). Blood was obtained from NL, IHL, and HL rats by exsanguination via cardiac puncture. For IHL and HL rats, blood was collected 72 and 36 h after intraperitoneal doses of P407, respectively. The blood was centrifuged at 2500g for 10 min. The NL, IHL, and HL rat plasma were spiked with PSZ and VCR methanolic solution to allow for final concentrations of 3.5 and 1 mg/L, respectively. The volume of methanol added to each tube did not exceed 0.05% (v/v). Tubes were incubated for 1 h in a 37 °C water bath shaker [15, 20]. One milliliter of each tube was transferred to an ultrafiltration device (n = 4) (Centrifree®; Millipore, Carrigtwohill, Ireland), which was then placed in a fixed-angle centrifuge rotor and spun at 2000g for 10 min at 37 °C. The samples were then analyzed for PSZ and VCR concentrations. In HL and IHL protein-binding tests, the ultrafiltration devices were pre-incubated with 5% Triton® (X-100) for 12 h to overcome the binding of drugs and lipids to the filter. The devices were then used after being rinsed several times with double-distilled water [16, 22].
2.5 Assay
A validated high-performance liquid chromatography assay was used for the measurement of PSZ and VCR concentrations, with itraconazole as an internal standard in plasma, Sorenson sodium phosphate buffer (pH 7.4), liver tissue and lung tissues [21]. The validated lower limit of quantitation was 50 ng/mL based on 0.2 mL of plasma, tissue homogenate, or buffer. For standard curve construction, drug-free plasma, tissue homogenate, and Sorenson sodium phosphate buffer (pH 7.4) were used and spiked with appropriate amounts of PSZ and VCR. The calibration curves relating the peak area ratio to the expected concentration were highly linear from 50 to 5000 ng/mL of PSZ and VCR in NL, IHL, HL plasma, tissue homogenate and buffer (r 2 > 0.997).
2.6 Data and Statistical Analysis
Non-compartmental methods were applied to calculate the pharmacokinetic parameters. The elimination rate constant (λz) was calculated by subjecting the plasma concentrations in the terminal phase to linear regression analysis. The terminal phase t½ was calculated using the equation t½ = 0.693/λz. The area under the plasma concentration–time curve from time zero to infinity (AUC0–∞) was calculated using the combined log-linear trapezoidal rule from time zero post-dose to the time of the last measured concentration, plus the quotient of the last measured concentration divided by λz. The concentration at time zero after intravenous dosing was estimated by back extrapolation of the log-linear regression line using the first three measured plasma concentrations. The clearance was calculated as the quotient of dose to AUC0–∞ and the V d as the quotient of clearance by λz. The V d of the central compartment is calculated as the quotient of dose by the concentration at time zero. The PSZ maximum plasma concentration and the time at which it occurred were determined by visual examination of the data.
The plasma-unbound fraction was determined by dividing the PSZ or VCR concentration in the filtrate by that measured in the pre-filtered plasma. All compiled data were reported as mean ± standard deviation, unless otherwise indicated. Significance of comparisons of means for the interactions of lipoprotein status, VCR pharmacokinetics, PSZ pharmacokinetics, and protein binding were assessed by two-way analysis of variance (ANOVA), followed by Tukey’s multiple comparison post-hoc test (Prism, Irvine, CA, USA). In all cases, the level of significance was set at p = 0.05.
3 Results
3.1 Pharmacokinetics and Protein Binding
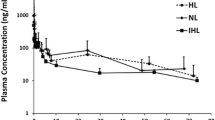
Upon co-administration of 40 mg/kg of PSZ and 0.15 mg/kg of VCR in NL rats, PSZ showed higher plasma concentrations than VCR at all timepoints (Fig. 1). Both PSZ and VCR showed few pharmacokinetic alterations compared with when solely administered [20, 22]. However, PSZ showed a significantly lower unbound fraction (fu = 0.011 ± 0.007), oral clearance (CL/f), oral volume of distribution (Vd/f), and t 1/2 values (Table 1). It also exhibited statistically higher C max2 and AUC0–∞ (Table 1; Fig. 1) than those previously reported for PSZ when administered alone in NL rats [22]. Co-administered VCR demonstrated a twofold higher unbound fraction (fu = 0.336 ± 0.022) than that previously reported for VCR when administered alone in NL rats. This was accompanied by a confirmatory trend of decreasing Co by half [20]. However, the decrease in Co was not statistically significant owing to the high variability between the rats. Moreover, the VCR Vc increased significantly with an increasing trend in the t 1/2 (Table 2).
