Abstract
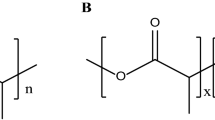
An attempt was made to formulate mesalamine-loaded alginate microspheres by emulsion cross linking method for local treatment of ulcerative colitis. Microspheres were filled in HPMC capsules enteric coated with Eudragit FS-30D. The effect of process variables like drug-alginate concentration ratio and emulsifier concentration was optimized. Microspheres were evaluated for particle size, shape and entrapment efficiency. Spherical shaped microspheres were confirmed by scanning electron microscopy. In vitro drug release study showed a burst drug release pattern in the initial hour necessitating encapsulation of alginate microspheres. In vivo study in rats performed by colonic inflammatory lesions demonstrated remarkable reduction in ulcer index treated with microspheres. Histopathological study confirmed no signs of ulceration or bleeding. Microspheres (drug:alginate concentration, 1:1) was then filled in HPMC capsule shells, enteric coated with Eudragit FS 30D to gain 30 mg weight. Due to solubility of Eudragit FS30D above pH 7, an encapsulated system released alginate microspheres at colon region. Capsules with weight gain of 30 mg coating polymer (B3) resisted the drug release at intestinal region for about 6 h with 10% drug release. In vitro drug release of formulation B3 showed 64.89 and 90.52% drug release within 10 h, in phosphate buffer pH 7.4 and in presence of rat faecal content in simulated colonic fluid, respectively. The later rapid drug release was due to action of colonic microflora at alginate microspheres. Therefore, mesalamine-loaded alginate microspheres enteric coated in HPMC capsules can be potential delivery system for local treatment of ulcerative colitis.





Similar content being viewed by others
References
Ahmed AE, Ahmed A et al (2012) Influence of some formulation variables on the optimization of pH-dependent, colon-targeted, sustained-release mesalamine microspheres. AAPS PharmSciTech 13(1):75–84
Alhnan MA, Basit AW (2011) Engineering polymer blend microparticles: an investigation into the influence of polymer blend distribution and interaction. Eur J Pharm Sci 42(1–2):30–6
Ashford M, Fell J, Attwoods D, Sharma H, Woodhead PJ (1993a) An in vivo investigation into the suitability of pH dependent polymers for colon targeting. Int J Pharm 95:193–199
Ashford M, Fell J, Attwoods D, Woodhead PJ (1993b) An in vitro investigation into the suitability of pH dependent polymers for colon targeting. Int J Pharm 91:241–245
Ashford M, Fell J, Attwoods D, Sharma H, Woodhead PJ (1994) Studies on pectin formulation for colonic drug delivery. J Control Release 30:225–232
Baykara T, Kiliçarslan M (2003) The effect of the drug/polymer ratio on the properties of the verapamil HCl loaded microspheres. Int J Pharm 252:99–109
Bondesen S (1997) Intestinal fate of 5-aminosalicylicacid: regional and systemic kinetic studies in relation to bound nanoparticles for the therapy of inflammatory bowel disease. J Control Release 123:211–218
Cheng CL, Gehrke SH, Ritschel WA (1994) Development of an azopolymer based colonic release capsule for delivering proteins/macromolecules. Methods Find Exp Clin Pharmacol 16(4):271–8
Chourasia MK, Jain SK (2003) Pharmaceutical approaches to colon targeted drug delivery systems. J Pharm Pharm Sci 6(1):33–66
Cole ET et al (2002) Enteric coated HPMC capsules designed to achieve intestinal targeting. Int J Pharm 231:83–95
Das MK, Senapati PC (2008) Furosemide-loaded alginate microspheres prepared by ionic cross-linking technique: morphology and release characteristics Indian. J Pharm Sci 70(1):77–84
Frieri G, Pimpo MT, Palumbo GC, Onori L, Viscido A, Latella G et al (1999) Rectal and colonic mesalazine concentration in ulcerative colitis: oral vs. oral plus topical treatment. Aliment Pharmacol Ther 13:1413–1417
Harder S, Fuhr V, Beerman D, Staib AH (1990) Ciprofloxacin absorption in different regions of human gastrointestinal tract: investigation with HF-capsule. Br J Clin Pharmacol 30:35–39
Jain V, Singh R (2010) Development and characterization of eudragit RS 100 loaded microsponges and its colonic delivery using natural polysaccharides. Acta Pol Pharmaica 67:407–415
Jayaprakash R, Sam T, Mathew (2012) Colon specific drug delivery systems: a review on various pharmaceutical approaches. J Appl Pharm Sci 02(01):163–169
Jitendra J et al (2013) Development and in-vitro evaluation of colon specific satranidazole tablet for the treatment of amoebiasis. Asian J Pharm 7:55–60
Jose K, Dhanya T, Cinu A, Aleykutty NA et al (2010) Multiparticulate system for colon targeted delivery of ondansetron. Indian J Pharm Sci 72(1):58–64
Khan AKA, Piris J, Truelove SC (1977) An experiment to determine the active therapeutic moiety of sulphasalazine. Lancet 2(8044):892–5
Klotz U, Schwab M (2005) Topical delivery of therapeutic agents in the treatment of inflammatory bowel disease. Adv Drug Deliv Rev 57:267–279
Kopeckova P, Rathi R, Takada S, Rihova B, Berenson MM, Kopeck J (1994) Bioadhesive N-(2-hydroxypropyl) methacrylamide copolymers for colon—specific drug delivery. J Control Release 28:211–222
