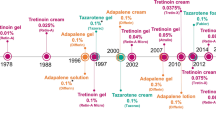
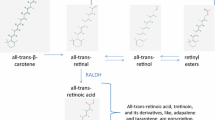
Abstract
Vitamin A and its derivatives have long been purported to support healthy skin. This has naturally led to their appearance in over-the-counter (OTC) products that fall under the umbrella term “cosmeceuticals,” a term used to describe OTC cosmetic products with biologically active ingredients. The lure of these products includes their accessibility and the claims made by marketing campaigns that tap into the psyche of the consumer. Compared to prescription retinoids, however, the scientific evidence behind the efficacy of retinol-containing cosmeceuticals is less abundant. Here, we review the relevant scientific data to date regarding the use of retinols and retinoids for photoaging, dyspigmentation, and acne.



Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Kuenzli S, Saurat J. Chapter 127. Retinoids. Bolognia JL, Jorizzo JL, Rapini RP, eds. Dermatology. Elsevier;2003:1991–2006.
Sachs DL, Voorhees JJ. Chapter 8. Vitamin A: retinoids and the treatment of aging Skin. Farris PK editor. Cosmeceuticals and Cosmetic Practice. John Wiley & Sons; 2014. This book chapter provides a broad overview of retinoids, their mechanism of action, and the clinical benefits and adverse effects of both retinoids and retinols.
Gallego O et al. Comparative functional analysis of human medium-chain dehydrogenases, short-chain dehydrogenases/reductases and aldo-keto reductases with retinoids. Biochem J. 2006;399:101–9.
Darlenski R, Surbur C, Fluhr JW. Topical retinoids in the management of photodamaged skin: from theory to evidence-based practical approach. Br J Dermatol. 2010;163(6):1157–65.
American Academy of Dermatology. Cosmeceuticals. 2014. Available at http://www.aad.org/media-resources/stats-and-facts/cosmetic-treatments/cosmeceuticals. Accessed October 2 2014.
FDA: US Food and Drug Administration. Cosmeceutical. 2014. Available at http://www.fda.gov/Cosmetics/Labeling/Claims/ucm127064.htm. Accessed October 2 2014.
Sorg O, Antille C, Kaya G, Saurat JH. Retinoids in cosmeceuticals. Dermatol Ther. 2006;19:289–96.
Kang S, Duell EA, Fisher GJ, Datta SC, Wang ZQ, Reddy AP, et al. Application of retinol to human skin in vivo induces epidermal hyperplasia and cellular retinoid binding proteins characteristic of retinoic acid but without measurable retinoic acid levels or irritation. J Investig Dermatol. 1995;105(4):549–56.
Ferguson J, Johnson BE. Photosensitivity due to retinoids: clinical and laboratory studies. Br J Dermatol. 1986;115(3):275–83.
Gaspar LR, Tharmann J, Maja Campos PM, Leibsch M. Skin phototoxicity of cosmetic formulations containing photounstable and photostable UV-filters and vitamin A palmitate. Toxicol In Vitro. 2013;27(1):418–25.
Murray J, Potts A. The phototoxic and photoallergy potential of clindamycin phosphate 1.2%/ tretinoin 0.025% gel for facial acne: results of two single-center, evaluator-blinded, randomized, vehicle-controlled phase 1 studies in healthy volunteers. J Drugs Dermatol. 2014;13(1):16–22.
Pham AK, Dinulos JG. Cosmeceuticals for children: should you care? Curr Opin Pediatr. 2014;26(4):446–51.
Busse K, Koo J. Introducing the delayed retinoid burn: a case report and discussion of this potential risk of retinoid-phototherapy combination management. J Am Acad Dermatol. 2011;64(5):1011–2.
Hubbard BA, Unger JG, Rohrich RJ. Reversal of skin aging with topical retinoids. Plast Reconstr Surg. 2014;133(4):481e–90. This recent review explores the biological processes of skin aging and evaluates the efficacy of various topical therapies with emphasis on evidence-based clinical outcomes of topical retinoid and retinol therapy.
Ho ET et al. A randomized, double-blind, controlled comparative trial of the anti-aging properties of non-prescription tri-retinol 1.1 % vs. prescription tretinoin 0.025 %. J Drugs Dermatol. 2012;11(1):64–9.