Elevation of the lipoprotein levels in plasma resulted in some alterations in the pharmacokinetic profile of the co-administered PSZ and VCR (Tables 1, 2; Figs. 2, 3). For PSZ, increasing the plasma lipoprotein levels demonstrated a significant increase in C max1 (NL < IHL = HL) and AUC0–24h (NL = IHL < HL) similar to what was previously reported for PSZ administered alone in such rats [22]. The PSZ unbound fraction in IHL and HL rats could not be measured because it decreased to a level below our analytical assay lower limit of quantification, especially with the presence of an interfering background peak that resulted in false higher concentrations in such groups [21]. The remainder of the PSZ pharmacokinetic parameters including CL/f, V d/f, and t 1/2 did not show any change with the elevation of the plasma lipoprotein levels.
For VCR, increasing the plasma lipoproteins levels was previously reported not to alter VCR pharmacokinetic parameters when administered alone [20]. However, upon co-administration with PSZ, HL rats showed a significant decrease (~71%) in AUC0–24h and AUC0–∞, accompanied by a significant increase in V d/f/kg compared with NL rats (Table 2; Fig. 3). The clearance showed an increasing trend though it was not significant.
3.2 Tissue Distribution
Normolipidemic rats co-administered PSZ with VCR showed significantly higher PSZ liver AUC0–48h than those administered PSZ alone (Table 3). However, PSZ lung AUC0–48h was not significantly different between the two rat groups (Table 3) [22]. Posaconazole liver and lung Kp (rate of tissue uptake calculated by dividing the drug tissue concentrations by drug plasma concentrations) values were also similar between both groups (Fig. 4) [22]. Hyperlipidemic rats co-administered PSZ and VCR showed significantly lower PSZ uptake for the first 8 h with a delayed appearance of C max (~24 h) in liver and lung tissues (Figs. 4, 5). However, PSZ liver and lung AUC0–48h were significantly higher in HL rat groups (Table 3; Fig. 5).
Mean ± standard deviation liver or lung to plasma ratios of posaconazole (PSZ) vs. time curves in normolipidemic (NL) and hyperlipidemic (HL) rats after 40 mg/kg of oral PSZ, which was co-administered with 0.15 mg/kg of intravenous vincristine 4 h later (n = 4 rats at each timepoint). Asterisk indicates statistical significance, p < 0.05
Co-administered VCR showed a significantly higher tissue, liver, and lung uptake in HL rats along all tested points (Fig. 6). VCR liver and lung AUC0–48h were significantly higher in HL rats (Table 4; Fig. 7). The C max for tissue was achieved relatively faster than that of PSZ (Figs. 5, 7).
Mean ± standard deviation liver/lung to plasma ratios of vincristine (VCR) vs. time curves in normolipidemic (NL) and hyperlipidemic (HL) rats after 40 mg/kg of oral posaconazole, which was co-administered with 0.15 mg/kg of intravenous VCR 4 h later (n = 4 rats at each timepoint). Asterisk indicates statistical significance, p < 0.05
4 Discussion
The study demonstrates that hyperlipidemia augments the drug interaction between PSZ and VCR. It results in a delayed PSZ tissue penetration. However, it decreases VCR plasma availability and increases its tissue penetration. This finding raises the possibility that patients with ALL with elevated lipoprotein levels co-administered both drugs are prone to a decreased VCR efficacy accompanied by more side effects, in addition to lower PSZ antifungal activity against invasive fungal infections.
Posaconazole co-administration with VCR was reported to increase the latter side effects, mainly peripheral neuropathy. A few case reports have also demonstrated VCR central toxicity, leading to seizures in ALL patients [14, 26, 28]. This was generally interpreted by the PSZ inhibition of CYP3A4, thus decreasing VCR clearance and increasing the latter side effects [14]. In our current model and study design, we suggest other resolutions as well. To study the effect of hyperlipidemia using our rat model, we gave the PSZ as a single dose followed by VCR 4 h later after PSZ reaches its C max. Thus, CYP3A4 inhibition may not be fully attained by PSZ. Instead, it is proven that both PSZ and VCR are excreted through the bile and undergo enterohepatic recirculation [20, 22]. This was clearly shown in this study also, where the two drugs demonstrated two peaks on the plasma concentration–time curve of each drug upon co-administration (Fig. 1). Moreover, our results show that co-administration of both drugs in NL rats leads to significantly higher PSZ liver concentrations and AUC0–48 than in the NL rats administered PSZ alone (Table 3) [22]. This was not the case for PSZ lung concentrations, indicating potential competition with VCR on bile elimination rather than an effect on tissue uptake. This was accompanied by a significant decrease in PSZ CL/f and an increase in AUC0–∞ as well as C max2 (Table 1) in NL rats co-administered both drugs compared with those administered PSZ alone [22]. On the other hand, VCR has shown an increased unbound fraction and higher V c and a trend of a prolonged half-life in co-administered NL rat groups [20].
