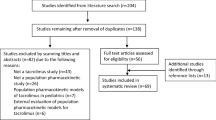
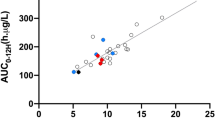
Abstract
This review summarises the available data on the population pharmacokinetics of tacrolimus and use of Maximum A Posteriori (MAP) Bayesian estimation to predict tacrolimus exposure and subsequent drug dosage requirements in solid organ transplant recipients. A literature search was conducted which identified 56 studies that assessed the population pharmacokinetics of tacrolimus based on non-linear mixed effects modelling and 14 studies that assessed the predictive performance of MAP Bayesian estimation of tacrolimus area under the plasma concentration–time curve (AUC) from time zero to the end of the dosing interval. Studies were most commonly undertaken in adult kidney transplant recipients and investigated the immediate-release formulation. The pharmacokinetics of tacrolimus were described using one- and two-compartment disposition models with first-order elimination in 61 and 39 % of population pharmacokinetic studies, respectively. Variability in tacrolimus whole blood apparent clearance amongst transplant recipients was most commonly related to cytochrome P450 (CYP) 3A5 genotype (rs776746), patient haematocrit, patient weight, post-operative day and hepatic function (aspartate aminotransferase). Bias, as calculated using estimation of the mean predictive error (MPE) or mean percentage predictive error (MPPE) associated with prediction of the tacrolimus AUC, ranged from −15 to 9.95 %. Imprecision, as calculated through estimation of the root mean squared error (RMSE) or mean absolute prediction error (MAPE), was generally much poorer overall, ranging from 0.81 to 40. r 2 values ranged from 0.27 to 0.99 %. Of the Bayesian forecasting strategies that used two or more tacrolimus concentrations, 71 % showed bias of 10 % or less; however, only 39 % showed imprecision of 10 % or less. The combination of sampling times at 0, 1 and 3 h post-dose consistently showed bias and imprecision values of less than 15 %. No studies to date have examined how closely MAP Bayesian dosage predictions of tacrolimus actually achieve target AUC by comparing dosage prediction from one occasion with a future measured AUC. Further research involving larger prospective studies including more diverse transplant groups and the extended-release formulation of tacrolimus is needed. Several questions require further examination, including the following. Do Bayesian forecasting methods currently use the most appropriate population pharmacokinetic models and optimal sampling times for dosage prediction? Does Bayesian forecasting perform well when applied to make dosage predictions on a subsequent occasion? How can Bayesian forecasting be simplified for use in the clinical setting? And, are patient outcomes improved with dosage prediction based on Bayesian forecasting compared with trough concentration monitoring?
Similar content being viewed by others
References
Staatz C, Tett SE. Clinical pharmacokinetics of once-daily tacrolimus in solid-organ transplant patients. Clin Pharmacokinet. 2015;54(10):993–1025.
Hesselink D, van Gelder T, van Schaik R. The pharmacogenetics of calcineurin inhibitors: one step closer toward individualised immunosuppression. Pharmacogenomics. 2005;6(4):323–37.
Staatz C, Tett S. Clinical pharmacokinetics and pharmacodynamis of tacrolimus in solid organ transplantation. Clin Pharmacokinet. 2004;43(10):623–53.
Shuker N, van Gelder T, Hesselink A. Intra-patient variability in tacrolimus exposure: causes, consequences for clinical management. Transplant Rev. 2015;29(2):78–84.
Scott L, McKeage K, Keam S, et al. Tacrolimus: a further update of its use in the management of organ transplantation. Drugs. 2000;63(12):1247–97.
de Jonge H, Kuypers DR, Verbeke K, Vanrenterghem Y. Reduced C 0 concentrations and increased dose requirements in renal allograft recipients converted to the novel once-daily tacrolimus formulation. Transplantation. 2010;90(5):523–9.
Wallemacq P, Armstrong V, Brrunet M, et al. Opportunities to optimise tacrolimus therapy in solid organ transplantation: report of the European Consensus Conference. Ther Drug Monit. 2009;31(2):139–52.