Green C, Orchard G, Cerio R, et al. A clinicopathological study of the effects of topical retinyl propionate cream in skin photoageing. Clin Exp Dermatol. 1998;23:162–7.
Han H-S, Kwon Y-J, Park M-S, et al. Efficacy validation of synthesized retinol derivatives in vitro: stability, toxicity, and activity. Bioorg Med Chem. 2003;11:3839–45.
Lee MS et al. A newly synthesized photostable retinol derivative (retinyl N-formyl aspartamate) for photodamaged skin: profilometric evaluation of a 24-wks study. J Am Acad Dermatol. 2006;55:220–4.
Draelos Z, Dahl A, Yatskayer M, Chen N, Krol Y, Oresajo C. Dyspigmentation, skin physiology, and a novel approach to skin lightening. J Cosmet Dermatol. 2013;12(4):247–53.
Rivas S, Pandya AG. Treatment of melasma with topical agents, peels and lasers: an evidence-based review. Am J Clin Dermatol. 2013;14(5):359–76.
Sorg O et al. The potential depigmenting activity of retinaldehyde. Dermatology. 2013;227:231–7. This study evaluated the efficacy of retinaldehyde as a depigmenting agent with evaluation of its melanocytic effects in animal and human epidermis.
Pechere M et al. Antibacterial activity of retinaldehyde against Propionibacterium acnes. Dermatology. 1999;199 Suppl 1:29–31.
Pechere M et al. The antibacterial activity of topical retinoids: the case of retinaldehyde. Dermatology. 2002;205:153–8.
Bowe WP, Shalita AR. Effective over-the-counter acne treatments. Semin Cutan Med Surg. 2008;27(3):170–6.
Draelos ZD, Shalita AR, Thiboutot D, Oresajo C, Yatskayer M, Raab S. A multicenter, double-blind study to evaluate the efficacy and safety of 2 treatments in participants with mild to moderate acne vulgaris. Cutis. 2012;89(6):287–93.
Rouss S. Prevention. Your guide to retinol and retinoids. Prevention. 2011. Available at http://www.prevention.com/beauty/beauty/best-anti-aging-product-retinoids-vitamin. Accessed October 11 2014.
Thornfeldt C. Cosmeceuticals containing herbs: fact, fiction, and future. Dermatol Surg. 2005;31:873–80.
Bellemere G. Ethnic patterns in response to retinol: an ex vivo study. J Am Acad Dermatol. 2010;62(3,S1):AB25.
Teixeira Z, Dreiss CA, Lawrence MJ, Heenan RK, Machado D, Justo GZ, et al. Retinyl palmitate polymeric nanocapsules as carriers of bioactives. J Colloid Interface Sci. 2012;382(1):36–47.
Gold M et al. Open label study of 1% retinol and double-blind placebo-controlled study of 0.5% retinol both in an oil-free aqueous protein rich suspension. J Am Acad Dermatol. 2013;68(4,Supp1):AB25.
Hougeir FG, Kircik L. A review of delivery systems in cosmetics. Dermatol Ther. 2012;25:234–7. This review discusses some of the most popular delivery systems used in cosmetic products.
Jain AK, Jain A, Garg NK, Agarwal A, Jain A, Jain SA, et al. Adapalene loaded solid lipid nanoparticles gel: an effective approach for acne treatment. Colloids Surf B: Biointerfaces. 2014;121:222–9.
Bhatia G, Zhou Y, Banga AK. Adapalene microemulsion for transfollicular drug delivery. J Pharm Sci. 2013;102:2622–31.
Iida S, Takushima A, Ohura N, Sato S, Kurita M, Harii K. Silicone sheet containing all-trans retinoic acid and hydroquinone for the treatment of epidermal melanosis. Dermatol Surg. 2013;39(8):1237–42.
Compliance with Ethics Guidelines
Conflict of Interest
Shauna Higgins and Naissan O. Wesley declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Cosmetic Dermatology
Rights and permissions
About this article
Cite this article
Higgins, S., Wesley, N.O. Topical Retinoids and Cosmeceuticals: Where Is the Scientific Evidence to Recommend Products to Patients?. Curr Derm Rep 4, 56–62 (2015). https://doi.org/10.1007/s13671-015-0102-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13671-015-0102-9