Posaconazole prophylactic therapy against invasive Aspergillus and Candida infections in patients with ALL has shown mortality benefits as well as better outcomes towards invasive fungal infections refractory to itraconazole therapy [18]. Posaconazole has greater and faster lung penetration than other azole antifungal treatments [13]. It was previously proven that hyperlipidemia altered PSZ liver and lung distribution, resulting in a delayed onset and decreased magnitude of its activity and/or drug-interaction pattern [22]. In the current study, hyperlipidemia affected co-administered PSZ in the same manner, resulting in a greater delay in its onset of action and/or interaction in the lung and/or liver. However, co-administered PSZ demonstrated higher lung and liver concentrations in HL rats than in NL rats and higher uptake after 24 h, which was accompanied by lower plasma C max2. The delayed onset of action of PSZ in the lung as well as the higher tissue concentrations of PSZ in HL rats upon co-administration with VCR could be attributed to the combined effect of the competitive inhibition between both drugs on efflux and influx proteins. In addition to the effect of hyperlipidemia reported previously to downregulate the expression and/or the functional activity of (MDR1) P-glycoprotein and other efflux proteins [4, 22]. In fact, hyperlipidemia alone and VCR co-administration alone did not alter the PSZ AUC0–48h in lung tissues. Whereas, the AUC0–48h of PSZ in lungs of HL rats that were co-administered VCR increased ~2.5 fold (Table 3). Looking at the liver tissues, co-administration with VCR increased the PSZ AUC0–48h by 1.9-fold in NL rats. Whereas, hyperlipidemia alone showed a trend of increasing PSZ AUC0–48h that was not statistically significant. Upon combining both factors, the AUC0–48h value increased 2.5-fold (Table 3). This necessitates further investigation into the efflux and influx proteins responsible for PSZ and VCR in the future to determine the effect of hyperlipidemia on these proteins and to describe in detail the molecular mechanisms involved in such drug interactions.
Although no effect was detected previously for the increased lipoprotein levels on VCR pharmacokinetics and protein binding, HL rats co-administered VCR and PSZ showed significantly lower VCR AUC0–24h and AUC0–∞ accompanied by a significant increase in V d/f/kg compared with NL rats. This was not attributed to the initial dose for such group where the concentration at time zero values among the different lipidemic groups were consistent. In addition, VCR liver and lung uptake and concentrations were consistently higher in HL rats than NL rats. Thus, HL accompanied by PSZ co-administration results in decreased VCR plasma availability and increased tissue distribution. This may result in lower efficacy for anticancer drugs and increased side effects.
5 Conclusion
Hyperlipidemia in rat resulted in alterations in PK of PSZ and delayed onset of activity and decreased tissue uptake. Whereas, HL by itself did not alter the PK of VCR. Upon coadministration, PSZ showed significantly lower Cl/F and significantly higher AUC and liver concentrations but unchanged lung concentrations. Whereas, VCR showed larger unbound fraction and volume of distribution. Coadministration associated with elevation in plasma lipoproteins resulted in further delay in PSZ tissue distribution however, a total higher liver and lung AUC0-48. On the other hand, it resulted in decreased VCR plasma availability and increased its tissue uptake and concentration. Thus as a conclusion, monitoring the cholesterol and triglyceride levels in ALL patients is advisable to avoid the aggravated neurological side effects of VCR and the lack or delay of activity of both PSZ and VCR.
References
Bin Jardan YA, Brocks DR. The pharmacokinetics of dronedarone in normolipidemic and hyperlipidemic rats. Biopharm Drug Dispos. 2016;37(6):345–51.
Blaser BW, Kim HT, Alyea EP, et al. Hyperlipidemia and statin use after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transpl. 2012;8:575–83.
Bourdelin M, Grenouillet F, Daguindau E, et al. Posaconazole prophylaxis in neutropenic patients with hematological malignancies: limits in clinical practice. Med Mycol. 2014;52:728–35.