Laskow D, Vincenti F, Neylan J, et al. An open-label, concentration-ranging trial of FK506 in primary kidney transplantation. Transplantation. 1996;62(7):900–5.
Kershner R, Fitzsimmons W. Relationship of FK506 whole blood concentrations and efficacy and toxicity after liver and kidney transplantation. Transplantation. 1996;62(7):920–6.
Saint-Marcoux F, Woillard J, Jurado C, et al. Lessons from routine dose adjustment of tacrolimus in renal transplant patients based on global exposure. Ther Drug Monit. 2013;35(3):322–7.
Venkataramanan R, Shaw L, Sarkozi L, et al. Clinical utility of monitoring tacrolimus blood concentrations in liver transplant patients. J Clin Pharmacol. 2001;41:542–51.
Bottiger Y, Brattstrom C, Tyden G, et al. Tacrolimus whole blood concentrations correlate closely to side-effects in renal transplant recipients. Br J Clin Pharmacol. 1998;48:4445–8.
Staatz C, Taylor P, Tett S. Low tacrolimus concentrations and increased risk of early acute rejection in adult renal transplantation. Nephrol Dial Transplant. 2001;16:1905–9.
Undre N, Hooff J, Christiaans M, et al. Low systemic exposure to tacrolimus correlates with acute rejection. Transplant Proc. 1999;31:296–8.
Daher Abdi Z, Premaud A, Essig M, et al. Exposure to mycophenolic acid better predicts immunosuppressive efficacy than exposure to calcineurin inhibitors in renal transplant patients. Clin Pharmacol Ther. 2014;96(4):508–15.
Bouamar R, Shuker N, Hesselink DA, et al. Tacrolimus predose concentrations do not predict the risk of acute rejection after renal transplantation: a pooled analysis from three randomized-controlled clinical trials(dagger). Am J Transplant. 2013;13(5):1253–61.
Knoop C, Thiry P, Saint-Marcoux F, et al. Tacrolimus pharmacokinetics and dose monitoring after lung transplantation for cystic fibrosis and other conditions. Am J Transplant. 2005;5(6):1477–82.
Wong K, Shek C, Chau K, et al. Abbreviated tacrolimus area-under-the-curve monitoring for renal transplant recipients. Am J Kidney Dis. 2000;35(4):660–6.
Ting L, Villeneuve E, Ensom M. Beyond cyclosporine: a systematic review of limited sampling strategies for other immunosuppressants. Ther Drug Monit. 2006;28(3):419–30.
van Der meer A, Marcus M, Touw D, et al. Optimal sampling strategy development methodology using Maximum A Posteriori Bayesian estimation. Ther Drug Monit. 2011;33:133–46.
Staatz C, Tett S. Maximum A Posteriori Bayesian estimation of mycophenolic acid area under the concentration-time curve: is this clinically useful for dosage prediction yet? Clin Pharmacokinet. 2011;50(12):759–72.
Beal A, Sheiner LB, Boeckmann A, et al. NONMEM user’s guides (1989–2009). Ellicott City: Icon Development Solutions; 2009.
Storset E, Holford N, Midtvedt K, et al. Importance of hematocrit for a tacrolimus target concentration strategy. Eur J Clin Pharmacol. 2014;70(1):65–77.
Andrews LM, Riva N, de Winter BC, et al. Dosing algorithms for initiation of immunosuppressive drugs in solid organ transplant recipients. Expert Opin Drug Metab Toxicol. 2015;11(6):921–36.
Woillard J, de Winter BC, Kamar N, et al. Population pharmacokinetic model and Bayesian estimator for two tacrolimus formulations–twice daily Prograf and once daily Advagraf. Br J Clin Pharmacol. 2011;71(3):391–402.
Musuamba F, Mourad M, Haufroid V, et al. Statistical tools for dose individualisation of mycophenolic acid and tacrolimus co-administered during the first month after renal transplantation. Br J Clin Pharmacol. 2013;75(5):1277–88.