Krishanaiah YS, Satyanarayan S, Prasad YV (1999) Studies of guar gum compression-coated 5 aminosalicylic acid tablets for colon specific drug delivery. Drug Dev Ind Pharm 2:651–657
Kshirsagar SJ et al (2009) In vitro In vivo comparison of two pH sensitive Eudragit polymers for colon specific drug delivery. J Pharm Sci Res 1(4):61–70
Lemoine D, Wauters F et al (1998) Preparation and characterization of alginate microsphere containing a model antigen. Int J Pharm 176:9–19
MacPherson B, Pfeiffer CJ (1976) Experimental colitis. Digestion 14(5–6):424–452
Martin DV (2000) clinical pharmacokinetics of slow release mesalazine. Clin Pharmacokinet 39(2):85–97
Mehta R, Chawla A, Sharma P, Pawar P (2013) Formulation and in vitro evaluation of Eudragit S-100 coated naproxen matrix tablets for colon-targeted drug delivery system. J Adv Pharm Technol Res 4(1):31–41
Morishita I, Morishita M, Takayania K, Machida Y, Nagai T (1993) Enteral insulin delivery by microsphere in three different formulations using Eudragit L-100 and S-100. Int J Pharm 91:29
Morris GP (1989) Hapten-induced model of chronic inflammation and ulceration in the rat colon. Gastroenterology 96(3):795–803
Nagpal M et al (2012) Formulation development and evaluation of alginate microspheres of ibuprofen. J Young Pharm 4(1):13–16
Pandit A, Mathur VB (2014) Formulation and development of two-compartment hpmc capsule for concurrent administration of drugs. Pharma Innov J 2(12):79–83
Pfister G, Bahadir M, Korte F (1986). Release characteristics of herbicide from calcium alginate formulations. J Control Release 3:229–233
Plaizier VJ, Van Molle M, Steppe K, Cherrette ´I. (1992) Enteric coating properties of Eudragit® Aquateric® and cellulose acetate trimellitate applied to capsules. Eur J Pharm Biopharm 38:145–149
Pozzi F, Furlani P, Gazzaniga A, Wilding IR, Davis SS (1994) The time-clock system: a new oral dosage form for fast and complete release of drug after a predetermined lag time. J Control release 31:99–108
Riley SA, Mani V, Goodman MJ, Herd ME, Dutt S, Turnberg LA (1988) Comparison of delayed release 5-aminosalicylic acid (mesalazine) and sulfasalazine in the treatment of mild to moderate ulcerative colitis relapse. Gut 29:669–674
Rubinstein A, Nakar D, Sintov A (1992) Colonic drug delivery: enhanced release of indomethacin from cross-linked chondroitin matrix in rat cecal content. Pharm Res 9:276–278
Rubinstein A, Radai M, Ezra M, Pathak S, Rokem JS (1993) In-vitro evaluation of calcium pectinate: a potential colon specific drug delivery carrier. Pharm Res 10:258–263
Sawarkar SP, Deshpande SG, Bajaj AN, Nikam VS (2015) In vivo evaluation of mesalamine colon-specific tablets using experimental-induced colitis rat animal model. AAPS PharmSciTech 16(6):1445–1454
Shukla S, Jain D et al (2010) Formulation and in vitro characterization of alginate microspheres loaded with diloxanide furoate for colon- specific drug delivery. Asian J Pharm 4:199–204
Tenjarla S. (2015) Dissolution of commercially available mesalamine formulations at various pH levels. Drugs in R&D 15(2):211–215
Tozaki H, Emi Y, Harisaka E, Fujitu P, Yamamoto A, Muranishi S (1997) Degradation of insulin and calcitonin and their protection by various protease inhibitors in rat caecal contents Implication in peptide delivery to the colon. J Pharm Pharmacol 49:164–168
Ueda S, Hata T, Asakura, Yamaguchi H, Kotani M et al (1994) Development of a novel drug release system, time-controlled explosion system (TES). Concept and design. J Drug Target 2:35–44
Ura J, Shirachi D, Ferrill M (1992) The chronotherapeutic approach to pharmaceutical treatment. Calif Pharm 23:46–53
Van den Mooter G, Samyn C, Kinget R (1994) The relation between swelling properties and enzymatic degradation of azo polymers designed for colon-specific drug delivery. Pharm Res 11:1737–1741
Van den Mooter G, Samyn C, Kinget R (1995) In –vivo evaluation of colon specific drug delivery system: an absorption study of theophylline from capsule coated with azo polymer in rats. Pharm Res 12:244–247
Varshosaz J, Dehkordi AJ, Golanfshan S (2006) Colon- specific delivery of mesalazine chitosan microspheres. J Microencapsul 23:329–39
Yotsuyanagi T, Ohkubo T, Ohhashi T, Ikeda K. (1987) Calcium-induced gelatin of alginic acid and pH-sensitive reswelling of dried gels. Chem Pharm Bull 35:1555-1563
Zingarelli B, Szabó C et al (1999) Reduced oxidative and nitrosative damage in murine experimental colitis in the absence of inducible nitric oxide synthase Inflammation and inflammatory bowel disease Gut 45:199–209
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors do not have conflict of interest.
Rights and permissions
About this article
Cite this article
Patole, V.C., Pandit, A.P. Mesalamine-loaded alginate microspheres filled in enteric coated HPMC capsules for local treatment of ulcerative colitis: in vitro and in vivo characterization. J. Pharm. Investig. 48, 257–267 (2018). https://doi.org/10.1007/s40005-017-0304-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40005-017-0304-1