Brocks DR, Chaudhary HR, Ben-Eltriki M, et al. Effects of serum lipoproteins on cyclosporine A cellular uptake and renal toxicity in vitro. Can J Physiol Pharmacol. 2014;92:140–8.
Brown MS, Goldstein, JL. Drugs used in the treatment of hyperliporoteinemias. In: Gilman AG, Goodman LS, Gilman A. The pharmacological basis of therapeutics. 8th ed. New York: McGraw-Hill Medical; 1990. pp. 874–96.
Chaudhary HR, Brocks DR. The single dose poloxamer 407 model of hyperlipidemia; systemic effects on lipids assessed using pharmacokinetic methods, and its effects on adipokines. J Pharm Pharm Sci. 2013;16:65–73.
Courtney R, Pai S, Laughlin M, et al. Pharmacokinetics, safety, and tolerability of oral posaconazole administered in single and multiple doses in healthy adults. Antimicrob Agents Chemother. 2003;47:2788–95.
Courtney R, Wexler D, Radwanski E, et al. Effect of food on the relative bioavailability of two oral formulations of posaconazole in healthy adults. Br J Clin Pharmacol. 2004;57:218–22.
Dennison JB, Jones DR, Renbarger JL, Hall SD. Effect of CYP3A5 expression on vincristine metabolism with human liver microsomes. J Pharmacol Exp Ther. 2007;321:553–63.
Dodds Ashley ES, Varkey JB, Krishna G, et al. Pharmacokinetics of posaconazole administered orally or by nasogastric tube in healthy volunteers. Antimicrob Agents Chemother. 2009;53:2960–4.
Dores GM, Devesa SS, Curtis RE, et al. Acute leukemia incidence and patient survival among children and adults in the United States, 2001–2007. Blood. 2012;119:34–43.
Eiden C, Palenzuela G, Hillaire-buys D, et al. Posaconazole-increased vincristine neurotoxicity in a child: a case report. J Pediatr Hematol/Oncol. 2009;31:292–5.
Felton T, Troke PF, Hope WW. Tissue penetration of antifungal agents. Clin Microbiol Rev. 2014;27:68–88.
Hamdy DA, El-Geed H, El-Salem S, Zaidan M. Posaconazole–vincristine coadministration triggers seizure in a young female adult: a case report. Case Rep Hematol. 2012;2012:343742.
Hamdy DA, Brocks DR. Experimental hyperlipidemia causes an increase in the electrocardiographic changes associated with amiodarone. J Cardiovasc Pharmacol. 2009;53:1–8.
Hamdy DA, Brocks DR. Effect of hyperlipidemia on ketoconazole–midazolam drug–drug interaction in rat. J Pharm Sci. 2011;100:4986–92.
Hamdy DA, Brocks DR. The effect of increased lipoprotein levels on the pharmacokinetics of ketoconazole enantiomers in the rat. Xenobiotica. 2011;41:137–43.
Hamdy DA, El-Salem S, El-Geed H, Zaidan M. Posaconazole, a prophylactic therapy in patients with haematological cancer: drug use evaluation study. Eur J Hosp Pharm. 2013;20:223–6.
Himes RH, Kersey RN, Heller-Bettinger I, Samson FE. Action of the vinca alkaloids vincristine, vinblastine, and desacetyl vinblastine amide on microtubules in vitro. Cancer Res. 1976;36(10):3798–802.
Khalil HA, Belal TS, El-Yazbi AF, Hamdy DA. The effect of increased lipoproteins levels on the disposition of vincristine in rat. Lipids Health Dis. 2016;15:152.
Khalil HA, El-Yazbi AF, Belal TS, Hamdy DA. High performance liquid chromatographic assay for the simultaneous determination of posaconazole and vincristine in rat plasma. Int J Anal Chem. 2015;2015:743915.
Khalil HA, Elnaggar MM, Belal TS, et al. The effect of hyperlipidemia on the pharmacokinetics, hepatic and pulmonary uptake of posaconazole in rat. Eur J Pharm Sci. 2016;91:190–5.
Krieter P, Flannery B, Musick T, et al. Disposition of posaconazole following single-dose oral administration in healthy subjects. Antimicrob Agents Chemother. 2004;48:3543–51.
Li Y, Theuretzbacher U, Clancy CJ, et al. Pharmacokinetic/pharmacodynamic profile of posaconazole. Clin Pharmacokinet. 2010;49:379–96.