Musuamba F, Mourad M, Haufroid V, et al. A simultaneous d-optimal designed study for population pharmacokinetic analyses of mycophenolic acid and tacrolimus early after renal transplantation. J Clin Pharmacol. 2012;52(12):1833–43.
Zhang JLS, Xue L, Ding X, Zhang H, Miao L. The genetic polymorphisms of POR*28 and CYP3A5*3 significantly influence the pharmacokinetics of tacrolimus in Chinese renal transplant recipients. Int J Clin Pharmacol Ther. 2015;53(9):728–36.
Antignac M, Hulot JS, Boleslawski E, et al. Population pharmacokinetics of tacrolimus in full liver transplant patients: modelling of the post-operative clearance. Eur J Clin Pharmacol. 2005;61(5–6):409–16.
Lee J, Hahn H, Son I, et al. Factors affecting the apparent clearance of tacrolimus in korean adult liver transplant recipients. Pharmacotherapy. 2006;26(8):1069–77.
Zhu L, Yang J, Zhang Y, et al. Effects of CYP3A5 genotypes, ABCB1 C3435T and G2677T/A polymorphism on pharmacokinetics of tacrolimus in Chinese adult liver transplant patients. Xenobiotica. 2015;45(9):840–6.
Jalil M, Hawwa AF, McKiernan PJ, et al. Population pharmacokinetic and pharmacogenetic analysis of tacrolimus in paediatric liver transplant patients. Br J Clin Pharmacol. 2014;77(1):130–40.
Kassir N, Labbé L, Delaloye J, et al. Population pharmacokinetics and Bayesian estimation of tacrolimus exposure in paediatric liver transplant recipients. Br J Clin Pharmacol. 2014;77(6):1051–63.
Saint-Marcoux F, Knoop C, Debord J, et al. Pharmacokinetic study of tacrolimus in cystic fibrosis and non-cystic fibrosis lung transplant patients and design of Bayesian estimators using limited sampling strategies. Clin Pharmacokinet. 2005;44(12):1317–28.
Kuypers D, Claes K, Evenepoel P, et al. Time-related clinical determinants of long-term tacrolimus pharmacokinetics in combination therapy with mycophenolic acid and corticosteroids—a prospective study in one hundred de novo renal transplant recipients. Clin Pharmacokinet. 2004;43(11):741–62.
de Jonge H, Vanhove T, de Loor H, et al. Progressive decline in tacrolimus clearance after renal transplantation is partially explained by decreasing CYP3A4 activity and increasing haematocrit. Br J Clin Pharmacol. 2015;80(3):548–59.
Jacobson PA, Schladt D, Oetting WS, et al. Lower calcineurin inhibitor doses in older compared to younger kidney transplant recipients yield similar troughs. Am J Transplant. 2012;12(12):3326–36.
Glowacki F, Lionet A, Buob D, et al. CYP3A5 and ABCB1 polymorphisms in donor and recipient: impact on tacrolimus dose requirements and clinical outcome after renal transplantation. Nephrol Dial Transplant. 2011;26(9):3046–50.
Barbarino J, Staatz C, Venkataramanan R, et al. PharmGKB summary: cyclosporine and tacrolimus pathways. Pharmacogenet Genomics. 2013;23(10):563–85.
Kurzawski M, Droździk M. Pharmacogenetics in solid organ transplantation: genes involved in mechanism of action and pharmacokinetics of immunosuppressive drugs. Pharmacogenomics. 2013;14(9):1099–118.
Picard N, Marquet P. The influence of pharmacogenetics and cofactors on clinical outcomes in kidney transplantation. Expert Opin Drug Metab Toxicol. 2011;7(6):731–43.
Dessilly G, Elens L, Panin N, et al. ABCB1 1199G>A genetic polymorphism (Rs2229109) influences the intracellular accumulation of tacrolimus in HEK293 and K562 recombinant cell lines. PLoS One. 2014;9(3):e91555.
Monostory K, Toth K, Kiss A, et al. Personalizing initial calcineurin inhibitor dosing by adjusting to donor CYP3A-status in liver transplant patients. Br J Clin Pharmacol. 2015;80(6):1429–37.
Pulk R, Schlad D, Oetting W, et al. Multigene predictors of tacrolimus exposure in kidney transplant recipients. Pharmacogenomics. 2015;16(8):841–54.
Boivin A, Cardinal H, Barama A, et al. Influence of SLCO1B3 genetic variations on tacrolimus pharmacokinetics in renal transplant recipients. Drug Metab Pharmacokinet. 2013;28(3):274–7.
Thervet E, Anglicheau D, Legendre C, et al. Role of pharmacogenetics of immunosuppressive drugs in organ transplantation. Ther Drug Monit. 2008;30:143–50.
Thervet E, Loriot MA, Barbier S, Buchler M, Ficheux M, Choukroun G, et al. Optimization of initial tacrolimus dose using pharmacogenetic testing. Clin Pharmacol Ther. 2010;87(6):721–6.
Shuker N, Bouamar R, van Schaik RH, et al. A randomized controlled trial comparing the efficacy of CYP3A5 genotype-based with bodyweight-based tacrolimus dosing after living donor kidney transplantation. Am J Transplant. 2015;. doi:10.1111/ajt.13691 (Epub 2015 Dec 29).
Holford N, Buclin T. Safe and effective variability—a criterion for dose individualization. Ther Drug Monit. 2012;34(5):565–8.
Ekberg H, Bernasconi C, Tedesco-Silva H, et al. Calcineurin inhibitor minimization in the Symphony study: observational results 3 years after transplantation. Am J Transplant. 2009;9(8):1876–85.
van Lent-Evers N, Mathot R, Geus W, et al. Impact of goal-orientated and model-based clinical pharmacokinetic dosing of aminoglycosides on clinical outcome: a cost-effectiveness analysis. Ther Drug Monit. 1999;21(1):63–73.
Evans W, Relling MV, Rodman J, et al. Conventional compared with individualised chemotherapy for childhood acute lymphoblastic leukaemia. N Engl J Med. 1998;338(8):499–505.
Storset E, Asberg A, Skauby M, et al. Improved tacrolimus target concentration achievement using computerized dosing in renal transplant recipients-a prospective, randomized study. Transplantation. 2015;99:2158–66.
Boots J, Van Duijnhoven E, Christiaans M, et al. Glucose metabolism in renal transplant recipients on tacrolimus: the effect of steroid withdrawal and tacrolimus trough level reduction. J Am Soc Nephrol. 2002;13:221–7.
Cosio F, Amer H, Grande JP, et al. Comparison of low versus high tacrolimus levels in kidney transplantation: assessment of efficacy by protocol biopsies. Transplantation. 2007;83(4):411–6.
Lemaitre F, Blanchet B, Latournerie M, et al. Pharmacokinetics and pharmacodynamics of tacrolimus in liver transplant recipients: inside the white blood cells. Clin Biochem. 2015;48(6):406–11.
Barraclough K, Staatz C, Isbel N. Pharmacokinetic, pharmacogenetic and pharmacodynamic monitoring of tacrolimus, mycophenolate and oral corticosteroids in adult kidney transplantation. In: Lim WH, editor. The complex evolution of kidney transplantation: pre-transplant donor and recipient assessment, transplant surgery, immunosuppression, high risk transplants and management of post transplant complications. Organ Transplant Research Horizons; Renal and Urologic Disorders. New York: Nova Science Publisher; 2014. p. 185–261.
Fuchs A, Csajka C, Thoma Y, et al. Benchmarking therapeutic drug monitoring software: a review of available computer tools. Clin Pharmacokinet. 2013;52(1):9–22.
Pharmacology LUHLo. ImmunoSuppressants Bayesian dose Adjustment (ISBA). Limoges; 2005. https://pharmaco.chu-limoges.fr. Accessed 4 Apr 2016.