Lipp HP. Posaconazole: clinical pharmacokinetics and drug interactions. Mycoses. 2011;54(Suppl. 1):32–8.
Mahapatra M, Kumar R, Choudhry VP. Seizures as an adverse drug reaction after therapeutic dose of vincristine. Ann Hematol. 2007;86:153–4.
Mantadakis E, Amoiridis G, Kondi A, Kalmanti M. Possible increase of the neurotoxicity of vincristine by the concurrent use of posaconazole in a young adult with leukemia. J Pediatr Hematol/Oncol. 2007;29:130.
Mantadakis E, Amoiridis G, Kondi A, Kalmanti M. Possible increase of the neurotoxicity of vincristine by the concurrent use of posaconazole in a young adult with leukemia. J Pediatr Hematol Oncol. 2007;29:130.
Martin J, Compston N. Vincristine sulphate in the treatment of lymphoma and leukaemia. Lancet. 1963;282:1080–3.
Mendis S, Puska P, Norrving B. Global atlas on cardiovascular disease prevention and control. Geneva: World Health Organization; 2011.
Moriyama B, Henning SA, Leung J, et al. Adverse interactions between antifungal azoles and vincristine: review and analysis of cases. Mycoses. 2012;55:290–7.
Nomeir AA, Kumari P, Hilbert MJ, et al. Pharmacokinetics of SCH 56592, a new azole broad-spectrum antifungal agent, in mice, rats, rabbits, dogs, and cynomolgus monkeys. Antimicrob Agents Chemother. 2000;44:727–31.
Padmavathy B, Devaraj SN, Devaraj H. Effect of amiodarone, an antiarrhythmic drug, on serum and liver lipids and serum marker enzymes in rats. Indian J Biochem Biophys. 1992;29:522–4.
Patel JP, Hamdy DA, El-kadi AO, Brocks DR. Effect of serum lipoproteins on stereoselective halofantrine metabolism by rat hepatocytes. Chirality. 2012;24:558–65.
Rosenthal S, Kaufman S. Vincristine neurotoxicity. Ann Intern Med. 1974;80:733–7.
Said R, Tsimberidou AM. Pharmacokinetic evaluation of vincristine for the treatment of lymphoid malignancies. Expert Opin Drug Metab Toxicol. 2014;10:483–94.
Shayeganpour A, Jun AS, Brocks DR. Pharmacokinetics of amiodarone in hyperlipidemic and simulated high fat-meal rat models. Biopharm Drug Dispos. 2005;26:249–57.
Tozuka M, Yamauchi K, Hidaka H, et al. Characterization of hypertriglyceridemia induced by l-asparaginase therapy for acute lymphoblastic leukemia and malignant lymphoma. Ann Clin Lab Sci. 1997;27:351–7.
van Hasselt JG, van Eijkelenburg NK, Beijnen JH, et al. Design of a drug–drug interaction study of vincristine with azole antifungals in pediatric cancer patients using clinical trial simulation. Pediatr Blood Cancer. 2014;61:2223–9.
van Schie RM, Bruggemann RJ, Hoogerbrugge PM, Te Loo DM. Effect of azole antifungal therapy on vincristine toxicity in childhood acute lymphoblastic leukaemia. J Antimicrob Chemother. 2011;66:1853–6.
Wasan KM, Looije NA. Emerging pharmacological approaches to the treatment of obesity. J Pharm Pharm Sci. 2005;8:259–71.
Zhou XJ, Martin M, Placidi M, et al. In vivo and in vitro pharmacokinetics and metabolism of vincaalkaloids in rat. II. Vinblastine and vincristine. Eur J Drug Metab Pharmacokinet. 1990;15(4):323–32.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was financially supported by the Science and Technology Development Fund in Egypt (Grant No. 5575).
Conflict of interest
Hadeel A. Khalil, Mohammed A.W. ElKhatib, Tarek S. Belal, Ahmed F. El-Yazbi, and Dalia A. Hamdy report no conflicts of interest directly relevant to the content of this study.
Ethics approval
Experimental protocols involving animals were approved by the Ethics Committee of the Faculty of Pharmacy, Alexandria University.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Khalil, H.A., ElKhatib, M.A.W., Belal, T.S. et al. Hyperlipidemia Alters the Pharmacokinetics of Posaconazole and Vincristine Upon Co-Administration in Rats. Drugs R D 17, 287–296 (2017). https://doi.org/10.1007/s40268-017-0178-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40268-017-0178-8