Barraclough K, Isbel NM, Kirkpatrick CM, et al. Evaluation of limited sampling methods for estimation of tacrolimus exposure in adult kidney transplant recipients. Br J Clin Pharmacol. 2011;71(2):207–23.
Benkali K, Premaud A, Picard N, et al. Tacrolimus population pharmacokinetic-pharmacogenetic analysis and Bayesian estimation in renal transplant recipients. Clin Pharmacokinet. 2009;48(12):805–16.
Benkali K, Rostaing L, Premaud A, et al. Population pharmacokinetics and Bayesian estimation of tacrolimus exposure in renal transplant recipients on a new once-daily formulation. Clin Pharmacokinet. 2010;49(10):683–92.
Antignac M, Barrou B, Farinotti R, et al. Population pharmacokinetics and bioavailability of tacrolimus in kidney transplant patients. Br J Clin Pharmacol. 2007;64(6):750–7.
Scholten E, Cremers S, Schoemaker R, et al. AUC-guided dosing of tacrolimus prevents progressive systemic overexposure in renal transplant recipients. Kidney Int. 2005;67:2440–7.
Staatz C, Willis C, Taylor PJ, et al. Population pharmacokinetics of tacrolimus in adult kidney transplant recipients. Clin Pharmacol Ther. 2002;72(6):660–9.
Saint-Marcoux F, Debord J, Undre N, et al. Pharmacokinetic modeling and development of bayesian estimators in kidney transplant patients receiving the tacrolimus once-daily formulation. Ther Drug Monit. 2010;32(2):129–35.
Press R, Ploeger B, den Hartigh J, et al. Explaining variability in tacrolimus pharmacokinetics to optimize early exposure in adult kidney transplant recipients. Ther Drug Monit. 2009;31:187–97.
Saint-Marcoux F, Debord J, Parant F, et al. Development and evaluation of a simulation procedure to take into account various assays for the Bayesian dose adjustment of tacrolimus. Ther Drug Monit. 2011;33:171–7.
Velickovic-Radovanovic R, Catic-Djordjevic A, Milovanovic J, et al. Population pharmacokinetics of tacrolimus in kidney transplant patients. Int J Clin Pharmacol Ther. 2010;48(6):375–82.
Han N, Yun HY, Hong JY, et al. Prediction of the tacrolimus population pharmacokinetic parameters according to CYP3A5 genotype and clinical factors using NONMEM in adult kidney transplant recipients. Eur J Clin Pharmacol. 2013;69(1):53–63.
Ogasawara K, Chitnis S, Gohh R, et al. Multidrug resistance-associated protein 2 (MRP2/ABCC2) haplotypes significantly affect the pharmacokinetics of tacrolimus in kidney transplant recipients. Clin Pharmacokinet. 2013;52(9):751–62.
Zuo X, Ng CM, Barrett JS, et al. Effects of CYP3A4 and CYP3A5 polymorphisms on tacrolimus pharmacokinetics in Chinese adult renal transplant recipients: a population pharmacokinetic analysis. Pharmacogenet Genom. 2013;23(5):251–61.
Andreu F, Colom H, Grinyo J, et al. Development of a population PK model of tacrolimus for adaptive dosage control in stable kidney transplant patients. Ther Drug Monit. 2015;37(2):246–55.
Bergmann T, Hennig S, Barraclough K, et al. Population pharmacokinetics of tacrolimus in adult kidney transplant patients: impact of CYP3A5 genotype on starting dose. Ther Drug Monit. 2014;36:62–70.
Asberg A, Midtvedt K, van Guilder M, et al. Inclusion of CYP3A5 genotyping in a nonparametric population model improves dosing of tacrolimus early after transplantation. Transplant Int. 2013;26(12):1198–207.
Golubovic B, Vucicevic K, Radivojevic D, et al. Total plasma protein effect on tacrolimus elimination in kidney transplant patients–population pharmacokinetic approach. Eur J Pharm Sci. 2014;52:34–40.
Han N, Ha S, Yun H, et al. Population pharmacokinetic–pharmacogenetic model of tacrolimus in the early period after kidney transplantation. Basic Clin Pharmacol Toxicol. 2014;114:400–6.
Zhao W, Elie V, Roussey G, et al. Population pharmacokinetics and pharmacogenetics of tacrolimus in de novo pediatric kidney transplant recipients. Clin Pharmacol Ther. 2009;86(6):609–18.
Zhao W, Fakhoury M, Baudouin V, et al. Population pharmacokinetics and pharmacogenetics of once daily prolonged-release formulation of tacrolimus in pediatric and adolescent kidney transplant recipients. Eur J Clin Pharmacol. 2013;69(2):189–95.
Jacobo-Cabral C, Garcia-Roca P, Romero-Tejeda EM, et al. Population pharmacokinetic analysis of tacrolimus in Mexican paediatric renal transplant patients: role of CYP3A5 genotype and formulation. Br J Clin Pharmacol. 2015;80(4):630–41.
Staatz C, Willis C, Taylor PJ, et al. Toward better outcomes with tacrolimus therapy: population pharmacokinetics and individualized dosage prediction in adult liver transplantation. Liver Transpl. 2003;9(2):130–7.
Macchi-Andanson M, Charpiat B, Jelliffe RW, et al. Failure of traditional trough levels to predict tacrolimus concentrations. Ther Drug Monit. 2001;23(2):129–33.
Fukatsu S, Yano I, Igarashi T, et al. Population pharmacokinetics of tacrolimus in adult recipients receiving living-donor liver transplantation. Eur J Clin Pharmacol. 2001;57(6–7):479–84.
Fukudo M, Yano I, Fukatsu S, et al. Forecasting of blood tacrolimus concentrations based on the Bayesian method in adult patients receiving living-donor liver transplantation. Clin Pharmacokinet. 2003;42(13):1161.
Zahir H, McLachlan A, Nelson A, et al. Population pharmacokinetic estimation of tacrolimus apparent clearance in adult liver transplant recipients. Ther Drug Monit. 2005;27:422–30.
Li D, Lu W, Zhua J, et al. Population pharmacokinetics of tacrolimus and CYP3A5, MDR1 and IL-10 polymorphisms in adult liver transplant patients. J Clin Pharm Ther. 2007;32:505–15.
Blanchet B, Duvoux C, Costentin C, et al. Pharmacokinetic-pharmacodynamic assessment of tacrolimus in liver-transplant recipients during the early post-transplantation period. Ther Drug Monit. 2008;30:412–8.
Zhang X, Wang Z, Fan J, et al. The impact of sulfonylureas on tacrolimus apparent clearance revealed by a population pharmacokinetics analysis in Chinese adult liver-transplant patients. Ther Drug Monit. 2012;34:126–33.
Oteo I, Lukas JC, Leal N, et al. Tacrolimus pharmacokinetics in the early post-liver transplantation period and clinical applicability via Bayesian prediction. Eur J Clin Pharmacol. 2013;69(1):65–74.
Valdivieso N, Oteo I, Valdivieso A, et al. Tacrolimus dose individualization in “de novo” patients after 10 years of experience in liver transplantation: pharmacokinetic considerations and patient pathophysiology. Int J Clin Pharmacol Ther. 2013;51(7):606–14.
Sam W, Tham L, Holmes M, et al. Population pharmacokinetics of tacrolimus in whole blood and plasma in Asian liver transplant patients. Clin Pharmacokinet. 2006;45(1):59–75.
Zhu L, Wang H, Sun X, et al. The population pharmacokinetic models of tacrolimus in Chinese adult liver transplantation patients. J Pharm. 2014;2014:713650. doi:10.1155/2014/713650
Moes DJ, van der Bent SA, Swen JJ, et al. Population pharmacokinetics and pharmacogenetics of once daily tacrolimus formulation in stable liver transplant recipients. Eur J Clin Pharmacol. 2016;72(2):163–74.
Lu YX, Su QH, Wu KH, et al. A population pharmacokinetic study of tacrolimus in healthy Chinese volunteers and liver transplant patients. Acta Pharmacol Sin. 2015;36(2):281–8.
Wallin J, Bergstrand M, Wilczek HE, et al. Population pharmacokinetics of tacrolimus in pediatric liver transplantation: early posttransplantation clearance. Ther Drug Monit. 2011;33(6):663–72.
Staatz C, Taylor P, Lynch S, et al. Population pharmacokinetics of tacrolimus in children who receive cut-down or full liver transplants. Transplantation. 2001;72(6):1051–61.
Sam W, Aw M, Quak H, et al. Population pharmacokinetics of tacrolimus in Asian paediatric liver transplant patients. Br J Clin Pharmacol. 2000;50:531–41.
Sanchez M, Manzanares C, Santos-Buelga D, et al. Covariate effects on the apparent clearance of tacrolimus in paediatric liver transplant patients undergoing conversion therapy. Clin Pharmacokinet. 2001;40(1):63–71.
Yasuhara M, Hashida T, Toraguchi M, et al. Pharmacokinetics and pharmacodynamics of FK 506 in pediatric patients receiving living-related donor liver transplantations. Transplant Proc. 1995;27(1):1108–10.
Guy-Viterbo V, Scohy A, Verbeeck RK, et al. Population pharmacokinetic analysis of tacrolimus in the first year after pediatric liver transplantation. Eur J Clin Pharmacol. 2013;69(8):1533–42.
Fukudo M, Yano I, Masuda S, et al. Population pharmacokinetic and pharmacogenomic analysis of tacrolimus in pediatric living-donor liver transplant recipients. Clin Pharmacol Ther. 2006;80(4):331–45.
Guy-Viterbo V, Baudet H, Elens L, et al. Influence of donor–recipient CYP3A4/5 genotypes, age and fluconazole on tacrolimus pharmacokinetics in pediatric liver transplantation: a population approach. Pharmacogenomics. 2014;15(9):1207–21.
Musuamba F, Guy-Viterbo V, Reding R, et al. Population pharmacokinetic analysis of tacrolimus early after pediatric liver transplantation. Ther Drug Monit. 2014;36(1):54–61.
Yang J, Liao SS, Zhu LQ, et al. Population pharmacokinetic analysis of tacrolimus early after Chinese pediatric liver transplantation. Int J Clin Pharmacol Ther. 2015;53(1):75–83.
Monchaud C, de Winter BC, Knoop C, et al. Population pharmacokinetic modelling and design of a bayesian estimator for therapeutic drug monitoring of tacrolimus in lung transplantation. Clin Pharmacokinet. 2012;51(3):175–86.
Op den Buijsch R, van de Plas A, Stolk L, et al. Evaluation of limited sampling strategies for tacrolimus. Eur J Clin Pharmacol. 2007;63(11):1039–44.
Zhao W, Fakhoury M, Baudouin V, et al. Limited sampling strategy for estimating individual exposure of tacrolimus in pediatric kidney transplant patients. Ther Drug Monit. 2011;33(6):681–7.
Zhao W, Maisin A, Baudouin V, et al. Limited sampling strategy using Bayesian estimation for estimating individual exposure of the once-daily prolonged-release formulation of tacrolimus in kidney transplant children. Eur J Clin Pharmacol. 2013;69(5):1181–5.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No sources of funding were used to assist in the preparation of this review. Dr Brooks was a student; Dr Tett, Dr Staatz and Dr Isbel receive salaries from university/hospital sources.
Conflict of interest
Dr Brooks, Dr Tett, Dr Isbel and Dr Staatz have no conflicts of interest that are relevant to the content of this review.
Rights and permissions
About this article
Cite this article
Brooks, E., Tett, S.E., Isbel, N.M. et al. Population Pharmacokinetic Modelling and Bayesian Estimation of Tacrolimus Exposure: Is this Clinically Useful for Dosage Prediction Yet?. Clin Pharmacokinet 55, 1295–1335 (2016). https://doi.org/10.1007/s40262-016-0396-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40262-016-0396-1